Abstract
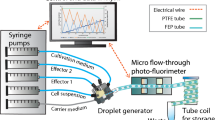
This paper discusses the vortical flow, mixing and cell culture of Pichia pastoris using a centrifugal microfluidic (CM) chamber. The resultant “spiral toroidal vortex” in the chamber is made up of a primary vortex induced from inertial acceleration/deceleration of the chamber superposed by a secondary toroidal vortex due to Coriolis acceleration acting on the primary vortex. A validated numerical fluid-flow model with minimized numerical diffusion effect has been developed to investigate the flow and consequently mixing of two-color liquids through cyclic constant acceleration-and-deceleration in the same rotation direction until homogeneous mixing of the two liquids in the CM chamber has been established. The specific mixing time is found to improve with increase in acceleration/deceleration and angular span of the chamber. An experimental CM platform with three cell-culture chambers of different angular spans has been built and Pichia pastoris cell culture has been successfully demonstrated. Cell growth can be monitored over time on the extracted samples by measuring the optical density at 600-nm wave-length. Comparing with conventional cell culture, Pichia pastoris cultured on CM platform exhibits a very short lag (cell preparation/budding) phase prior to the log phase (cell growth). While it takes 8 to 12 h for the conventional shake flask in the lag phase, it takes only 2 h for the CM platform irrespective of initial cell concentration (8.1 × 104 to 8.1 × 105/ml), acceleration/deceleration (10 to 32/s2) and angular span of the culture chamber (π/12 to π/4), representing significant time reduction. This is largely attributed to better growth conditions due to enhanced mixing and appropriate shear-stress stimulation from the efficient spiral toroidal vortex.












Similar content being viewed by others
Abbreviations
- C :
-
Concentration (g/m3)
- D :
-
Diffusion coefficient (m2/s)
- H :
-
Height of chamber (m)
- MT :
-
Mixing time when the mixing quality α reaches 90 % (s)
- P :
-
Pressure (Pa)
- q :
-
Velocity vector (m/s)
- r :
-
Radial coordinate (m)
- SMT :
-
Specific mixing time (s/L)
- t :
-
Time variable (s)
- V :
-
Chamber volume (m3)
- z :
-
Vertical coordinate (m)
- α :
-
Mixing quality (−)
- θ :
-
Angular span of chamber (rad)
- ν:
-
Kinematic viscosity (m2/s)
- ρ :
-
Fluid density (kg/m3)
- σ:
-
Standard deviation (−)
- ϕ :
-
Azimuthal coordinate (rad)
- Ω :
-
Rotation speed (rad/s)
- i :
-
Grid location, also inner
- M :
-
Maximum value
- o :
-
Outer
References
A. Bhagat, H. Bow, H. Hou, S. Tan, J. Han, C. Lim, Microfluidics for cell separation. Med. Biol. Eng. Comput. 48(10), 999–1014 (2010)
M.R. Bringer, C.J. Gerdts, H. Song, J.D. Tice, R.F. Ismagilov, Microfluidic systems for chemical kinetics that rely on chaotic mixing in droplets. Philos. Trans. R. Soc. London, Ser. A 362, 1087–1104 (2004)
D. Chakraborty, M. Madou, S. Chakraborty, Anomalous mixing behavior in rotationally actuated microfluidic devices. Lab Chip 11, 2823–2826 (2011)
W.J. Chang, D. Akin, M. Sedlak, M.R. Ladisch, R. Bashir, Poly(dimethylsiloxane) (PDMS) and silicon hybrid biochip for bacterial culture. Biomed. Microdev. 5, 281–290 (2003)
J.M. Cregg, I. Tolstorukov, A. Kusari, J. Sunga, K. Madden, T. Chappell, Expression in the yeast Pichia pastoris. Meth. Enzymol. 463, 169–189 (2009)
J. Ducrée, S. Haeberle, S. Lutz, S. Pausch, F. Stetten, R. Zengerle, The centrifugal microfluidic bio-disk platform. J. Micromech. Microeng. 17, 103–115 (2007)
K.N. Faber, W. Harder, M. Veenhuis, Review: methylotropic yeasts as factories for the production of foreign proteins. Yeast 11, 1331–1344 (1995)
D. Gao, H. Liu, Y. Jiang, J. Lin, Recent developments in microfluidic devices for in vitro cell culture for cell-biology research. Trac- Trend. Anal. Chem. 35, 150–164 (2012)
R. Gómez, R. Bashir, A.K. Bhunia, Microscale electronic detection of bacterial metabolism. Sensors Actuators B 86, 198–208 (2002)
