Abstract
The American Cancer Society (ACS) recommends at least 150 min of moderate intensity physical activity per week, alcohol intake of ≤1 drink per day, and maintaining a body mass index (BMI) of <25 kg/m2 for breast cancer prevention. Adherence to these guidelines has been linked to lower overall mortality in average-risk populations, it is not known if mortality reduction extends to women at higher risk given their family history of breast cancer. We followed 2,905 women from a high-risk Breast Cancer Family Registry in New York, of which 77 % were white non-Hispanic and 23 % were Hispanic. We collected information on BMI, physical activity, and alcohol intake at baseline and prospectively followed our cohort for outcomes based on questionnaires and National Death Index linkage. We used Cox regression to examine the relation between adherence to ACS guidelines and overall mortality and examined effect modification by race, age, and BRCA status. There were 312 deaths after an average of 9.2 ± 4.1 years of follow-up. Adherence to all three ACS recommendations was associated with 44–53 % lower mortality in women unaffected with breast cancer at baseline [Hazard Ratio (HR) 0.56, 95 % CI (0.33–0.93)] and in women affected with breast cancer at baseline [HR 0.47, 95 % CI (0.30–0.74)]. These associations remained after stratification by age, race, and BRCA status {e.g., BRCA1 and/or BRCA2 carriers [HR 0.39, 95 % CI (0.16–0.97)]}. These results support that women at high risk, similar to women at average risk, may also have substantial benefits from maintaining the ACS guidelines.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The American Cancer Society (ACS) recommends health guidelines for breast cancer prevention that include lifestyle behaviors for regular physical activity, moderate alcohol consumption, and maintaining a healthy body mass index (BMI) [1]. The current recommendations for women are as follows: (1) “to be as lean as possible throughout life without being underweight”; (2) “to get at least 150 min of moderate intensity or 75 min of vigorous intensity activity each week (or a combination of these) preferably spread throughout the week”; and (3) “to drink no more than one drink per day” [1].
Epidemiological studies have provided evidence that individuals who adhere to these ACS guidelines and other similar health guidelines on physical activity, alcohol consumption, and body size have better health outcomes including reduced risk of chronic disease, including diabetes, myocardial infarction, stroke, and cancer [2] as well as reduced mortality [3–5]. For example, recent results from a large European cohort consisting of nearly 400,000 participants from the World Cancer Research Fund/American Institute for Cancer Research indicate that there is an 18 % reduction in incidence of total cancers [6] and a 34 % reduction in mortality when comparing those in the highest group of adherence to the lowest group [7]. Similarly, in the U.S. in an ACS cohort of 100,000 men and women from 21 different states with population-based cancer registries, those who adhered to ACS cancer prevention guidelines had a 42 % reduction in all-cause mortality and a 24 % reduction in cancer-specific mortality [8]. In addition to mortality reductions, adherence to ACS guidelines has also been associated with lower cancer incidence. For example, in a cohort of over 93,000 postmenopausal women enrolled from 40 U.S. clinical centers, adherence to ACS cancer prevention guidelines was associated with a 22 % reduction in the incidence of breast cancer, a 52 % reduction of colorectal cancer incidence, and a 27 % reduction in endometrial cancer incidence [9]. Women who adhered to cancer prevention guidelines had a 27 % reduction in all-cause mortality, a 61 % lower risk of colorectal cancer death, and a 33 % lower risk of death from breast cancer [9].
These cohorts suggest a large unrealized potential for cancer prevention in populations at average risk of cancer. Family history of cancer dramatically increases risk of most cancers including breast cancer, of which only a portion is explained by mutations in BRCA1 and BRCA2 genes [10] and other highly penetrant genes [11–13]. Despite the increased risk in many cancers from a family history of cancer, some studies suggest that family members may not adhere to cancer prevention guidelines. For example, the largest U.S.-based family cohort, the Sister Study [14], found that only 12 % of women with a sister affected with breast cancer adhere to all three guidelines [15]. Further, the Sister Study found that there was substantial heterogeneity in meeting these guidelines by women of different racial and ethnic groups [15]. Specifically, they found that only 12.7 % of white women and 4.5 % of black women met all three guidelines (4.5 %) [15].
