Abstract
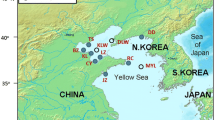
Genetic characteristics of four wild (Mie, Fukui, Shimane and Nagasaki) and five cultured populations (selectively bred for 12 years with an origin of Ehime population) of the Japanese pearl oyster, Pinctada fucata martensii, were evaluated by using AFLP markers. Six primer pairs generated 1019 loci in total, among which 45.2–55.1 % was polymorphic among populations. Although there was no significant difference in gene diversity between wild (0.170–0.174) and cultured (0.158–0.173) populations, genetic relatedness in cultured populations (0.316–0.450) was about three times higher than that in wild populations (0.110–0.165). In addition, genetic differentiation was about twenty times larger in cultured populations (Nei’s distance: 0.0111) than in wild populations (Nei’s distance: 0.0005). These results mean that selective breeding can cause marked inbreeding as well as large genetic differentiation among cultured populations in a short period. On the other hand, it was suggested that genetic homogenization in the wild, probably due to a large-scale transport of cultured oysters, had progressed in the sea around Japan. In conclusion, it is necessary to prevent inbreeding by the reconsideration of the style of selective breeding in cultured populations, while the escape of gametes or spats of cultured strains in the wild should be avoided for the preservation of genetic characteristics in native populations of P. f. martensii.



Similar content being viewed by others
References
Arnaud-Haond S, Vonau V, Bonhomme F, Boudry P, Blanc F, Prou J, Seaman T, Goyard E (2004) Spatio-temporal variation in the genetic composition of wild populations of pearl oyster (Pinctada margaritifera cumingii) in French Polynesia following 10 years of juvenile translocation. Mol Ecol 13:2001–2007
Atsumi T, Masuda T (2008) Relationships between filtration rate and temperature tolerance of Japanese, Chinese and hybrid pearl oysters, Pinctada fucata martensii. Fish Genet Breed Sci 37:43–49
Atsumi T, Komaru A, Okamoto C (2004) Genetic relationship among the Japanese pearl oysters Pinctada fucata martensii and other pearl oysters. Fish Genet Breed Sci 33:135–142
Beaumont AR, Khamdan SAA (1991) Electrophoretic and morphometric characters in population differentiation of the pearl oyster, Pinctada fucata (Leach), from around Bahrain. J Moll Stud 57:433–441
Bensch S, Åkesson M (2005) Ten years of AFLP in ecology and evolution: why so few animals? Mol Ecol 14:2899–2914
Colgan D, Ponder W (2002) Genetic discrimination of morphologically similar, sympatric species of pearl oysters (Mollusca: Bivalvia: Pinctada) in eastern Australia. Mar Freshw Res 53:697–709
Felsenstein J (2005) PHYLIP (Phylogeny Inference Package), Version 3.6. Distributed by the author. Department of Genome Sciences, University of Washington, Seattle [http://www.evolution.genetics.washington.edu/phylip.html]
Hayami I (2000) Subfamily Pteriacea. In: Okutani T (ed) Marine mollusks in Japan. Tokai University Press, Tokyo, pp 879–883
Huete-Pérez JA, Quezada F (2013) Genomic approaches in marine biodiversity and aquaculture. Biol Res 46:353–361
Ishikawa T, Okamoto C, Hayashi M, Aoki H, Isowa K, Komaru A (2009) The heritability of the shell-closing strength in the Japanese pearl oyster, Pinctada fucata martensii. Aquac Sci 57:77–82
Jameson HL (1921) The Japanese artificially induced pearl. Nature 107:396–398
Jiang W, Wei Y (1985) Observation on the viability of offspring from mating of close relatives. Hereditas (Beijing) 7:19–21
Krauss SL (2000) Accurate gene diversity estimates from amplified fragment length polymorphism (AFLP) markers. Mol Ecol 9:1241–1245
Kurita O, Hayashi M (2004) Study on character and discrimination of the breeding for Akoya oyster (Pinctada fucata). Rep Mie Pref Ind Res Inst 29:71–74
Kurokawa T, Suzuki T, Okauchi M, Miwa S, Nagai K, Nakamura K, Honjo T, Nakajima K, Ashida K, Funakoshi S (1999) Experimental infections of a disease causing mass mortalities of Japanese pearl oyster Pinctada fucata martensii by tissue transplantation and cohabitation. Nippon Suisan Gakkaishi 65:241–251
Lansman RA, Shade RO, Shapira JF, Avise JC (1981) The use of restriction endonucleases to measure mitochondrial DNA sequence relatedness in natural populations. III. Techniques and potential applications. J Mol Evol 17:214–226
Lemer S, Planes S (2012) Translocation of wild populations: conservation implications for the genetic diversity of the black-lipped pearl oyster Pinctada margaritifera. Mol Ecol 21:2949–2962