R. Gorkin, J. Park, J. Siegrist, M. Amasia, B.S. Lee, J.M. Park, J. Kim, H. Kim, M. Madou, Y.K. Cho, Centrifugal microfluidics for biomedical applications. Lab Chip 10(14), 1758–1773 (2010)
M. Grumann, A. Geipel, L. Riegger, R. Zengerle, J. Ducrée, Batch-mode mixing on centrifugal microfluidic platforms. Lab Chip 5, 560–565 (2005)
C.W. Huang, G.B. Lee, A microfluidic system for automatic cell culture. J. Micromech. Microeng. 17, 1266–1274 (2007)
M. Huang, S. Fan, W. Xing, C. Liu, Microfluidic cell culture system studies and computational fluid dynamics. Math. Comput. Model. 52(11–12), 2036–2042 (2010)
P.J. Hung, P.J. Lee, P. Sabounchi, R. Lin, L.P. Lee, Continuous perfusion microfluidic cell culture array for high-throughput cell-based assays. Biotechnol. Bioeng. 89(1), 1–8 (2005)
N.K. Inamdar, J.T. Borenstein, Microfluidic cell culture models for tissue engineering. Curr. Opin. Biotechnol. 22(5), 681–689 (2011)
N. Kim, C.M. Dempsey, J.V. Zoval, J. Sze, M.J. Madou, Automated microfluidic compact disc (CD) cultivation system of Caenorhabditis elegans. Sensors Actuators B 122, 511–518 (2007)
T.J. Kim, S.J. Kim, H.I. Jung, Physical stimulation of mammalian cells using micro-bead impact within a microfluidic environment to enhance growth rate. Microfluid Nanofluid 6, 131–138 (2009)
K. Kojima, H. Moriguchi, A. Hattori, T. Kaneko, K. Yasuda, Two-dimensional network formation of cardiac myocytes in agar microculture chip with 1480 nm infrared laser photo-thermal etching. Lab Chip 3, 292–296 (2003)
C.Y. Lee, G.B. Lee, J.L. Lin, F.C. Huang, L.C. Sheng, Integrated microfluidic systems for cell lysis, mixing/pumping and DNA amplification. J. Micromech. Microeng. 15(6), 1215 (2005)
K.F. Lei, M.H. Wu, P.Y. Liao, Y.M. Chen, T.M. Pan, Development of a micro-scale perfusion 3-D cell culture biochip with an incorporated electrical impedance measurement scheme for the quantification of cell number in a 3-D cell culture construct. Microfluid Nanofluid 12, 117–125 (2012)
M. Madou, J. Zoval, G. Jia, H. Kido, J. Kim, N. Kim, Lab on a CD. Annu. Rev. Biomed. Eng. 8(1), 601–628 (2006)
D. Mark, S. Haeberle, G. Roth, F. von Stetten, R. Zengerle, Microfluidic lab-on-a-chip platforms: requirements, characteristics and applications. Chem. Soc. Rev. 39(3), 1153 (2010)
N.T. Nguyen, S.T. Wereley, Fundamentals and applications of microfluidics, 2nd edn. (Artech House, Boston, 2006)
J. Nielsen, J. Villadsen, Bioreaction engineering principles (Plenum Press, New York, 1994)
M.J. Powers, K. Domansky, M.R. Kaazempur-Mofrad, A. Kalezi, A. Capitano, A. Upadhyaya, P. Kurzawski, K.E. Wack, D.B. Stolz, R. Kamm, L.G. Griffith, A microfabricated array bioreactor for perfused 3-D liver culture. Biotechnol. Bioeng. 78, 257–269 (2002)
Y. Ren, W.W.F. Leung, Numerical and experimental investigation on flow and mixing in batch-mode centrifugal microfluidics. Int. J. Heat Mass Transfer (in press) (2012)
G. Roman, Y. Chen, P. Viberg, A. Culbertson, C. Culbertson, Single-cell manipulation and analysis using microfluidic devices. Anal. Bioanal. Chem. 387(1), 9–12 (2007)
A. Sin, K.C. Chin, M.F. Jamil, Y. Kostov, G. Rao, M.L. Shuler, The design and fabrication of three-chamber microscale cell culture analog devices with integrated dissolved oxygen sensors. Biotechnol. 20, 338–345 (2004)
M. Stangegaard, S. Petronis, A.M. Jørgensen, C.B.V. Christensenc, M. Dufva, A biocompatible micro cell culture chamber (μCCC) for the culturing and on-line monitoring of eukaryote cells. Lab Chip 6, 1045–1051 (2006)
A. Takano, M. Tanaka, N. Futai, On-chip CO2 incubation for pocket-sized microfluidic cell culture. Microfluid Nanofluid 12, 907–915 (2012)
T. Thorsen, S. Maerkl, S. Quake, Microfluidic large scale integration. Science 298, 580–584 (2002)
Y.C. Toh, J. Voldman, Fluid shear stress primes mouse embryonic stem cells for differentiation in a self-renewing environment via heparan sulfate proteoglycans transduction. FASEB J. 25(4), 1208–1217 (2011)