Given that adherence to cancer prevention guidelines has been shown to substantially reduce mortality in average-risk cohorts (range of 27–42 % reduction in mortality), we investigated whether this reduction in mortality from meeting the ACS breast cancer guidelines holds in higher risk populations in ethnically diverse women from our New York Breast Cancer Family Registry. Specifically, we examined the effect of adhering to ACS Guidelines for breast cancer prevention in the United States, across white and Hispanic women with a family history or personal history of breast cancer. The recommendations include adherence to physical activity (≥150 min per week), alcohol intake (≤1 drink per day), and body mass index guidelines (<25 kg/m2). We evaluated levels of adherence with respect to all-cause mortality.
Materials and methods
The Breast Cancer Family Registry (BCFR), established in 1995, is a collaborative project with participants from six research sites in the USA, Canada, and Australia [16]. For this study, we included participants from the New York site of the BCFR [17–19], a clinic-based site that identified participants along with family members based on the following criteria: (1) a female with either breast or ovarian cancer age 45 or less at diagnosis; (2) a male with a history of breast cancer; (3) a female with a history of both breast and ovarian cancer; (4) a female with one first-degree relative or two second-degree relatives with a history of either breast or ovarian cancer; (5) a family with a known BRCA1 or BRCA2 mutation. We included women who completed a baseline questionnaire, identified themselves as either ‘White/Caucasian’ or ‘Hispanic/Latina’, and for whom height, weight, physical activity, and alcohol intake data were available. 1,000 women were affected with breast cancer at enrollment (815 white, 185 Hispanic) and 1,905 women were unaffected relatives of those with breast cancer (1,433 white, 472 Hispanic). For these analyses, we examined women in two separate cohorts, those who were affected with breast cancer at the time of baseline (n = 1,000) and those who were unaffected women from families with a history of breast cancer (n = 1,905). We inlcuded outcomes through December 31, 2012, the endpoint for the start of analyses. The Institutional Review Board of Columbia University Medical Center approved the study and all participants provided informed consent.
We calculated BMI based on self-reported height and weight at baseline [weight (kg)/height (m)2] and categorized BMI according to ACS guidelines (<25 or ≥25 kg/m2). We asked women to report their average hours per week of moderate physical activity (e.g., brisk walking, golf, volleyball, cycling on level streets, recreation tennis, or softball) and strenuous exercise (e.g., swimming laps, aerobics, calisthenics, running, jogging, basketball, cycling on hills, racquetball) from the previous three years. We categorized physical activity (≥150 or <150 min of moderate or vigorous activity/week) to assess adherence to ACS guidelines. Alcohol consumption per week in the last 3 years was self-reported and included details regarding beer, wine or wine coolers, and hard liquor consumption. We categorized alcohol consumption according to ACS guidelines (≤1 or >1 serving/day). We evaluated confounding based on data from the baseline questionnaire including education (<high school, high school or vocational tech, some college or graduate), age (continuous), smoking status (never, former, current), history of mammogram (yes, no), and hormone replacement therapy (HRT) use (never, former, current) at baseline. We calculated the number of first- and second-degree family members with breast cancer from self and relative reported family history. We obtained vital status through self or relative report at follow-up and matching of participants to the National Death Index (NDI). In the analyses, we considered NDI-reported deaths if NDI assigned scores that met recommended criteria [20].
We tested the associations between adherence to ACS guidelines and all-cause mortality using Cox proportional hazards models and tested for confounding by age at baseline, education, smoking, number of relatives with breast cancer, mammogram history, and HRT use through a 10 % change in the beta coefficient for the categorical constructs representing adherence. In addition, we assessed interaction with race, age, and BRCA status through stratified analyses and statistical tests of multiplicative interaction. We used adherence to 1 ACS recommendation as the reference due to small samples in the 0 adherence group. We used SAS 9.3 (SAS Institute, Cary, NC) to conduct all statistical analyses.
Results
We included 2,905 women in this study, 2,248 white women and 657 Hispanic women, and followed them for an average of 9.17 ± 4.12 years during which time 312 deaths occurred. The mean age ± SD for white and Hispanic women was 50 ± 15.2 and 45 ± 15.2 years, respectively. Compared to Hispanics, white women were more likely to report some college degree or higher (86.0 vs. 38.1 %), were less likely to report being overweight or obese (37.7 vs. 62.1 %), and were less likely to report smoking or alcohol consumption (52.5 vs. 66.4 % and 45.8 vs. 72.7 %). White women were also more likely to be past or current users of hormone replacement therapy (22.1 vs. 12.9 %). Fewer women in the affected cohort reported never smoking compared to unaffected women (46.4 vs. 56.0 % in Whites and 62.2 vs. 68.0 % in Hispanics). As expected, almost all the women who were affected with breast cancer reported having had a mammogram, though within the unaffected cohort white women were more likely to have had a recent screening mammogram as compared to Hispanic women (81.3 vs. 63.1 %). On average, affected Hispanic women reported less exercise than affected white women (3.7 vs. 4.6 h/week) (Table 1).
