Abstract
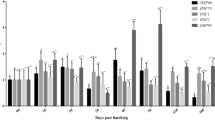
The brine shrimp Artemia franciscana is an important live feed for fish and shellfish larviculture. Cysts of Artemia are naturally found in a wide variety of harsh environments worldwide where they are exposed to different changing environmental conditions. Artemia cysts are also exposed to different hydration/dehydration (H/D) conditions during the post-harvest processing period in the Artemia processing industry. In this study, cysts of two strains of A. franciscana originating from two different geographical locations—one a natural population from Great Salt Lake (GSL), Utah, USA, and the other cultured in the Vinh Chau (VC) salt ponds, Vietnam—where they experience different environmental factors, were exposed to successive hydration/dehydration (H/D) cycles with the aim of determining the effects of these conditions on the stress (i.e., induction of Hsp70) and functional (i.e., resistance toward abiotic and pathogenic biotic factors) responses of the emerged nauplii. Our results showed that a short period of H/D of the cysts did not appear to have a deleterious effect on the emerged nauplii of both strains, as was evidenced by the absence of significant difference in the survival of nauplii emerged from the control and treated cysts, upon challenge with a thermal stressor or with pathogenic Vibrio campbellii. A limited exposure to H/D treatment even leads to the induction of enhanced thermotolerance in GSL nauplii. In essence, these observations add some insights to our current understanding of stress responses in Artemia under the described experimental conditions. In addition, the impact of H/D cycles associated with stress response should be taken into consideration when Artemia is considered as a model organism for future research and applications.

Similar content being viewed by others
References
Baruah K, Ranjan J, Sorgeloos P, Bossier P (2010) Efficacy of heterologous and homologous heat shock protein 70s as protective agents to Artemia franciscana challenged with Vibrio campbellii. Fish Shellfish Immunol 29:733–739
Baruah K, Ranjan JK, Sorgeloos P, MacRae T, Bossier P (2011) Priming the prophenoloxidase system of Artemia franciscana by heat shock proteins protects against Vibrio campbellii challenge. Fish Shellfish Immunol 31:134–141
Baruah K, Norouzitallab P, Roberts RJ, Sorgeloos P, Bossier P (2012) A novel heat-shock protein inducer triggers heat shock protein 70 to protect Artemia franciscana against abiotic stressors. Aquaculture 334–337:152–158
Baruah K, Norouzitallab P, Shihao L, Sorgeloos P, Bossier P (2013) Feeding truncated heat shock protein 70 s protect Artemia franciscana against virulent Vibrio campbellii challenge. Fish Shellfish Immunol 34:183–191
Baruah K, Norouzitallab P, Linayati L, Sorgeloos P, Bossier P (2014) Reactive oxygen species generated by a heat shock protein (Hsp) inducing product contributes to Hsp70 production and Hsp70-mediated protective immunity in Artemia franciscana against pathogenic vibrios. Dev Comp Immunol 46:470–479
Baruah K, Huy TT, Norouzitallab P, Niu Y, Gupta SK, De Schryver P, Bossier P (2015) Probing the protective mechanism of poly-ß-hydroxybutyrate against vibriosis by using gnotobiotic Artemia franciscana and Vibrio campbellii as host-pathogen model. Sci Rep 5:9427. doi:10.1038/srep09427
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chrousos GP, Gold PW (1992) The concepts of stress and stress system disorders. Overview of physical and behavioural homeostasis. J Am Med Assoc 267:1244–1252
Clegg JS (2007) Protein stability in Artemia embryos during prolonged anoxia. Biol Bull 212:74–81
Clegg JS, Willsie JK, Jackson SA (1999) Adaptive significance of a small heat shock/α-crystallin protein in encysted embryos of the brine shrimp, Artemia franciscana. Am Zool 39:836–847
Clegg JS, Jackson SA, Hoa NV, Sorgeloos P (2000) Thermal resistance, development rate and heat shock proteins in Artemia franciscana, from San Francisco Bay and Southern Vietnam. J Exp Mar Biol Ecol 252:85–96
Defoirdt T, Crab R, Wood TK, Sorgeloos P, Verstrate W, Bossier P (2006a) Quorum sensing-disrupting brominated furanones protect the gnotobiotic brine shrimp Artemia franciscana from pathogenic Vibrio harveyi, Vibrio campbellii, and Vibrio parahaemolyticus Isolates. Appl Environ Microbiol 72:6419–6423
Defoirdt T, Halet D, Sorgeloos P, Bossier P, Verstraete W (2006b) Short-chain fatty acids protect gnotobiotic Artemia franciscana from pathogenic Vibrio campbellii. Aquaculture 261:804–808
Defoirdt T, Halet D, Boon N, Van De Wiele T, Sorgeloos P, Bossier P, Verstraete W (2007) The bacterial storage compound poly-b-hydroxybutyrate protects Artemia franciscana from pathogenic Vibrio campbellii. Environ Microbiol 9:445–452
El-Magsodi M, Bossier P, Sorgeloos P, Van Stappen G (2014) Hatching and nutritional quality of Artemia cysts progressively deteriorates as a function of increased exposure to hydration/dehydration cycles. Aquac Int 22:1515–1532
