Abstract
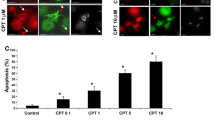
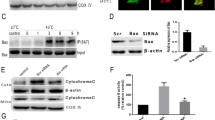
Apoptotic microtubule network (AMN) is organized during apoptosis, forming a cortical structure beneath the plasma membrane which plays a critical role in preserving cell morphology and plasma membrane integrity. The aim of this study was to examine the effect of cold/warming exposure on apoptotic microtubules and plasma membrane integrity during the execution phase of apoptosis. We demonstrated in camptothecin-induced apoptotic H460 cells that cold/warming exposure disorganized apoptotic microtubules and allowed the access of active caspases to the cellular cortex and the cleavage of essential proteins in the preservation of plasma membrane permeability. Cleavage of cellular cortex and plasma membrane proteins, such as α-spectrin, paxilin, focal adhesion kinase and calcium ATPase pump (PMCA-4) involved in cell calcium extrusion resulted in increased plasma permeability and calcium overload leading apoptotic cells to secondary necrosis. The essential role of caspase-mediated cleavage in this process was demonstrated because the addition of the pan-caspase inhibitor z-VAD during cold/warming exposure that induces AMN depolymerization avoided the cleavage of cortical and plasma membrane proteins and prevented apoptotic cells to undergo secondary necrosis. Likewise, apoptotic microtubules stabilization by taxol during cold/warming exposure also prevented cellular cortex and plasma membrane protein cleavage and secondary necrosis. Furthermore, microtubules stabilization or caspase inhibition during cold/warming exposure was also critical for proper phosphatidylserine externalization and apoptotic cell clearance by macrophages. These results indicate that cold/warming exposure of apoptotic cells induces secondary necrosis which can be prevented by both, microtubule stabilization or caspase inhibition.






Similar content being viewed by others
Abbreviations
- AMN:
-
Apoptotic microtubule network
- CPT:
-
Camptothecin
- CYTO:
-
Cytochalasin
- ΔΨm:
-
Mitochondria membrane potential
- FAK:
-
Focal adhesion kinase
- GADPH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- LDH:
-
Lactic dehydrogenase
- PMCA-4:
-
Plasma membrane Ca2+ ATPase
- PS:
-
Phosphadidylserine
- ROCK-1:
-
Rho-associated, coiled-coil containing protein kinase 1
References
Kerr JF, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26(4):239–257
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407(6805):770–776
Hammill AK, Uhr JW, Scheuermann RH (1999) Annexin V staining due to loss of membrane asymmetry can be reversible and precede commitment to apoptotic death. Exp Cell Res 251(1):16–21
Savill J, Dransfield I, Gregory C, Haslett C (2002) A blast from the past: clearance of apoptotic cells regulates immune responses. Nat Rev Immunol 2(12):965–975
Zong WX, Thompson CB (2006) Necrotic death as a cell fate. Genes Dev 20(1):1–15
Kaczmarek A, Vandenabeele P, Krysko DV (2013) Necroptosis: the release of damage-associated molecular patterns and its physiological relevance. Immunity 38(2):209–223
Mills JC, Stone NL, Pittman RN (1999) Extranuclear apoptosis. The role of the cytoplasm in the execution phase. J Cell Biol 146(4):703–708
Pittman S, Geyp M, Fraser M, Ellem K, Peaston A, Ireland C (1997) Multiple centrosomal microtubule organising centres and increased microtubule stability are early features of VP-16-induced apoptosis in CCRF-CEM cells. Leuk Res 21(6):491–499
Pittman SM, Strickland D, Ireland CM (1994) Polymerization of tubulin in apoptotic cells is not cell cycle dependent. Exp Cell Res 215(2):263–272