Approximately, half the women who were unaffected with breast cancer at the time of enrollment adhered to the ACS guidelines for physical activity and BMI <25 kg/m2. However, white women were more likely to exercise ≥150 min/week (66.8 vs. 55.8 %) and were more likely to have a BMI <25 kg/m2 (64.5 vs. 34.9 %) than Hispanic women. The majority of unaffected women adhered with the alcohol consumption guideline of <1 drink per day, though Hispanic women were more adherent than white women (90.9 vs. 83.3 %). Overall, the majority of women in the unaffected cohort did not adhere to all three of the ACS guidelines, though white women were more likely to report adherence to all three (36.2 vs. 17.6 %; p < 0.001) (Table 2).
Women who were affected with breast cancer at the time of enrollment had moderate adherence to the ACS guidelines for physical activity and BMI <25 kg/m2. However, white women were more likely to exercise ≥150 min/week (62.0 vs. 42.7 %) and were more likely to maintain a BMI <25 kg/m2 (57.8 vs. 34.1 %). White women and Hispanic women who were affected with breast cancer were equally adherent with the alcohol consumption of <1 drink/day (86.0 and 86.0 %). Overall, women affected with breast cancer were not adherent with ACS guidelines. However, white women were more likely to report adherence to all three recommendations (31.8 vs. 12.4 %; p < 0.001) (Table 2).
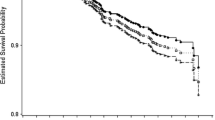
After adjusting for age and education, we observed a 44–53 % reduction in rate of overall mortality in the unaffected (HR 0.56; 95 %CI (0.33–0.93)) and affected groups (HR 0.47; 95 %CI (0.30–0.74) (Table 3). We did not observe confounding by the other factors considered (age at baseline, education, smoking, number of relatives with breast cancer, mammogram history, and HRT use). The inverse associations with adherence and mortality remained after stratification by age but the association was only statistically significant in unaffected women ≥50 years (HR 0.45; 95 % CI (0.24–0.84) and in affected women <50 years [HR 0.19; 95 %CI (0.08–0.47); Fig. 1]. Adherence to all three guidelines was associated with a 27 % reduced mortality 95 % CI (0.28–1.89) in unaffected women <50 years and 32 % reduced mortality 95 % CI (0.41–1.12) in affected women >50 years. The inverse association with adherence remained in all groups except for unaffected Hispanic women, after stratification by race and ethnicity (Table 3). Stratification by BRCA carrier status showed that adherence to all three recommendations was associated with a 61 % lower mortality [HR in carriers = 0.39; 95 % CI (0.16–0.97)]; adherence to all three recommendations in non-carriers was associated with a 50 % lower mortality [HR in non-carriers = 0.50; 95 %CI (0.36–0.70)] (Fig. 2).
Discussion
Women in our cohort who reported adherence to all three ACS guidelines had a 44–53 % reduction in all-cause mortality as compared with women who were only adhered to one guideline. Only 3.0 % of our cohort reported not adhering to any of the three ACS guidelines, which limited separate analysis of this group. These results remained after examining confounding by age at baseline, education, smoking, number of relatives with breast cancer, mammogram history, and HRT use. These results are also consistent with other cohort studies with participants of average risk including the European Prospective Investigation into Cancer and Nutrition (EPIC) [3] where participants who adhered to zero out of four health recommendations had a four-fold increase in all-cause mortality, as well as the Cancer Prevention Study II Nutrition Cohort [8] and the Women’s Health Initiative [9] which showed a 42 and 27 % reduction of all-cause mortality, respectively, when comparing those most adherent to American Cancer Society guidelines to those least adherent.
