Abstract
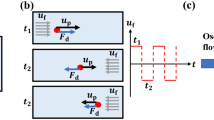
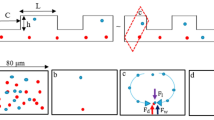
This paper provides a novel application of the optical radiation pressure for microfluidic particle transportation without precise focusing or alignment of the laser beam to the device and target. An optical manipulation of particles in a microfluidic platform is highly exploited in life science or biomedical analysis using optical tweezers with the use of a gradient force of the optical radiation pressure. Our method utilizes the other term of the radiation pressure, namely scattering force, to manipulate particles in a microchannel. The migration distance of particle depends on the amount of light received by the particle. Therefore, particle movement with long retention distance can be achieved by large-area irradiation even with low energy density. In our experiments, two proof-of-concept microfluidic chips were designed and investigated; one was a lateral particle sorting using a monolithic microfluidic chip integrated with a planar SU-8 waveguide and beam expander, the other was a vertical sorting using a 10-cm-long polydimethylsiloxane channel with whole-area irradiation. Experimental results show that 1, 2 and 5 μm polystyrene beads can be transported by the optical scattering force and that particle migration is achieved with the irradiated energy density <10 mW/mm2. The present method has practical potential for simple and fuss-free use of the optical radiation pressure without spot focusing or precise alignment process of the laser beam and damage to the device and sample.











Similar content being viewed by others
References
Airoudj A, Debarnot D, Beche B, Poncin-Epaillard F (2008) Development of an optical ammonia sensor based on polyaniline/epoxy resin (SU-8) composite. Talanta 77:1590–1596
Ashkin A (1980) Application of laser radiation of pressure. Science 210:1081–1088
Ashkin A (1992) Forces of a single-beam gradient laser trap on a dielectric sphere in the ray optics regime. Biophys J 61:569–582
Ashkin A, Dziedzic JM, Bjorkholm JE, Chu S (1986) Observation of a single-beam gradient force optical trap for dielectric particles. Opt Lett 11:288–290
Friend J, Yeo L (2010) Fabrication of microfluidic devices using polydimethylsiloxane. Biomicrofluidics 4:026592
Gascoyne PRC, Vykoukal J (2002) Particle separation by dielectrophoresis. Electrophoresis 23:1973–1983
Gossett DR, Weaver WM, Mach AJ, Hur SC, Tse HT, Lee W, Amini H, Di Carlo D (2010) Label-free cell separation and sorting in microfluidic systems. Anal Bioanal Chem 397:3249–3267
Hart SJ, Terray AV (2003) Refractive-index-driven separation of colloidal polymer particles using optical chromatography. Appl Phys Lett 83:5316–5318
Hoeb M, Radler JO, Klein S, Stutzmann Brandt MMB (2007) Light-induced dielectrophoretic manipulation of DNA. Biophys J 93:1032–1038
Hou HW, Bhagat AS, Lee WC, Huang S, Han J, Lim CT (2011) Microfluidic devices for blood fractionation. Micromachines 2:319–343
Imasaka T, Kawabata Y, Kaneta T, Ishidzu Y (1995) Optical chromatography. Anal Chem 67:1763–1765
Ishida A, Toki H, Motosuke M, Honami S (2012) Particle accumulation by AC electroosmosis in microfluidic device with co-planar electrodes. J Therm Sci Tech 7:475–486
Janasek D, Franzke J, Manz A (2006) Scaling and the design of miniaturized chemical-analysis systems. Nature 442:05059
Johnson FS (1954) The solar constant. J Meteorol 11:431–439
Kayani AA, Khoshmanesh K, Ward SA, Mitchell A, Kalantar-zadeh K (2012) Optofluidics incorporating actively controlled micro- and nano-particles. Biomicrofluidics 6:031501
Kim SB, Kim SS (2003) Radiation forces on spheres in loosely focused Gaussian beam: ray-optics regime. J Opt Soc Am B 23(5):897–903
Kim SB, Yon SY, Sung HJ, Kim SS (2008) Cross-type optical particle separation in a microchannel. Anal Chem 80:6023–6028
Kuntaegowdanahalli SS, Bhagat AS, Kumar G, Papautsky I (2009) Inertial microfluidics for continuous particle separation in spiral microchannels. Lab Chip 9:2973–2980
Lairell T, Petersson F, Nilsson A (2007) Chip integrated strategies for acoustic separation and manipulation of cells and particles. Chem Soc Rev 36:492–506