Moss DK, Betin VM, Malesinski SD, Lane JD (2006) A novel role for microtubules in apoptotic chromatin dynamics and cellular fragmentation. J Cell Sci 119(Pt 11):2362–2374
Moss DK, Lane JD (2006) Microtubules: forgotten players in the apoptotic execution phase. Trends Cell Biol 16(7):330–338
Sanchez-Alcazar JA, Rodriguez-Hernandez A, Cordero MD, Fernandez-Ayala DJ, Brea-Calvo G, Garcia K, Navas P (2007) The apoptotic microtubule network preserves plasma membrane integrity during the execution phase of apoptosis. Apoptosis 12(7):1195–1208
Moss DK, Wilde A, Lane JD (2009) Dynamic release of nuclear RanGTP triggers TPX2-dependent microtubule assembly during the apoptotic execution phase. J Cell Sci 122(Pt 5):644–655
Oropesa M, de la Mata M, Maraver JG, Cordero MD, Cotan D, Rodriguez-Hernandez A, Dominguez-Monino I, de Miguel M, Navas P, Sanchez-Alcazar JA (2011) Apoptotic microtubule network organization and maintenance depend on high cellular ATP levels and energized mitochondria. Apoptosis 16(4):404–424
Oropesa-Avila M, Fernandez-Vega A, de la Mata M, Maraver JG, Cordero MD, Cotan D, de Miguel M, Calero CP, Paz MV, Pavon AD, Sanchez MA, Zaderenko AP, Ybot-Gonzalez P, Sanchez-Alcazar JA (2013) Apoptotic microtubules delimit an active caspase free area in the cellular cortex during the execution phase of apoptosis. Cell Death Dis 4:e527. doi:10.1038/cddis.2013.58
Huot J, Houle F, Rousseau S, Deschesnes RG, Shah GM, Landry J (1998) SAPK2/p38-dependent F-actin reorganization regulates early membrane blebbing during stress-induced apoptosis. J Cell Biol 143(5):1361–1373
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Akhmanova A, Steinmetz MO (2008) Tracking the ends: a dynamic protein network controls the fate of microtubule tips. Nat Rev Mol Cell Biol 9(4):309–322
Wade RH (2009) On and around microtubules: an overview. Mol Biotechnol 43(2):177–191
Lee JC, Timasheff SN (1977) In vitro reconstitution of calf brain microtubules: effects of solution variables. Biochemistry 16(8):1754–1764
Olmsted JB, Borisy GG (1975) Ionic and nucleotide requirements for microtubule polymerization in vitro. Biochemistry 14(13):2996–3005
Regula CS, Pfeiffer JR, Berlin RD (1981) Microtubule assembly and disassembly at alkaline pH. J Cell Biol 89(1):45–53
Suprenant KA, Marsh JC (1987) Temperature and pH govern the self-assembly of microtubules from unfertilized sea-urchin egg extracts. J Cell Sci 87(Pt 1):71–84
Fygenson DK, Braun E, Libchaber A (1994) Phase diagram of microtubules. Phys Rev E Stat Phys Plasmas Fluids Relat Interdiscip Topics 50(2):1579–1588
Modig C, Wallin M, Olsson PE (2000) Expression of cold-adapted beta-tubulins confer cold-tolerance to human cellular microtubules. Biochem Biophys Res Commun 269(3):787–791
Afford S, Randhawa S (2000) Demystified: apoptosis. Mol Pathol 53(2):55–63
Silva MT, do Vale A, dos Santos NM (2008) Secondary necrosis in multicellular animals: an outcome of apoptosis with pathogenic implications. Apoptosis 13(4):463–482
Silva MT (2010) Secondary necrosis: the natural outcome of the complete apoptotic program. FEBS Lett 584(22):4491–4499
Poon IK, Hulett MD, Parish CR (2010) Molecular mechanisms of late apoptotic/necrotic cell clearance. Cell Death Differ 17(3):381–397
Fadok VA, Bratton DL, Konowal A, Freed PW, Westcott JY, Henson PM (1998) Macrophages that have ingested apoptotic cells in vitro inhibit proinflammatory cytokine production through autocrine/paracrine mechanisms involving TGF-beta, PGE2, and PAF. J Clin Invest 101(4):890–898