Women with a first-degree relative with breast cancer have a two-fold increased risk of developing breast cancer in their lifetime [11], which is substantially higher based on the ages of onset of the relatives. Although women with a genetic mutation at the BRCA1 or BRCA2 gene are at substantially higher lifetime risk, only 5–10 % of women with a family history are carriers [21–24]. Women in our cohort are at an overall higher risk than women in the general population. Ten-year breast cancer risks observed in this cohort were 7.03 % for women 35–49 years and 8.49 % for women 50 years and above; 3.1 times that of an average-risk population when comparing to age-standardized SEER rates [25]. Despite having a personal history of breast cancer, only 28.2 % were adherent to all three guidelines for physical activity, maintaining a healthy BMI, and alcohol consumption and only 31.6 % of unaffected women from families at high risk for breast cancer were adherent to all three guidelines. While adherence was low in our cohort, it should be noted that the majority of women reported adherence with the recommendation for one or less alcoholic beverage per day (85 %). The rates of adherence were considerably lower for maintaining a healthy BMI (57 %) and participating in physical activity for at least 150 min each week (60 %). However, differences in reporting of these three constructs may affect some of the adherence as measured.
Adherence was low across all subgroups, although white women were more adherent with all three lifestyle behaviors combined as compared to Hispanic women (36.2 vs. 17.6 % in unaffected women; 31.8 vs. 12.4 % in affected women). This same trend held for each of the individual behaviors except for alcohol consumption. While white and Hispanic women were equally adherent in the affected cohort (86.0 vs. 86.0 %), white women were less adherent than Hispanic women (83.3 vs. 90.9 %) in the unaffected cohort. These results are consistent with other high-risk cohorts. Spector et al. [15] showed that white and black women who are sisters of women with breast cancer had low adherence to recommendations for physical activity (26.4 and 18.2 %, respectively) and maintaining a healthy BMI (42.5 and 16.7 %, respectively). However, black women were more likely to comply with ACS alcohol recommendations [15]. A prospective cohort study by Thomson et al. showed that cancer incidence, all-cause, and cancer-specific mortality were reduced through adherence to a greater number of ACS guidelines which suggests that adherence to ACS recommendations can be beneficial across all subgroups [9]. Furthermore this same cohort [9] showed benefits from adherence in affected women and all-cause mortality across racial and ethnic subgroups; an important finding as many minority populations are often diagnosed at later stages of disease and experience worse outcomes [26–29].
Breast cancer incidence, particularly for cancer with distal involvement is increasing in young women (<40 years) [30]. The increase is seen across all racial and ethnic subgroups but the rate of increase is higher in non-white women [31]. Both Hispanic and black women are more likely to present at a later stage at the time of diagnoses and have higher rates of breast cancer-specific mortality than non-Hispanic white women [32]. The Sister’s Study indicates differences in adherence with cancer prevention guidelines between white and black women [15], and our data indicate that there are similar differences between white and Hispanic women in adherence. Continuing to urge clinicians to advise all patients and their families to adhere to cancer prevention guidelines is an obvious first step. However, our study along with the existing evidence suggests that specific recognition and solutions need to be addressed so that interventions can be targeted to groups with the greatest need.
This large, prospective cohort is focused on women that are higher risk for breast cancer from their family history. Due to smaller numbers than some of the very large cohorts, we were not able to consider cancer-specific mortality. As we relied on self report, it is possible that women reported adherence to the guidelines when in actuality they would not have adhered. This would be non-differential misclassification, however, as all participants from this cohort reported their adherence at baseline without knowing their outcomes. Therefore, associations may be even stronger than we estimated if there are no large unmeasured confounders. Although we were able to consider empirical confounding by a number of factors, as our exposures were not randomized it is possible that the effects observed would be smaller if there are large, unmeasured, positive confounders. Because the data for physical activity, alcohol consumption, and body size were collected at baseline, we were also unable to address whether adherence to these guidelines later in life reduces mortality as it is possible that the women who complied with the guidelines have been engaging in healthy behaviors throughout their life.
For women not already meeting guidelines, clinical trials focused on changing behaviors later in life support the challenge of long-term maintenance of lifestyle changes [33–35]. As research may indicate that behavior changes later in life have a lower probability of being maintained, families at high risk of breast cancer should be informed at an earlier stage in life that making healthy behavioral changes with respect to alcohol consumption, physical activity, and maintaining a healthy weight have positive benefits associated with a reduction in overall mortality [36]. Breast cancer prevention, such as changes in these reported lifestyle behaviors, are most likely to have the greatest impact when initiated earlier in life and maintained over the lifecourse [37].
