Abstract
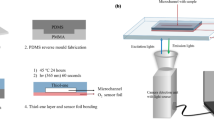
Polydimethylsiloxane (PDMS) is a commonly used polymer in the fabrication of microfluidic devices due to such features as transparency, gas permeability, and ease of patterning with soft lithography. The surface characteristics of PDMS can also be easily changed with oxygen or low pressure air plasma converting it from a hydrophobic to a hydrophilic state. As part of such a transformation, surface methyl groups are removed and replaced with hydroxyl groups making the exposed surface to resemble silica, a gas impermeable substance. We have utilized Platinum(II)-tetrakis(pentaflourophenyl)porphyrin immobilized within a thin (~1.5 um thick) polystyrene matrix as an oxygen sensor, Stern-Volmer relationship, and Fick’s Law of simple diffusion to measure the effects of PDMS composition, treatment, and storage on oxygen diffusion through PDMS. Results indicate that freshly oxidized PDMS showed a significantly smaller diffusion coefficient, indicating that the SiO2 layer formed on the PDMS surface created an impeding barrier. This barrier disappeared after a 3-day storage in air, but remained significant for up to 3 weeks if PDMS was maintained in contact with water. Additionally, higher density PDMS formulation (5:1 ratio) showed similar diffusion characteristics as normal (10:1 ratio) formulation, but showed 60 % smaller diffusion coefficient after plasma treatment that never recovered to pre-treatment levels even after a 3-week storage in air. Understanding how plasma surface treatments contribute to oxygen diffusion will be useful in exploiting the gas permeability of PDMS to establish defined normoxic and hypoxic oxygen conditions within microfluidic bioreactor systems.




Similar content being viewed by others
Abbreviations
- EOF:
-
Electroosmotic flow
- PDMS:
-
Polydimethylsiloxane
- Pt-TPFP:
-
Platinum(II)-tetrakis(pentaflourophenyl)porphyrin
- TTB:
-
Thick tissue bioreactor
References
M. Adler, M. Polinkovsky, E. Gutierrez, and A. Groisman, Lab Chip 10, 388 (2010)
Y. Amao, I. Okura, Tanpakushitsu Kakusan Koso 43, 1492 (1998)
Y. Amao, T. Miyashita, I. Okura, J. Fluor. Chem. 107, 101 (2001)
R. Cairns, I. Papandreou, N. Denko, Mol. Cancer Res. 4, 61 (2006)
E.R. Carraway, J.N. Demas, B.A. Degraff, Anal. Chem. 63, 332 (1991a)
E.R. Carraway, J.N. Demas, B.A. Degraff, J.R. Bacon, Anal. Chem. 63, 337 (1991b)
D.C. Duffy, J.C. McDonald, O.J. Schueller, G.M. Whitesides, Anal. Chem. 70, 4974 (1998)
C.S. Effenhauser, G.J. Bruin, A. Paulus, M. Ehrat, Anal. Chem. 69, 3451 (1997)
K. Efimenko, W.E. Wallace, J. Genzer, J. Colloid Interface Sci. 254, 306 (2002)
D. Erickson, D. Sinton, D. Li, Lab Chip 3, 141 (2003)
J.L. Fritz, M.J. Owen, J. Adhes. 54, 33 (1995)
Y. Gao, D. Majumdar, B. Jovanovic, C. Shaifer, P.C. Lin, A. Zijlstra, D.J. Webb, D. Li, Biomed. Microdevices 13, 539 (2011)
R.A. Gatenby, R.J. Gillies, Nat. Rev. Cancer 8, 56 (2008)
R.A. Gatenby, K. Smallbone, P.K. Maini, F. Rose, J. Averill, R.B. Nagle, L. Worrall, R.J. Gillies, Br.J. Cancer 97, 646 (2007)
H. Hillborg, J.F. Ankner, U.W. Gedde, G.D. Smith, H.K. Yasuda, K. Wikstrom, Polymer 41, 6851 (2000)
B.H. Jo, L.M. Van Lerberghe, K.M. Motsegood, D.J. Beebe, J. Microelectromech. Syst. 9, 76 (2000)
G.E. Khalil, A. Chang, M. Gouterman, J.B. Callis, L.R. Dalton, N.J. Turro, and S. Jockusch, Rev. Sci. Instrum. 76, 054101 (2005)
M.S. Kim, J.H. Yeon, J.K. Park, Biomed. Microdevices 9, 25 (2007)
E. Leclerc, Y. Sakai, T. Fujii, Biotechnol. Prog. 20, 750 (2004)
G.B. Lee, C.H. Lin, K.H. Lee, Y.F. Lin, Electrophoresis 26, 4616 (2005)