Serhan CN, Savill J (2005) Resolution of inflammation: the beginning programs the end. Nat Immunol 6(12):1191–1197
Krysko DV, D’Herde K, Vandenabeele P (2006) Clearance of apoptotic and necrotic cells and its immunological consequences. Apoptosis 11(10):1709–1726
Gaipl US, Munoz LE, Grossmayer G, Lauber K, Franz S, Sarter K, Voll RE, Winkler T, Kuhn A, Kalden J, Kern P, Herrmann M (2007) Clearance deficiency and systemic lupus erythematosus (SLE). J Autoimmun 28(2–3):114–121
Unal-Cevik I, Kilinc M, Can A, Gursoy-Ozdemir Y, Dalkara T (2004) Apoptotic and necrotic death mechanisms are concomitantly activated in the same cell after cerebral ischemia. Stroke 35(9):2189–2194
Tabas I (2004) Apoptosis and plaque destabilization in atherosclerosis: the role of macrophage apoptosis induced by cholesterol. Cell Death Differ 11(Suppl 1):S12–S16. doi:10.1038/sj.cdd.4401444
Mukaro VR, Hodge S (2011) Airway clearance of apoptotic cells in COPD. Curr Drug Targets 12(4):460–468
Kirkham P (2007) Oxidative stress and macrophage function: a failure to resolve the inflammatory response. Biochem Soc Trans 35(Pt 2):284–287
Vandivier RW, Fadok VA, Hoffmann PR, Bratton DL, Penvari C, Brown KK, Brain JD, Accurso FJ, Henson PM (2002) Elastase-mediated phosphatidylserine receptor cleavage impairs apoptotic cell clearance in cystic fibrosis and bronchiectasis. J Clin Invest 109(5):661–670
Uller L, Persson CG, Erjefalt JS (2006) Resolution of airway disease: removal of inflammatory cells through apoptosis, egression or both? Trends Pharmacol Sci 27(9):461–466
Watt AP, Brown V, Courtney J, Kelly M, Garske L, Elborn JS, Ennis M (2004) Neutrophil apoptosis, proinflammatory mediators and cell counts in bronchiectasis. Thorax 59(3):231–236
Silva MT (2010) Bacteria-induced phagocyte secondary necrosis as a pathogenicity mechanism. J Leukoc Biol 88(5):885–896
Savill J, Fadok V (2000) Corpse clearance defines the meaning of cell death. Nature 407(6805):784–788
Voll RE, Herrmann M, Roth EA, Stach C, Kalden JR, Girkontaite I (1997) Immunosuppressive effects of apoptotic cells. Nature 390(6658):350–351
Fadok VA, Bratton DL, Frasch SC, Warner ML, Henson PM (1998) The role of phosphatidylserine in recognition of apoptotic cells by phagocytes. Cell Death Differ 5(7):551–562
Castedo M, Hirsch T, Susin SA, Zamzami N, Marchetti P, Macho A, Kroemer G (1996) Sequential acquisition of mitochondrial and plasma membrane alterations during early lymphocyte apoptosis. J Immunol 157(2):512–521
Nicotera P, Leist M, Ferrando-May E (1998) Intracellular ATP: a switch in the decision between apoptosis and necrosis. Toxicol Lett 102–103:139–142
Moreira ME, Barcinski MA (2004) Apoptotic cell and phagocyte interplay: recognition and consequences in different cell systems. An Acad Bras Cienc 76(1):93–115
Acknowledgments
This work was supported by FIS PI10/00543 Grant, FIS EC08/00076 Grant, Ministerio de Sanidad, Spain and Fondo Europeo de Desarrollo Regional (FEDER-Unión Europea), SAS 111242 Grant, Servicio Andaluz de Salud-Junta de Andalucía, Proyecto de Investigación de Excelencia de la Junta de Andalucía CTS-5725, and by Asociación de Enfermos de Patología Mitocondrial (AEPMI).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Oropesa-Ávila, M., Fernández-Vega, A., de la Mata, M. et al. Apoptotic cells subjected to cold/warming exposure disorganize apoptotic microtubule network and undergo secondary necrosis. Apoptosis 19, 1364–1377 (2014). https://doi.org/10.1007/s10495-014-1015-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-014-1015-y