The magnitude of the associations we observed was as large as or larger than many other primary, secondary, and tertiary breast cancer prevention interventions. For example, mammography screening is associated with a 19 % reduction in mortality with a greater reduction in mortality for screening over age 50 [38, 39]. Chemopreventive medications have also been linked with reduced risk in high-risk women as well as adjuvant treatment for women with estrogen receptor-positive breast cancers in the range of 31–67 % and a reduction in breast cancer mortality of 30 % [40]. Our findings suggest a large potential for reduced mortality from following these guidelines. However, adherence to these behaviors is currently low in the general population [6] as well as in high-risk cohorts [15]. Adherence across high-risk minority groups is even lower [15]. Adherence to chemoprevention and mammography screening, in contrast, is much higher in high-risk cohorts with regular mammography screening and five-year use of chemoprevention of tamoxifen with 40–83 % [41–44] and 41–72 % [45] adherence, respectively. Thus, substantial gains can be made in primary, secondary, and tertiary prevention in all women, and especially those at higher risk, from improving adherence with screening, chemoprevention, and alcohol, physical activity, and body size guidelines.
Conclusion
In summary, we found that among women with a family or personal history of breast cancer, adherence to all three ACS breast cancer prevention guidelines for physical activity, alcohol consumption, and maintaining a healthy weight was associated with a reduction in mortality compared to women who were only adherent to one guideline. Even in white and Hispanic women with a family history of breast cancer, adherence with the three cancer prevention guidelines remained low. Substantial gains can be made in prevention for women, especially those at high risk based on family history, by improving adherence to these lifestyle behaviors.
Abbreviations
- ACS:
-
American Cancer Society
- BCFR:
-
Breast Cancer Family Registry
- BMI:
-
Body mass index
- EPIC:
-
European prospective investigation into cancer and nutrition
- HR:
-
Hazard ratio
- HRT:
-
Hormone replacement therapy
- NDI:
-
National Death Index
References
Kushi LH, Doyle C, McCullough M, Rock CL, Demark-Wahnefried W, Bandera EV, Gapstur S, Patel AV, Andrews K, Gansler T (2012) American Cancer Society Guidelines on nutrition and physical activity for cancer prevention: reducing the risk of cancer with healthy food choices and physical activity. Cancer J Clin 62(1):30–67. doi:10.3322/caac.20140
Ford ES, Bergmann MM, Kroger J, Schienkiewitz A, Weikert C, Boeing H (2009) Healthy living is the best revenge: findings from the European Prospective Investigation Into Cancer and Nutrition-Potsdam study. Arch Intern Med 169(15):1355–1362. doi:10.1001/archinternmed.2009.237
Khaw KT, Wareham N, Bingham S, Welch A, Luben R, Day N (2008) Combined impact of health behaviours and mortality in men and women: the EPIC-Norfolk prospective population study. PLoS Med 5(1):e12. doi:10.1371/journal.pmed.0050012
Knoops KT, de Groot LC, Kromhout D, Perrin AE, Moreiras-Varela O, Menotti A, van Staveren WA (2004) Mediterranean diet, lifestyle factors, and 10-year mortality in elderly European men and women: the HALE project. JAMA 292(12):1433–1439. doi:10.1001/jama.292.12.1433
Cerhan JR, Potter JD, Gilmore JM, Janney CA, Kushi LH, Lazovich D, Anderson KE, Sellers TA, Folsom AR (2004) Adherence to the AICR cancer prevention recommendations and subsequent morbidity and mortality in the Iowa Women’s Health Study cohort. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. Cancer Epidemiol Biomark Prev 13(7):1114–1120
