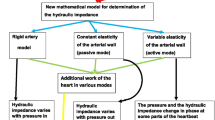
Abstract
In many studies, the functional state of vessels of different caliber was determined by fitting the lumped parameters of a mathematical model of the bed in order to fit the vascular input impedance (Z in) data. However, reliability of the results obtained in such a way remains uncertain. In this study, we employed a mathematical model with seven lumped parameters and Z in experimental data to analyze the distribution of resistance across the arterial bed of the hind limb in anesthetized cats, to test reliability of this distribution and to describe the process of ascending arterial dilation followed occlusion of iliac artery. The vascular bed was divided into three segments: large arteries, medium-sized arterial vessels and precapillary resistance vessels together with venous part of the bed. Based on the data of Z in measured in a wide frequency range (from 0 to 150 Hz) we showed that pharmacologically induced constriction and dilation of the arterial microvessels were reflected in the model by the changes in the resistance of distal precapillary vessels only, whereas the local constriction or dilation of femoral and iliac arteries as well as artificial stenosis of the femoral artery resulted exclusively in the changes of the resistance describing the state of large arteries. Using the input impedance method we could demonstrate and quantitatively describe the process of ascending arterial dilation during the post-occlusion (reactive) hyperemia. All these results prove that the model of vascular bed with seven lumped elements used in combination with input hydraulic impedance data can be an effective tool permitted to quantitatively analyze the functional state of arterial vessels of different caliber and to describe the changes in resistance of arterial vessels during vascular reactions.








Similar content being viewed by others
References
Balashov, S. A., A. M. Melkumyants, A Yu Naumov, and K. V. Sheptutsolov. Determination of the parameters of hydraulic models of vascular bed by the input impedance method. Fluid Dyn. 36(1):48–56, 2001.
Bard, Y. Nonlinear Parameter Estimation. New York: Academic Press, 1974. 341 pp.
Bauer, R. D., R. Busse, and A. Schabert. The input impedance of the peripheral vascular termination in skeletal muscle. Pflugers Arch. 403:308–311, 1985.
Bendat, J. S., and A. G. Piersol. Engineering Applications of Correlation and Spectral Analysis. New York, John Wiley & Sons, 1980, 315 pp.
Burattini, R., and P. O. Di Salvia. Development of systemic arterial mechanical properties from infancy to adulthood interpreted by four-element windkessel models. J. Appl. Physiol. 103:66–79, 2006.
Burattini, R., R. Fogliardi, and K. B. Campbell. Lumped model of terminal aortic impedance in the dog. Ann. Biomed. Eng. 22:381–391, 1994.
Burattini, R., and G. Gnudi. Computer identification of models for the arterial tree input impedance: comparison between two new simple models and first experimental results. Med. Biol. Eng. Comput. 20:134–144, 1982.
Draperm, N. R., H. Smith. Applied Regression Analysis. New York: Wiley & Sons, Inc., 2nd ed., 1981, 709 pp.
Fenger-Gron, J., M. J. Mulvany, and K. L. Christensen. Mesenteric blood pressure profile of conscious, freely moving rats. J. Physiol. 488:753–760, 1995.
Folkow, B., R. R. Sonnenshein, and D. L. Wright. Loci of neurogenic and metabolic effects on precapillary vessels of skeletal muscle. Acta Physiol. Scand. 81(4):459–471, 1971.
Fronek, K., and B. W. Zweifach. Microvascular pressure distribution in skeletal muscle and vasodilatation. Am. J. Physiol. 228:791–796, 1975.
Gessner, U. Vascular input impedance. In: Cardiovascular Fluid Dynamics, edited by D. H. Bergel. London: Academic Press, 1972, Vol. 1, Chap. 10, pp. 315–349.
Kartamyshev, S. P., S. A. Balashov, and A. M. Melkumyants. Role of endothelium sensitivity to shear stress in noradrenaline-induced constriction of feline femoral arterial bed under constant flow and constant pressure perfusions. J. Vasc. Res. 44:1–10, 2007.
Khayutin, V. M., V. P. Nikolsky, A. N. Rogoza, and E. V. Lukoshkova. Endothelium determines stabilization of the pressure drop in arteries. Acta Physiol. Scand. 148:295–304, 1993.