E. Verpoorte, N.F. De Rooij, Microfluidics meets MEMS. Proc. IEEE 91(6), 930–953 (2003)
H.Y. Wang, N. Bao, C. Lu, A microfluidic cell array with individually addressable culture chambers. Biosens. Bioelectron. 24, 613–617 (2008)
D.B. Weibel, P.G. Garstecki, G.M. Whitesides, Combining microscience and neurobiology. Curr. Opin. Neurobiol. 15, 560–567 (2005)
G.M. Whitesides, The origins and the future of microfluidics. Nature 442(7101), 368–373 (2006)
M.H. Wu, J.L. Lin, J. Wang, Z. Cui, Z. Cui, Development of high throughput optical sensor array for on-line pH monitoring in micro-scale cell culture environment. Biomed. Microdevices 11, 265–273 (2009)
M.H. Wu, S.B. Huang, G.B. Lee, Microfluidic cell culture systems for drug research. Lab Chip 10, 939–956 (2010)
C.H. Yeh, P.W. Lin, Y.C. Lin, Chitosan microfiber fabrication using a microfluidic chip and its application to cell cultures. Microfluid Nanofluid 8, 115–121 (2010)
J.H. Yeon, J.K. Park, Microfluidic cell culture systems for cellular analysis. Biochip J. 1(1), 17–27 (2007)
C. Yi, C.W. Li, S. Ji, M. Yang, Microfluidics technology for manipulation and analysis of biological cells. Anal. Chim. Acta 560, 1–23 (2006)
S.K. Yoo, J.H. Lee, S.S. Yun, M.B. Gu, J.H. Lee, Fabrication of a bio-MEMS based cell-chip for toxicity monitoring. Biosens. Bioelectron. 22, 1586–1592 (2007)
Acknowledgements
The authors acknowledge the funding support from the Hong Kong Grant Research Council under project no GRF/B-Q15S. Yong Ren also acknowledges the help from Dr Tabris Chung and Ms Kit Ying Choy for demonstration of cell counting and conventional cell culture technology.
Author information
Authors and Affiliations
Corresponding author
Appendix-Sensitivity study of grid size, time step and numerical diffusion
Appendix-Sensitivity study of grid size, time step and numerical diffusion
To validate our numerical model (i.e., chamber with r i = 35 mm, r o = 53 mm, θ = 15° and H = 0.5 mm), the grid size and time step sensitivity studies were conducted by checking the velocity profiles at t = 3 s when Δt = 5 s and dΩ/dt = ±20.9 rad/s2, as demonstrated in Fig. 13. It shows that the solution can reach convergence when Δt = 0.05 s and Δx = 0.1 mm. The dependence of numerical diffusion on the order of computational discretization scheme (i.e., backward Euler scheme) and grid size at t = 1 and 2 s respectively when Δt = 5 s and dΩ/dt = ±20.9 rad/s2 was investigated in Fig. 14, which demonstrates qualitatively that the numerical diffusion can be minimized using the 2nd order scheme and grid size of 0.1 mm. The same conclusion can be drawn from the quantitative results in Fig. 15, wherein the species concentration distribution profile versus radial position r (z = 0.25 mm, ϕ = 3.75°, see the white dash curve in Fig. 14,) is demonstrated and the curve with the steepest slope represents the condition with minimized numerical diffusion. In the meanwhile, the results are compared with the benchmarking condition when the physical diffusion is disabled (Diffusion coefficient, D = 0 mm2/s). It shows some variance (i.e., reduced diffusion layer thickness) from the optimal case when the 2nd order scheme, grid size of 0.1 mm and D = 0.015 mm2/s were used, indicating the mixing effect in this scenario is completely attributed to the numerical diffusion.
Order of computational discretization scheme and grid size effects to concentration distribution profile over radial position r (z = 0.25 mm, ϕ = 3.75°, see the white dash curve in Fig. A.2) at t = 1 and 2 s respectively when Δt = 5 s and dΩ/dt = ±20.9 rad/s2
Rights and permissions
About this article
Cite this article
Ren, Y., Chow, L.MC. & Leung, W.WF. Cell culture using centrifugal microfluidic platform with demonstration on Pichia pastoris . Biomed Microdevices 15, 321–337 (2013). https://doi.org/10.1007/s10544-012-9735-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-012-9735-7