Romaguera D, Vergnaud AC, Peeters PH, van Gils CH, Chan DS, Ferrari P, Romieu I, Jenab M, Slimani N, Clavel-Chapelon F, Fagherazzi G, Perquier F, Kaaks R, Teucher B, Boeing H, von Rusten A, Tjonneland A, Olsen A, Dahm CC, Overvad K, Quiros JR, Gonzalez CA, Sanchez MJ, Navarro C, Barricarte A, Dorronsoro M, Khaw KT, Wareham NJ, Crowe FL, Key TJ, Trichopoulou A, Lagiou P, Bamia C, Masala G, Vineis P, Tumino R, Sieri S, Panico S, May AM, Bueno-de-Mesquita HB, Buchner FL, Wirfalt E, Manjer J, Johansson I, Hallmans G, Skeie G, Benjaminsen Borch K, Parr CL, Riboli E, Norat T (2012) Is concordance with World Cancer Research Fund/American Institute for Cancer Research guidelines for cancer prevention related to subsequent risk of cancer? Results from the EPIC study. Am J Clin Nutr 96(1):150–163. doi:10.3945/ajcn.111.031674
Vergnaud AC, Romaguera D, Peeters PH, van Gils CH, Chan DS, Romieu I, Freisling H, Ferrari P, Clavel-Chapelon F, Fagherazzi G, Dartois L, Li K, Tikk K, Bergmann MM, Boeing H, Tjonneland A, Olsen A, Overvad K, Dahm CC, Redondo ML, Agudo A, Sanchez MJ, Amiano P, Chirlaque MD, Ardanaz E, Khaw KT, Wareham NJ, Crowe F, Trichopoulou A, Orfanos P, Trichopoulos D, Masala G, Sieri S, Tumino R, Vineis P, Panico S, Bueno-de-Mesquita HB, Ros MM, May A, Wirfalt E, Sonestedt E, Johansson I, Hallmans G, Lund E, Weiderpass E, Parr CL, Riboli E, Norat T (2013) Adherence to the World Cancer Research Fund/American Institute for Cancer Research guidelines and risk of death in Europe: results from the European Prospective Investigation into Nutrition and Cancer cohort study 1,4. Am J Clin Nutr 97(5):1107–1120. doi:10.3945/ajcn.112.049569
McCullough ML, Patel AV, Kushi LH, Patel R, Willett WC, Doyle C, Thun MJ, Gapstur SM (2011) Following cancer prevention guidelines reduces risk of cancer, cardiovascular disease, and all-cause mortality. Cancer Epidemiol Biomark Prev 20(6):1089–1097. doi:10.1158/1055-9965.EPI-10-1173
Thomson CA, McCullough ML, Wertheim BC, Chlebowski RT, Martinez ME, Stefanick ML, Rohan TE, Manson JE, Tindle HA, Ockene J, Vitolins MZ, Wactawski-Wende J, Sarto GE, Lane DS, Neuhouser ML (2014) Nutrition and physical activity cancer prevention guidelines, cancer risk, and mortality in the women’s health initiative. Cancer Prev Res (Phila) 7(1):42–53. doi:10.1158/1940-6207.CAPR-13-0258
Claus EB, Schildkraut J, Iversen ES Jr, Berry D, Parmigiani G (1998) Effect of BRCA1 and BRCA2 on the association between breast cancer risk and family history. J Natl Cancer Inst 90(23):1824–1829
Pharoah PD, Day NE, Duffy S, Easton DF, Ponder BA (1997) Family history and the risk of breast cancer: a systematic review and meta-analysis. Int J Cancer 71(5):800–809
Colditz GA, Willett WC, Hunter DJ, Stampfer MJ, Manson JE, Hennekens CH, Rosner BA (1993) Family history, age, and risk of breast cancer. Prospective data from the Nurses’ Health Study. JAMA 270(3):338–343
Houlston RS, McCarter E, Parbhoo S, Scurr JH, Slack J (1992) Family history and risk of breast cancer. J Med Genet 29(3):154–157
Weinberg CR, Shore DL, Umbach DM, Sandler DP (2007) Using risk-based sampling to enrich cohorts for endpoints, genes, and exposures. Am J Epidemiol 166(4):447–455. doi:10.1093/aje/kwm097
Spector D, Deroo LA, Sandler DP (2011) Lifestyle behaviors in black and white women with a family history of breast cancer. Prev Med 52(5):394–397. doi:10.1016/j.ypmed.2011.03.001
John EM, Hopper JL, Beck JC, Knight JA, Neuhausen SL, Senie RT, Ziogas A, Andrulis IL, Anton-Culver H, Boyd N, Buys SS, Daly MB, O’Malley FP, Santella RM, Southey MC, Venne VL, Venter DJ, West DW, Whittemore AS, Seminara D (2004) The Breast Cancer Family Registry: an infrastructure for cooperative multinational, interdisciplinary and translational studies of the genetic epidemiology of breast cancer. Breast cancer Res 6(4):R375–R389. doi:10.1186/bcr801
