Abstract
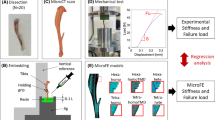
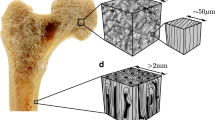
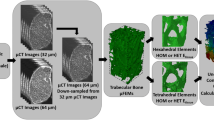
Image-based modeling is a popular approach to perform patient-specific biomechanical simulations. Accurate modeling is critical for orthopedic application to evaluate implant design and surgical planning. It has been shown that bone strength can be estimated from the bone mineral density (BMD) and trabecular bone architecture. However, these findings cannot be directly and fully transferred to patient-specific modeling since only BMD can be derived from clinical CT. Therefore, the objective of this study was to propose a method to predict the trabecular bone structure using a µCT atlas and an image registration technique. The approach has been evaluated on femurs and patellae under physiological loading. The displacement and ultimate force for femurs loaded in stance position were predicted with an error of 2.5% and 3.7%, respectively, while predictions obtained with an isotropic material resulted in errors of 7.3% and 6.9%. Similar results were obtained for the patella, where the strain predicted using the registration approach resulted in an improved mean squared error compared to the isotropic model. We conclude that the registration of anisotropic information from of a single template bone enables more accurate patient-specific simulations from clinical image datasets than isotropic model.








Similar content being viewed by others
References
Bonaretti, S., C. Seiler, C. Boichon, M. Reyes, and P. Büchler. Image-based vs. mesh-based statistical appearance models of the human femur: implications for finite element simulations. Med. Eng. Phys. 36:1626–1635, 2014.
Boutroy, S., M. L. Bouxsein, F. Munoz, and P. D. Delmas. In vivo assessment of trabecular bone microarchitecture by high-resolution peripheral quantitative computed tomography. J. Clin. Endocrinol. Metab. 90:6508–6515, 2005.
Burghardt, A. J., T. M. Link, and S. Majumdar. High-resolution computed tomography for clinical imaging of bone microarchitecture. Clin. Orthop. Relat. Res. 469:2179–2193, 2011.
Doblaré, M., and J. M. García. Application of an anisotropic bone-remodelling model based on a damage-repair theory to the analysis of the proximal femur before and after total hip replacement. J. Biomech. 34:1157–1170, 2001.
Dragomir-Daescu, D., S. McEligot, Y. Dai, R. C. Entwistle, C. Salas, L. J. Melton, K. E. Bennet, S. Khosla, and S. Amin. Robust QCT/FEA models of proximal femur stiffness and fracture load during a sideways fall on the hip. Ann. Biomed. Eng. 39:742–755, 2011.
Enns-Bray, W. S., J. S. Owoc, K. K. Nishiyama, and S. K. Boyd. Mapping anisotropy of the proximal femur for enhanced image based finite element analysis. J. Biomech. 47:3272–3278, 2014.
Harrigan, T. P., and R. W. Mann. Characterization of microstructural anisotropy in orthotropic materials using a second rank tensor. J. Mater. Sci. 19:761–767, 1984.
Hazrati Marangalou, J., K. Ito, M. Cataldi, F. Taddei, and B. van Rietbergen. A novel approach to estimate trabecular bone anisotropy using a database approach. J. Biomech. 46:2356–2362, 2013.
Hazrati Marangalou, J., K. Ito, and B. van Rietbergen. A novel approach to estimate trabecular bone anisotropy from stress tensors. Biomech. Model. Mechanobiol. 2014. doi:10.1007/s10237-014-0584-6.
Hellmich, C., C. Kober, and B. Erdmann. Micromechanics-based conversion of CT data into anisotropic elasticity tensors, applied to FE simulations of a mandible. Ann. Biomed. Eng. 36:108–122, 2008.
Hellmich, C., F.-J. Ulm, and L. Dormieux. Can the diverse elastic properties of trabecular and cortical bone be attributed to only a few tissue-independent phase properties and their interactions? Arguments from a multiscale approach. Biomech. Model. Mechanobiol. 2:219–238, 2004.
Horn, B. K. P. Closed-form solution of absolute orientation using unit quaternions. J. Opt. Soc. Am. A 4:629, 1987.
Ino, F., Y. Kawasaki, T. Tashiro, Y. Nakajima, Y. Sato, S. Tamura, and K. Hagihara. A parallel implementation of 2D/3D image registration for computer-assisted surgery. Int. J. Bioinform. Res. Appl. 2:341–358, 2006.
Kersh, M. E., P. K. Zysset, D. H. Pahr, U. Wolfram, D. Larsson, and M. G. Pandy. Measurement of structural anisotropy in femoral trabecular bone using clinical-resolution CT images. J. Biomech. 46:2659–2666, 2013.
Klein, S., M. Staring, K. Murphy, M. A. Viergever, and J. P. W. Pluim. Elastix: a toolbox for intensity-based medical image registration. IEEE Trans. Med. Imaging 29:196–205, 2010.
Kober, C., B. Erdmann, C. Hellmich, R. Sader, and H.-F. Zeilhofer. Consideration of anisotropic elasticity minimizes volumetric rather than shear deformation in human mandible. Comput. Methods Biomech. Biomed. Eng. 9:91–101, 2006.