Khayutin, V. M. Active arterial function: prompt adaptation of the vascular lumen to the blood flow and viscosity. In: Contemporary Problems of Biomechanics, edited by G. G. Chernyi and S. A. Regirer. Moscow: Mir Publ. & Boca Raton: CRC Press, 1990, pp. 142–207.
Lutchen, K. R., and A. C. Jackson. Reliability of parameter estimates from models applied to respiratory impedance data. J. Appl. Physiol. 62(2):403–413, 1987.
Marquardt, D. W. An algorithm for least squares estimation of nonlinear parameters –. J. Soc. Ind. Appl. Math. 11:431–441, 1963.
McDonald, D. A. Blood Flow in Arteries. London: Edward Arnold Ltd., 1960.
Melkumyants, A. M., S. A. Balashov, E. S. Veselova, and V. M. Khayutin. Continuous control of the lumen of feline conduit arteries by blood flow rate. Cardiovasc. Res. 21:863–870, 1987.
Olufsen, M. S., and A. Nadim. On deriving lumped models for blood flow and pressure in the systemic arteries. Math. Biosci. Eng. 1(1):61–80, 2004.
O’Rourke, M. F., and M. G. Taylor. Vascular impedance of the femoral bed. Circ. Res. 18:126–139, 1966.
O’Rourke, M. F., and M. G. Taylor. Input impedance of the systemic circulation. Circ. Res. 20:365–380, 1967.
Piene, H. Some physical properties of the pulmonary arterial bed deduced from pulsatile arterial flow and pressure. Acta. Physiol. Scand. 98:295–306, 1976.
Piene, H., and A. Hauge. Reduction of pulsatile hydraulic power in the pulmonary circulation caused by moderate vasoconstriction. Cardiovasc. Res. 10:503–513, 1976.
Press W. H., S. A. Teukolsky, W. T. Vetterling, and B. P. Flann, Numerical recipes in C. In: The Art of Scientific Computing, 2nd ed. Cambridge, UK: Cambridge Univ. Press, 1992.
Rogoza, A. N. A contact transducer for continuous measurement of the diameter of blood vessels. Bull. Exp. Biol. Med. 91:698–701, 1981.
Segal, S. S., and B. R. Duling. Communication between feed arteries and micro vessels in hamster striated muscle: segmental vascular responses are functionally coordinated. Circ. Res. 59:283-290, 1986.
Segers, P., S. Brimioulle, N. Stergiopulos, N. Westerhof, R. Naeije, M. Maggiorini, and P. Verdonck. Pulmonary arterial compliance in dogs and pigs: the three-element windkessel model revisited. Am. J. Physiol. 277(2):H725–H731, 1999.
Stergiopulos, N., B. E. Westerhof, and N. Westerhof. Total arterial inertance as the fourth element of the windkessel model. Am. J. Physiol. 276(1):H81–H88, 1999.
Vanderpool, R., R. Naeije, and N. C. Chesler. Impedance in isolated mouse lungs for the determination of site of action vasoactive agents and disease. Ann. Biomed. Eng. 38(5):1854–1861, 2010.
Westerhof, N., F. Bosman, C. J. De Vries, and A. Noordergraaf. Analog studies of the human systemic arterial tree. J. Biomech. 2:121–143, 1969.
Westerhof, N., G. Elzinga, and P. Sipkema. An artificial arterial system for pumping hearts. J. Appl. Physiol. 31:776–781, 1971.
Westerhof, N., J.-W. Lankhaar, and B. E. Westerhof. The arterial windkessel. Med. Biol. Eng. Comput. 47:131–141, 2009.
Acknowledgments
The work was supported by the Russian Foundation for Basic Research (Grant #13-04-01082). We are grateful to Dr. S. Revenko for valuable discussions and assistance.
Conflict of interest
The authors (A. Naumov, S. Balashov and A. Melkumyants) have no conflicts of interest (financial or otherwise).
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Scott I Simon oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Naumov, A.Y., Balashov, S.A. & Melkumyants, A.M. Use of Input Impedance to Determine Changes in the Resistance of Arterial Vessels at Different Levels in Feline Femoral Bed. Ann Biomed Eng 42, 1644–1657 (2014). https://doi.org/10.1007/s10439-014-1016-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-014-1016-6