Delgado-Cruzata L, Wu HC, Liao Y, Santella RM, Terry MB (2014) Differences in DNA methylation by extent of breast cancer family history in unaffected women. Epigenetics 9(2):243–248. doi:10.4161/epi.26880
Delgado-Cruzata L, Wu HC, Perrin M, Liao Y, Kappil MA, Ferris JS, Flom JD, Yazici H, Santella RM, Terry MB (2012) Global DNA methylation levels in white blood cell DNA from sisters discordant for breast cancer from the New York site of the Breast Cancer Family Registry. Epigenetics 7(8):868–874. doi:10.4161/epi.20830
Zipprich J, Terry MB, Brandt-Rauf P, Freyer GA, Liao Y, Agrawal M, Gurvich I, Senie R, Santella RM (2010) XRCC1 polymorphisms and breast cancer risk from the New York Site of the Breast Cancer Family Registry: a family-based case-control study. J Carcinog 9:4. doi:10.4103/1477-3163.62535
Bilgrad R (2009) National Death Index User’s Guide. Centers for Disease Control and Prevention, National Center for Health Statistics, Department of Health and Human Services, Atlanta
Neuhausen SL, Ozcelik H, Southey MC, John EM, Godwin AK, Chung W, Iriondo-Perez J, Miron A, Santella RM, Whittemore A, Andrulis IL, Buys SS, Daly MB, Hopper JL, Seminara D, Senie RT, Terry MB (2009) BRCA1 and BRCA2 mutation carriers in the Breast Cancer Family Registry: an open resource for collaborative research. Breast Cancer Res Treat 116(2):379–386. doi:10.1007/s10549-008-0153-8
Anglian Breast Cancer Study Group (2000) Prevalence and penetrance of BRCA1 and BRCA2 mutations in a population-based series of breast cancer cases. Br J cancer 83(10):1301–1308. doi:10.1054/bjoc.2000.1407
Malone KE, Daling JR, Doody DR, Hsu L, Bernstein L, Coates RJ, Marchbanks PA, Simon MS, McDonald JA, Norman SA, Strom BL, Burkman RT, Ursin G, Deapen D, Weiss LK, Folger S, Madeoy JJ, Friedrichsen DM, Suter NM, Humphrey MC, Spirtas R, Ostrander EA (2006) Prevalence and predictors of BRCA1 and BRCA2 mutations in a population-based study of breast cancer in white and black American women ages 35 to 64 years. Cancer Res 66(16):8297–8308. doi:10.1158/0008-5472.CAN-06-0503
Loman N, Johannsson O, Kristoffersson U, Olsson H, Borg A (2001) Family history of breast and ovarian cancers and BRCA1 and BRCA2 mutations in a population-based series of early-onset breast cancer. J Natl Cancer Inst 93(16):1215–1223
Quante AS, Whittemore AS, Shriver T, Strauch K, Terry MB (2012) Breast cancer risk assessment across the risk continuum: genetic and nongenetic risk factors contributing to differential model performance. Breast Cancer Res 14(6):R144. doi:10.1186/bcr3352
Biffl WL, Myers A, Franciose RJ, Gonzalez RJ, Darnell D (2001) Is breast cancer in young Latinas a different disease? Am J Surg 182(6):596–600
Smigal C, Jemal A, Ward E, Cokkinides V, Smith R, Howe HL, Thun M (2006) Trends in breast cancer by race and ethnicity: update 2006. Cancer J Clin 56(3):168–183
Ooi SL, Martinez ME, Li CI (2011) Disparities in breast cancer characteristics and outcomes by race/ethnicity. Breast Cancer Res Treat 127(3):729–738. doi:10.1007/s10549-010-1191-6
Howe HL, Wu X, Ries LA, Cokkinides V, Ahmed F, Jemal A, Miller B, Williams M, Ward E, Wingo PA, Ramirez A, Edwards BK (2006) Annual report to the nation on the status of cancer, 1975-2003, featuring cancer among U.S. Hispanic/Latino populations. Cancer 107(8):1711–1742. doi:10.1002/cncr.22193
Johnson RH, Chien FL, Bleyer A (2013) Incidence of breast cancer with distant involvement among women in the United States, 1976 to 2009. JAMA 309(8):800–805. doi:10.1001/jama.2013.776
Tehranifar P, Akinyemiju TF, Terry MB (2013) Incidence rate of breast cancer in young women. JAMA 309(23):2433–2434. doi:10.1001/jama.2013.6461
Banegas MP, Li CI (2012) Breast cancer characteristics and outcomes among Hispanic Black and Hispanic White women. Breast Cancer Res Treat 134(3):1297–1304. doi:10.1007/s10549-012-2142-1
