Abstract
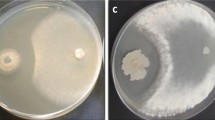
Black rot, caused by Xanthomonas campestris pv. campestris (Xcc) (Xanthomonadales: Xanthomonadaceae), is the most important and potentially destructive disease of cabbage. Twenty-three plant growth-promoting rhizobacteria (PGPR) strains were tested for antibiosis against Xcc in vitro. Seven strains with antibiosis activity significantly reduced disease in the greenhouse. Two mixtures of PGPR strains and four individual strains were then tested three times in the greenhouse and one time in the field. In the greenhouse test, all treatments resulted in significant disease suppression. Mixture-2 and strain AP218 caused the highest and most consistent disease reduction in two of the three trials. In the field test, both mixtures and two individual strains significantly reduced disease incidence and disease severity. In conclusion, mixture-2 exhibited consistent biocontrol of black rot of cabbage.
Similar content being viewed by others
References
Akhtar M (1989) Xanthomonas campestris pv. campestris causing black rot in cabbage. Pak J Agric Res 10:311–313
Alvarez AM (2000) Black rot of crucifers. In: Slusarenko AJ, Fraser RSS, Loon LCV (eds) Mechanisms of resistance to plant diseases. Kluwer, Dordrecht, pp 21–52
Brown JE, Lu TY, Stevens C, Khan VA, Lu JY, Wilson CL, Collins DJ, Wilson MA, Igwegbe ECK, Chalutz E, Droby S (2001) The effect of low dose ultraviolet light-C seed treatment on induced resistance in cabbage to black rot (Xanthomonas campestris pv. campestris). Crop Protect 20:873–883
Domenech J, Reddy M, Kloepper J, Ramos B, Gutierrez-Manero J (2006) Combined application of the biological product LS213 with Bacillus, Pseudomonas or Chryseobacterium for growth promotion and biological control of soil-borne diseases in pepper and tomato. BioControl 51:245–258
Hugouvieux V, Barber CE, Daniels MJ (1998) Entry of Xanthomonas campestris pv. campestris into hydathodes of Arabidopsis thaliana leaves: a system for studying early infection events in bacterial pathogenesis. Mol Plant-Microbe Interact 11:537–543
Jetiyanon K, Kloepper JW (2002) Mixtures of plant growth-promoting rhizobacteria for induction of systemic resistance against multiple plant diseases. Biol Control 24:285–291
Jetiyanon K, Fowler WD, Kloepper JW (2003) Broad-spectrum protection against several pathogens by PGPR mixtures under field conditions in Thailand. Plant Dis 87:1390–1394
Krauthausen HJ, Laun N, Wohanka W (2011) Methods to reduce the spread of the black rot pathogen, Xanthomonas campestris pv. campestris, in brassica transplants. J Plant Dis Protect 118:7–16
Mew T, Natural M (1993) Management of Xanthomonas diseases. In: Swings JG, Civerolo EL (eds) Xanthomonas. Chapman & Hall, London, pp 341–362
Mishra S, Arora NK (2012a) Evaluation of rhizospheric Pseudomonas and Bacillus as biocontrol tool for Xanthomonas campestris pv campestris. World J Microbiol Biotechnol 28:693–702
Mishra S, Arora NK (2012b) Management of black rot in cabbage by rhizospheric Pseudomonas species and analysis of 2,4-diacetylphloroglucinol by qRT-PCR. Biol Control 61:32–39
Murphey Coy R, Held DW, Kloepper JW (2014) Rhizobacterial inoculants increase root and shoot growth in Tifway hybrid bermudagrass. J Environ Hort 32:149–154
Nega E, Ulrich R, Werner S, Jahn M (2003) Hot water treatment of vegetable seed–an alternative seed treatment method to control seed borne pathogens in organic farming. J Plant Dis Protect 110:220–234
Ran C, Carrias A, Williams MA, Capps N, Dan BCT, Newton JC, Kloepper JW, Ooi EL, Browdy CL, Terhune JS, Liles MR (2012) Identification of Bacillus strains for biological control of catfish pathogens. PLoS ONE 7(9):e45793
Raupach GS, Kloepper JW (1998) Mixtures of plant growth-promoting rhizobacteria enhance biological control of multiple cucumber pathogens. Phytopathology 88:1158–1164
Saharan B, Nehra V (2011) Plant growth promoting rhizobacteria: a critical review. Life Sci Med Res 21:1–30
Schaad N, Alvarez A (1993) Xanthomonas campestris pv. campestris: cause of black rot of crucifers. In: Swings JG, Civerolo EL (eds) Xanthomonas. Chapman & Hall, London, pp 51–55
United States Department of Agriculture-National Agricultural Statistics Service (2015) http://www.nass.usda.gov/Statistics_by_Subject/result.php?4AFF75C4-BC1E−3F52-858D-2C5DEDC5E034§or=CROPS&group=VEGETABLES&comm=CABBAGE
Vicente JG, Holub EB (2013) Xanthomonas campestris pv. campestris (cause of black rot of crucifers) in the genomic era is still a worldwide threat to brassica crops. Mol Plant Pathol 14:2–18
Williams P, Staub T, Sutton J (1972) Inheritance of resistance in cabbage to black rot. Phytopathology 62:247–252
Wulff EG, Mguni CM, Mortensen CN, Keswani CL, Hockenhull J (2002) Biological control of black rot (Xanthomonas campestris pv. campestris) of brassicas with an antagonistic strain of Bacillus subtilis in Zimbabwe. Eur J Plant Pathol 108:317–325
Zhang S, Reddy MS, Kloepper JW (2004) Tobacco growth enhancement and blue mold disease protection by rhizobacteria: relationship between plant growth promotion and systemic disease protection by PGPR strain 90-166. Plant Soil 262:277–288
Zhang S, White TL, Martinez MC, McInroy JA, Kloepper JW, Klassen W (2010) Evaluation of plant growth-promoting rhizobacteria for control of Phytophthora blight on squash under greenhouse conditions. Biol Control 53:129–135
Acknowledgments
We acknowledge and thank John McInroy and Dr. Chia-Hui Hu for their advice and technical help and John Olive for the source of Xanthomonas campestris pv. campestris. Funding was provided in part by the USDA National Institute of Food and Agriculture of Organic Agriculture Research and Extension Initiative (Grant No. 2011-51300-30634).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Jesus Mercado Blanco
Rights and permissions
About this article
Cite this article
Liu, K., Garrett, C., Fadamiro, H. et al. Antagonism of black rot in cabbage by mixtures of plant growth-promoting rhizobacteria (PGPR). BioControl 61, 605–613 (2016). https://doi.org/10.1007/s10526-016-9742-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-016-9742-3