Haldar S, Chatterjee S, Sugimoto N, Das S, Chowdhury N, Hinenoya A et al (2011) Identification of Vibrio campbellii isolated from diseased farm-shrimps from south India and establishment of its pathogenic potential in an Artemia model. Microbiology 157:179–188
Hand SC, Menze MA, Borcar A, Patil Y, Covi JA et al (2011) Metabolic restructuring during energy-limited states: insights from Artemia franciscana embryos and other animals. J Insect Physiol 57:584–594
Kellogg VL (1906) A new Artemia its life conditions. Science 24:594–596
King AM, MacRae TH (2012) The small heat shock protein p26 aids development of encysting Artemia embryos, prevents spontaneous diapause termination and protects against stress. PLoS One 7:e43723
Kiss AJ, Muir TJ, JrRE Lee, Costanzo JP (2011) Seasonal variation in the hepatoproteome of the dehydration and free-tolerant wood frog, Ranasylvatica. Int J Mol Sci 12:8406–8414
Lavens P, Sorgeloos P (1987) The cryptobiotic state of Artemia cysts, its diapause deactivation and hatching: 27-63. In: Sorgeloos P, Bengtson DA, Decleir W, Jaspers E (eds) Artemia research and its applications, vol 3. Universa Press, Wetteren
Liang P, MacRae TH (1999) The synthesis of a small heat shock/α-crystallin protein in Artemia and its relationship to stress tolerance during development. Dev Biol 207:445–456
Lindquist S (1992) Heat shock proteins and stress tolerance in microorganisms. Curr Opin Genet Dev 2:748–755
Lindquist S, Craig EA (1988) The heat-shock proteins. Annu Rev Genet 22:631–677
MacRae TH (2003) Molecular chaperones, stress resistance and development in Artemia franciscana. Semin Cell Dev Biol 14:251–258
MacRae TH (2010) Gene expression, metabolic regulation and stress tolerance during diapause. Cell Mol Life Sci 67:2405–2424
Marques A, Dhont J, Sorgeloos P, Bossier P (2006a) Immunostimulatory nature of β-glucans and baker’s yeast in the challenge test of Artemia. Fish Shellfish Immunol 20:682–692
Marques A, Ollevier F, Verstraete W, Sorgeloos P, Bossier P (2006b) Gnotobiotically grown aquatic animals: opportunities to investigate host-microbe interactions. J Appl Microbiol 100:903–918
Mercier L, Palacios E, Campa-Còrdova AI, Tovar-Ramirezm D, Hernàndez-Herrera R, Racotta IS (2006) Metabolic and immune responses in pacific white leg shrimp Litopenaeus vannamei exposed to a repeated handling stress. Aquaculture 258:633–640
Ohtsuka K, Kawashima D, Asai M (2007) Dual functions of heat shock proteins: molecular chaperones inside of cells and danger signals outside of cells. Therm Med 23:11–22
Parsell DA, Linquist S (1993) The function of heat shock proteins in stress tolerance: degradation and reactivation of damaged proteins. Annu Rev Genet 27:437–496
Roberts RJ, Agius C, Saliba C, Bossier P, Sung YT (2010) Heat shock proteins (chaperones) in fish and shellfish and their potential role in relation to fish health: a review. J Fish Dis 33:789–801
Song LS, Wu LT, Ni DJ, Chang YQ, Xu W, Xing KZ (2006) The cDNA cloning and mRNA expression of heat shock protein 70 gene in the haemocytes of bay scallop (Argopectenirradians, Lamarck 1819) responding to bacteria challenge and naphthalin stress. Fish Shellfish Immunol 21:335–345
Sorgeloos P, Lavens P, Léger P, Tackaert W, Versichele D (1986) Manual for the culture and use of brine shrimp Artemia in aquaculture. In: FAO Fisheries Technical Paper No. 361 (ed.) Food and Agriculture Organization Rome, Italy, pp 91–95
Sorgeloos P, Dhert P, Candreva P (2001) Use of brine shrimp, Artemia sp., in marine fish larviculture. Aquaculture 200:147–159
Sung YY, Van Damme EJM, Sorgeloos P, Bossier P (2007) Non-lethal heat shock protects gnotobiotic Artemia franciscana larvae against virulent vibrios. Fish Shellfish Immunol 22:318–326
Sung YY, Roberts RJ, Bossier P (2011a) Enhancement of Hsp70 synthesis protects common carp Cyprinus carpio L. against lethal ammonia toxicity. J Fish Dis 35:563–568
Sung YY, MacRae TH, Sorgeloos P, Bossier P (2011b) Stress response for disease control in aquaculture. Rev Aquac 3:120–137
Tort L (2011) Stress and immune modulation in fish. Dev Comp Immunol 35:1366–1375
Triantaphyllidis GV, Abatzopoulos TJ, Sorgeloos P (1998) Review of the biogeography of the genus Artemia (Crustacea, Anostraca). J Biogeogr 25:213–226
Van Stappen G (1996) Artemia: Use of cysts. In: Manual on the production and use of live food for aquaculture (ed.) FAO Fisheries Technical Paper No. 361 Food and Agriculture Organization Rome, Italy, pp 107–136
Vanhaecke P, Sorgeloos P (1982) International Study on Artemia. XVIII. The hatching rate of Artemia cysts—a comparative study. Aquac Eng 1:263–273
Acknowledgments
The authors are grateful to the Ministry of Higher Education of the Libyan Government who supported this study through a doctoral Grant to the first author. The author Kartik Baruah is a postdoctoral fellow of Research Foundation Flanders (FWO; Brussels, Belgium).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
El-Magsodi, M.O., Baruah, K., Norouzitallab, P. et al. Hydration/dehydration cycles imposed on Artemia cysts influence the tolerance limit of nauplii against abiotic and biotic stressors. Aquacult Int 24, 429–439 (2016). https://doi.org/10.1007/s10499-015-9935-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-015-9935-2