Larsson, D., B. Luisier, M. E. Kersh, E. Dall’ara, P. K. Zysset, M. G. Pandy, and D. H. Pahr. Assessment of transverse isotropy in clinical-level CT images of trabecular bone using the gradient structure tensor. Ann. Biomed. Eng. 42:950–959, 2014.
Latypova, A., F. Levrero, D. Pioletti, B. Jolles, and A. Terrier. A musculoskeletal numerical knee model to assess patellar resurfacing in total knee arthroplasty. 2013. At <http://www.ors.org/Transactions/59/PS2–099/1709.html>.
Le Minh, H., W. M. Park, K. Kim, S.-W. Son, S.-H. Lee, and Y. H. Kim. A new patient-specific planning method based on joint contact force balance with soft tissue release in total knee arthroplasty. Int. J. Precis. Eng. Manuf. 14:2193–2199, 2013.
Lekadir, K., J. Hazrati-Marangalou, C. Hoogendoorn, Z. Taylor, B. van Rietbergen, and A. F. Frangi. Statistical estimation of femur micro-architecture using optimal shape and density predictors. J. Biomech. 48:598–603, 2015.
Luisier, B., E. Dall’Ara, and D. H. Pahr. Orthotropic HR-pQCT-based FE models improve strength predictions for stance but not for side-way fall loading compared to isotropic QCT-based FE models of human femurs. J. Mech. Behav. Biomed. Mater. 32:287–299, 2014.
Maquer, G., S. N. Musy, J. Wandel, T. Gross, and P. K. Zysset. Bone volume fraction and fabric anisotropy are better determinants of trabecular bone stiffness than other morphological variables. J. Bone Miner. Res. 30:1000–1008, 2015.
Muller, R., T. Hildebrand, and P. Ruegsegger. Non-invasive bone biopsy: a new method to analyse and display the three-dimensional structure of trabecular bone. Phys. Med. Biol. 39:145–164, 1994.
Ourselin, S., A. Roche, S. Prima, and N. Ayache. Block Matching: A General Framework to Improve Robustness of Rigid Registration of Medical Images. In: Medical image computing and computer-assisted interventio—MICCAI 2000 SE-57, edited by S. Delp, A. DiGoia, and B. Jaramaz. Berlin Heidelberg: Springer, 2000, pp. 557–566.
Pahr, D. H., and P. K. Zysset. From high-resolution CT data to finite element models: development of an integrated modular framework. Comput. Methods Biomech. Biomed. Eng. 12:45–57, 2009.
Rueckert, D., L. I. Sonoda, C. Hayes, D. L. Hill, M. O. Leach, and D. J. Hawkes. Nonrigid registration using free-form deformations: application to breast MR images. IEEE Trans. Med. Imaging 18:712–721, 1999.
San Antonio, T., M. Ciaccia, C. Müller-Karger, and E. Casanova. Orientation of orthotropic material properties in a femur FE model: a method based on the principal stresses directions. Med. Eng. Phys. 34:914–919, 2012.
Schwiedrzik, J. J., and P. K. Zysset. An anisotropic elastic–viscoplastic damage model for bone tissue. Biomech. Model. Mechanobiol. 12:201–213, 2013.
Seiler, C., X. Pennec, and M. Reyes. Capturing the multiscale anatomical shape variability with polyaffine transformation trees. Med. Image Anal. 16:1371–1384, 2012.
Tabor, Z., and E. Rokita. Quantifying anisotropy of trabecular bone from gray-level images. Bone 40:966–972, 2007.
Takahashi, A., H. Sano, M. Ohnuma, M. Kashiwaba, D. Chiba, M. Kamimura, T. Sugita, and E. Itoi. Patellar morphology and femoral component geometry influence patellofemoral contact stress in total knee arthroplasty without patellar resurfacing. Knee Surg. Sports Traumatol. Arthrosc. 20:1787–1795, 2012.
The CGAL Project. CGAL User and Reference Manual. CGAL Editorial Board, 2015. At <http://doc.cgal.org/4.6/Manual/packages.html>.
Trabelsi, N., and Z. Yosibash. Patient-specific finite-element analyses of the proximal femur with orthotropic material properties validated by experiments. J. Biomech. Eng. 133:061001, 2011.
Wolfram, U., B. Schmitz, F. Heuer, M. Reinehr, and H.-J. Wilke. Vertebral trabecular main direction can be determined from clinical CT datasets using the gradient structure tensor and not the inertia tensor—a case study. J. Biomech. 42:1390–1396, 2009.
Zysset, P. K., and A. Curnier. An alternative model for anisotropic elasticity based on fabric tensors. Mech. Mater. 21:243–250, 1995.
Acknowledgment
The authors would like to thank Dr. Enrico Dall’Ara for preparing, scanning and sharing the femoral data, Dr. Jakob Schwiedrzik for providing the UMAT implementation of the mechanical model, Prof. Dieter Pahr for giving us access to the Medtool software and Dr. Ghislain Maquer and Dr. Hadi Seyed Hosseini for their assistance on the preparation of the FE models.
Funding
The authors have no commercial, proprietary, or financial interest in any products or companies described in this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Amit Gefen oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Taghizadeh, E., Reyes, M., Zysset, P. et al. Biomechanical Role of Bone Anisotropy Estimated on Clinical CT Scans by Image Registration. Ann Biomed Eng 44, 2505–2517 (2016). https://doi.org/10.1007/s10439-016-1551-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-016-1551-4