Lind CE, Evans BS, Knauer J, Taylor JJ, Jerry DR (2009) Decreased genetic diversity and a reduced effective population size in cultured silver-lipped pearl oysters (Pinctada maxima). Aquaculture 286:12–19
Liu Z, Cordes J (2004) DNA marker technologies and their applications in aquaculture genetics. Aquaculture 238:1–37
Lynch M, Milligan BG (1994) Analysis of population genetic structure with RAPD markers. Mol Ecol 3:91–99
Masaoka T, Kobayashi T (2005a) Natural hybridization between Pinctada fucata and Pinctada maculata inferred from internal transcribed spacer regions of nuclear ribosomal RNA genes. Fish Sci 71:829–836
Masaoka T, Kobayashi T (2005b) Species identification of Pinctada imbricata using intergenic spacer of nuclear ribosomal RNA genes and mitochondrial 16S ribosomal RNA gene regions. Fish Sci 71:837–846
McGinty EL, Evans BS, Taylor JU, Jerry DR (2010) Xenografts and pearl production in two pearl oyster species, P. maxima and P. margaritifera: effect on pearl quality and a key to understanding genetic contribution. Aquaculture 302:175–181
Minami H, Akera S, Kijima A (2000) Genetic variability and genetic differences in and among localities in the Japanese pearl oyster, Pinctada fucata marfensi. Fish Genet Breed Sci 29:103–111
Nei M (1972) Genetic distance between populations. Am Nat 106:283–292
Reitzel A, Herrera S, Layden M, Martindale M, Shank T (2013) Going where traditional markers have not gone before: utility of and promise for RAD sequencing in marine invertebrate phylogeography and population genomics. Mol Ecol 22:2953–2970
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225
Shimizu T, Takagi M (2011) Allozyme genetic variability of wild and cultured populations of Japanese pearl oyster Pinctada fucata martensi in the Uwa Sea, Ehime Prefecture, Japan. Aquacult Sci 59:411–418
Southgate PC (2008) Pearl oyster culture. In: Southgate PC, Lucas JS (eds) The pearl oyster. Elsevier, Oxford, pp 231–272
Vekemans X, Beauwens T, Lemaire M, Roldán-Ruiz I (2002) Data from amplified fragment length polymorphism (AFLP) markers show indication of size homoplasy and of a relationship between degree of homoplasy and fragment size. Mol Ecol 11:139–151
Vos P, Hogers R, Bleeker M, Reijans M, Van de Lee T, Hornes M, Friters A, Pot J, Paleman J, Kuiper M (1995) AFLP: a new technique for DNA fingerprinting. Nucl Acids Res 23:4407–4414
Wada KT (1984) Breeding study of the pearl oyster, Pinctada fucata. Bull Nat Res Inst Aquacult 6:79–157
Wada KT (1986) Genetic variability at four polymorphic loci in Japanese pearl oysters, Pinctada fucata martensii, selected for six generations. Aquaculture 59:139–146
Wada KT (2011) Production of pearls. Seizando Shoten, Tokyo
Wada KT, Komaru A (1994) Effect of selection for shell coloration on growth rate and mortality in the Japanese pearl oyster, Pinctada fucata martensii. Aquaculture 125:59–65
Wang N, Kinoshita S, Riho C, Maeyama K, Nagai K, Watabe S (2009) Quantitative expression analysis of nacreous shell matrix protein genes in the process of pearl biogenesis. Comp Biochem Physiol B-Biochem Mol Biol 154:346–350
Yamada A (2014) History of pearls in the world: wealth and ambition in the five milleniums. Chuoukouron-sha, Japan
Yano M, Nagai K, Morimoto K, Miyamoto H (2007) A novel nacre protein N19 in the pearl oyster Pinctada fucata. Biochem Biophys Res Commun 362:158–163
Yokogawa K (1999) Introduction of exotic fishes and shellfishes into Japan and their biological characteristics. Fish Genet Breed Sci 28:1–25
Yu DH, Chu KH (2006) Genetic variation in wild and cultured populations of the pearl oyster Pinctada fucata from southern China. Aquaculture 258:220–227
Zhivotovsky LA (1999) Estimating population structure in diploids with multilocus dominant DNA markers. Mol Ecol 8:907–913
Acknowledgments
We thank Ms Yamamoto for cooperating to collect samples and Dr. Hideo Aoki for helpful suggestions on the manuscript. We also express sincere thanks to two anonymous reviewers for the critical comments and useful suggestions that have helped us to improve our manuscript considerably. This study was supported by the research fund of Japanese Government, Ministry of Aquaculture, Forestry and Fisheries during 2010–2012. The title of fund is “A research program aimed at utilizing advanced technologies in agriculture, forestry and fisheries: MAFF, Japan” (No. #22065).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miyake, T., Isowa, K., Ishikawa, T. et al. Evaluation of genetic characteristics of wild and cultured populations of the Japanese pearl oyster Pinctada fucata martensii by using AFLP markers. Aquacult Int 24, 537–548 (2016). https://doi.org/10.1007/s10499-015-9943-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-015-9943-2