Lee KH, Kim SB, Lee KS, Sung HJ (2011) Enhancement by optical force of separation in pinched flow fractionation. Lab Chip 11:354–357
Lee KH, Lee KS, Jung JH, Chang CB, Sung HJ (2013) Optical mobility of blood cells for label-free cell separation applications. Appl Phys Lett 102:141911
Liu C, Lagae L, Borghs G (2007) Manipulation of magnetic particles on chip by magnetophoretic actuation and dielectrophoretic levitation. Appl Phys Lett 90:184109
Mogensen KB, El-Ali J, Wolff A, Kutter JP (2003) Integration of polymer waveguide for optical detection in microfabricated chemical analysis systems. Appl Opt 42(19):4072–4079
Motosuke M, Shimakawa J, Akutsu D, Honami S (2010) Noncontact manipulation of microflow by photothermal control of viscous force. Int J Heat Fluid Flow 31:1005–1011
Motosuke M, Shimakawa J, Akutsu D, Honami S (2012) Particle migration by optical scattering force in microfluidic system with light-absorbing liquid. J Heat Transf 134:051025
Myers FB, Lee LP (2008) Innovations in optical microfluidic technologies for point-of-care diagnostics. Lab Chip 8:2015–2031
Nagrath S, Sequist LV, Maheswaran S, Bell DW, Irimia D, Ulkus L, Smith MR, Kwak EL, Digumarthy S, Muzikansky A, Ryan P, Balis UJ, Tompkins RG, Haber DA, Toner M (2007) Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 450:1235–1239
Neuman KC, Block SM (2004) Optical trapping. Rev Sci Instrum 75(9):2785–2809
Nussbaum Ph, Völkel R, Herzig HP, Eisner M, Haselbeck S (1997) Design, fabrication and testing of microlens arrays for sensor and microsystems. Appl Opt 6:617–636
Ozkan M, Wang M, Ozkan C, Flynn R, Birkbeck A, Esener S (2003) Optical manipulation of objects and biological cells in microfluidics devices. Biomed Microdevices 5:61–67
Pamme N (2007) Continuous flow separations in microfluidic devices. Lab Chip 7:1644–1659
Peterman EG, Gittes F, Schmidt C (2003) Laser-induced heating in optical traps. Biophys J 84:1308–1316
Russel WB (1981) Brownian motion of small particles suspended in liquids. Annu Rev Fluid Mech 13:425–455
Sajeesh P, Sen AK (2013) Particle separation and sorting in microfluidic device: a review. Microfluid Nanofluidics. doi:10.1007/s10404-013-1291-9
Santiago JG, Wereley ST, Meinhart CD, Beebe DJ, Adrian RJ (1998) A particle image velocimetry system for microfluidics. Exp Fluid 25:316–319
Seo J, Lee LP (2003) Disposable integrated microfluidics with self-aligned planar microlenses. Sens Actuator B 99:615–622
Svoboda K, Block SM (1994) Biological applications of optical forces. Annu Rev Biomol Struct 23:247–285
Viana NB, Rocha MS, Mesquita ON, Mazolli A, Maia Neto PA, Nussenzveig HM (2007) Towards absolute calibration of optical tweezers. Phys Rev E 75:021914
Xia Y, Whitesides GM (1998) Soft lithography. Annu Rev Mater Sci 28:153–184
Yalcin SE, Sharma Qian AS, Joo SW, Baysal O (2010) Manipulating particles in microfluidics by floating electrodes. Electrophoresis 31:3711–3718
Yamada M, Seki M (2005) Hydrodynamic filtration for on-chip particle concentration and classification utilizing microfluidics. Lab Chip 5:1233–1239
Yamada M, Nakashima M, Seki M (2004) Pinched flow fractionation: continuous size separation of particle utilizing a laminar flow profile in a pinched microchannel. Anal Chem 76:5465–5471
Zhbanov A, Yang S (2011) Microfluidics blood separations through optical sorting and deterministic lateral displacement. In: Proceedings of the International Conference on Quantum, Nano and Micro Technologies, pp 103–108
Acknowledgments
This work is partially supported by Grant-in-Aid for Young Scientists (A) No. 25709013 from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan. A part of microfabrication was performed in Center for Nano Lithography and Analysis, The University of Tokyo, supported by MEXT, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kotari, H., Motosuke, M. Simple applications of microparticle transportation by tender optical scattering force. Microfluid Nanofluid 18, 549–558 (2015). https://doi.org/10.1007/s10404-014-1459-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10404-014-1459-y