Chaudhry ZW, Brown RV, Fawole OA, Wilson R, Gudzune KA, Maruthur NM, Segal J, Hutfless SM (2013) Comparative effectiveness of strategies to prevent weight gain among women with and at risk for breast cancer: a systematic review. SpringerPlus 2(1):277. doi:10.1186/2193-1801-2-277
Spark LC, Reeves MM, Fjeldsoe BS, Eakin EG (2013) Physical activity and/or dietary interventions in breast cancer survivors: a systematic review of the maintenance of outcomes. J cancer surviv 7(1):74–82. doi:10.1007/s11764-012-0246-6
Winzer BM, Whiteman DC, Reeves MM, Paratz JD (2011) Physical activity and cancer prevention: a systematic review of clinical trials. Cancer Causes Control 22(6):811–826. doi:10.1007/s10552-011-9761-4
Toriola AT, Colditz GA (2013) Trends in breast cancer incidence and mortality in the United States: implications for prevention. Breast Cancer Res Treat 138(3):665–673. doi:10.1007/s10549-013-2500-7
Colditz GA, Bohlke K, Berkey CS (2014) Breast cancer risk accumulation starts early: prevention must also. Breast Cancer Res Treat 145(3):567–579. doi:10.1007/s10549-014-2993-8
Pace LE, Keating NL (2014) A systematic assessment of benefits and risks to guide breast cancer screening decisions. JAMA 311(13):1327–1335. doi:10.1001/jama.2014.1398
Independent UK Panel on Breast Cancer Screening (2012) The benefits and harms of breast cancer screening: an independent review. Lancet 380(9855):1778–1786. doi:10.1016/S0140-6736(12)61611-0
Davies C, Godwin J, Gray R, Clarke M, Cutter D, Darby S, McGale P, Pan HC, Taylor C, Wang YC, Dowsett M, Ingle J, Peto R (2011) Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet 378(9793):771–784. doi:10.1016/S0140-6736(11)60993-8
Price MA, Butow PN, Charles M, Bullen T, Meiser B, McKinley JM, McLachlan SA, Phillips KA (2010) Predictors of breast cancer screening behavior in women with a strong family history of the disease. Breast Cancer Res Treat 124(2):509–519. doi:10.1007/s10549-010-0868-1
Campitelli MA, Chiarelli AM, Mirea L, Stewart L, Glendon G, Ritvo P, Andrulis IL, Knight JA (2011) Adherence to breast and ovarian cancer screening recommendations for female relatives from the Ontario site of the Breast Cancer Family Registry. Eur J Cancer Prev 20(6):492–500. doi:10.1097/CEJ.0b013e3283476217
Madlensky L, Vierkant RA, Vachon CM, Pankratz VS, Cerhan JR, Vadaparampil ST, Sellers TA (2005) Preventive health behaviors and familial breast cancer. Cancer Epidemiol Biomark Prev 14(10):2340–2345. doi:10.1158/1055-9965.EPI-05-0254
Isaacs C, Peshkin BN, Schwartz M, Demarco TA, Main D, Lerman C (2002) Breast and ovarian cancer screening practices in healthy women with a strong family history of breast or ovarian cancer. Breast Cancer Res Treat 71(2):103–112
Murphy CC, Bartholomew LK, Carpentier MY, Bluethmann SM, Vernon SW (2012) Adherence to adjuvant hormonal therapy among breast cancer survivors in clinical practice: a systematic review. Breast Cancer Res Treat 134(2):459–478. doi:10.1007/s10549-012-2114-5
Acknowledgments
This work was supported by the Grant UM1 CA164920 from the USA National Cancer Institute. The content of this manuscript does not necessarily reflect the views or policies of the National Cancer Institute or any of the collaborating centers in the Breast Cancer Family Registry (BCFR), nor does mention of trade names, commercial products, or organizations imply endorsement by the USA Government or the BCFR.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Cloud, A.J., Thai, A., Liao, Y. et al. The impact of cancer prevention guideline adherence on overall mortality in a high-risk cohort of women from the New York site of the Breast Cancer Family Registry. Breast Cancer Res Treat 149, 537–546 (2015). https://doi.org/10.1007/s10549-014-3234-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-014-3234-x