L. Liu, K. Loutherback, D. Liao, D. Yeater, G. Lambert, A. Estevez-Torres, J.C. Sturm, R.H. Getzenberg, R.H. Austin, Lab Chip 10, 1807 (2010)
K. Lundgren, C. Holm, G. Landberg, Cell Mol. Life Sci. 64, 3233 (2007)
D.A. Markov, J.Q. Lu, P.C. Samson, J.P. Wikswo, L.J. McCawley, Lab Chip 12, 4560 (2012)
J.C. McDonald, G.M. Whitesides, Acc. Chem. Res. 35, 491 (2002)
J.C. McDonald, D.C. Duffy, J.R. Anderson, D.T. Chiu, H. Wu, O.J. Schueller, G.M. Whitesides, Electrophoresis 21, 27 (2000)
M.J. Owen, P.J. Smith, J. Adhes. Sci. Technol. 8, 1063 (1994)
M. Polinkovsky, E. Gutierrez, A. Levchenko, and A. Groisman, Lab Chip 9, 1073 (2009)
A. Prokop, Z. Prokop, D. Schaffer, E. Kozlov, J. Wikswo, D. Cliffel, F. Baudenbacher, Biomed. Microdevices 6, 325 (2004)
X. Ren, M. Bachman, C. Sims, G.P. Li, N. Allbritton, J. Chromatogr. B: Biomed. Sci. Appl. 762, 117 (2001)
Y. Rharbi, A. Yekta, M.A. Winnik, Anal. Chem. 71, 5045 (1999)
T. Saito, C.C. Wu, H. Shiku, T. Yasukawa, M. Yokoo, T. Ito-Sasaki, H. Abe, H. Hoshi, T. Matsue, Analyst 131, 1006 (2006)
P.C. Thomas, M. Halter, A. Tona, S.R. Raghavan, A.L. Plant, S.P. Forry, Anal. Chem. 81, 9239 (2009)
P.C. Thomas, S.R. Raghavan, S.P. Forry, Anal. Chem. 83, 8821 (2011)
O. Tredan, C.M. Galmarini, K. Patel, I.F. Tannock, J. Natl. Cancer Inst. 99, 1441 (2007)
J.A. Vickers, M.M. Caulum, C.S. Henry, Anal. Chem. 78, 7446 (2006)
X. Wang, C. Cheng, S. Wang, S. Liu, Microfluid. Nanofluidics. 6, 145 (2009)
Welty, J.R., Wicks, C.E., Wilson R.E., Rorrer, G, “Fundamentals of Momentum, Heat, and Mass Transfer” 4th ed. Wiley, 2001
Y. Zhang, M. Li, Q. Yao, C. Chen, Med. Sci. Monit. 13, RA175 (2007)
Acknowledgments
This research was supported by the Department of Defense Breast Cancer Research Program (DOD BCRP) grants W81XWH-09-1-0444 and W81XWH-10-1-0157 and NIH/NCI R21 CA126728-01 to L.J.M., by 1UH2TR000491-01, by the Vanderbilt Institute for Integrative Biosystems Research and Education (VIIBRE) and through the Searle Systems Biology and Bioengineering Undergraduate Research Experience (Searle SyBBURE) to E.M.L. We would like to thank VINSE for use of Fluorolog-3 FL3-111 Spectrophotofluorometer, L. Smith for help with establishing conditions for Figure S5, and J.P. Wikswo and P.C. Samson for useful discussions.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
Relationship between spin speed and the resulting thickness of PDMS membrane after curing at 65 °C for 4 h. Spinner: Laurell WS-400-6NPP; conditions: 2 s spread cycle at 600 rpm, 30 s at designated speed. Note: These parameters are given as a reference. The end-user would have to adjust them to accommodate their actual setup and equipment. Time course of fluorescence quenching for thin membranes is shown in Figure S4. (PDF 52.4 kb)
Figure S2
Derivations of the formulas used for non-linear fit to determine D. (PDF 38 kb)
Figure S3
Oxygen sensing film calibration. The Ksv coefficient from the Stern-Volmer relationship is found as a slope of the calibration curve. (PDF 15 kb)
Figure S4
Fluorescence quenching as a function of oxygen diffusing through the PDMS membranes of various thicknesses. (PDF 135 kb)
Figure S5
Changes in emission spectra for Pt-TFPP compound immobilized within polystyrene matrix in the presence of atmospheric oxygen (Ex = 540 +/−2.5 nm) visualized with Fluorolog-3 FL3-111 Spectrophotofluorometer. (PDF 15 kb)
Rights and permissions
About this article
Cite this article
Markov, D.A., Lillie, E.M., Garbett, S.P. et al. Variation in diffusion of gases through PDMS due to plasma surface treatment and storage conditions. Biomed Microdevices 16, 91–96 (2014). https://doi.org/10.1007/s10544-013-9808-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-013-9808-2