Abstract
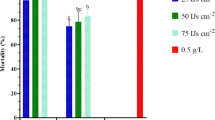
The larvae of Curculio elephas (Coleoptera: Curculionidae) and Polyphylla fullo (Coleoptera: Scarabaeidae) are major agricultural pests of chestnut and roots of cultivated crops, respectively. Previous research showed that they are relatively resistant to nematode infection. Accordingly, we evaluated the efficacy of Steinernema glaseri, S. weiseri or Heterorhabditis bacteriophora alone or in combination against these two insect pests. A nematode concentration of 50 or 100 infective juveniles (IJs)/larva for C. elephas or 50 or 100 IJs/cm2 for P. fullo at 25 °C was used. The highest (81 %) and the lowest (21 %) larval mortalities for C. elephas were obtained with S. weiseri+H. bacteriophora combined application and S. glaseri alone, respectively. The nature of the interactions (antagonism, additive, or synergy) for the larval mortality was evaluated. For C. elephas, S. weiseri combined with S. glaseri or H. bacteriophora was additive, whereas the combination of S. glaseri+H. bacteriophora was antagonistic. For P. fullo, the efficiency of nematodes used alone or combinations was very low, and there were no significant differences among the treatments at 50 or 100 IJs/cm2. The interaction was additive with the combinations of S. glaseri+H. bacteriophora against P. fullo larvae. No synergistic effect was observed for any combination against C. elephas and P. fullo larvae. Our results show that the EPN species tested, either alone or in combination, are not economically feasible for use against C. elephas or P. fullo, but that further research with other combinations of EPN species or EPNs with other entomopathogens is warranted.

Similar content being viewed by others
References
Abbott WS (1925) A method for computing the effectiveness of an insecticide. J Econ Entomol 18:265–267
Akhurst RJ (1983) Neoaplectana species: specificity of association with bacteria of the genus Xenorhabdus. Int J Syst Bacteriol 33:38–45
Alatorre-Rosas R, Kaya HK (1990) Interspecific competition between entomopathogenic nematodes in the genera Heterorhabditis and Steinernema for an insect host in sand. J Invertebr Pathol 55:179–188
Anonymous. 2008. T.C. Tarim ve Koyisleri Bakanligi Tarimsal Arastirmalar Genel Mudurlugu, Zirai Mucadele Teknik Talimatlari. Cilt 4, Ankara, 316–318
Ansari MA, Waeyenberge L, Moens M (2005) First record of Steinernema glaseri Steiner, 1929 (Rhabditida: Steinernematidae) from Belgium: a natural pathogen of Hoplia philantus (Coleoptera: Scarabeidae). Nematology 7:953–956
Avtzis DN, Cognato AI (2013) Genetic structure of Curculio elephas Gyll. (Coleoptera, Curculionidae) in Greece: an important pest of sweet chestnut. J Pest Sci 86:491–497
Barbercheck ME, Kaya HK (1990) Interactions between Beauveria bassiana and the entomogenous nematodes Steinernema feltiae and Heterorhabditis heliothidis. J Invertebr Pathol 55:225–234
Campbell JF, Lewis EE, Stock SP, Nadler S, Kaya HK (2003) Evolution of host search strategies in entomopathogenic nematodes. J Nematol 35:142–145
Choo HY, Kaya HK, Shea P, Noffsinger EM (1987) Ecological study of nematode parasitism in Ips beetles from California and Idaho. J Nematol 19:495–502
Choo HY, Koppenhöfer AM, Kaya HK (1996) Combination of two entomopathogenic nematode species for suppression of an insect pest. J Econ Entomol 89:97–103
Choo HY, Kaya HK, Huh J, Lee DW, Kim HH, Lee SM, Choo YM (2002) Entomopathogenic nematodes (Steinernema spp. and Heterorhabditis bacteriophora) and a fungus Beauveria brongniartii for biological control of the white grubs, Ectinohoplia rufipes and Exomala orientalis, in Korean golf courses. Biocontrol 47:177–192
Desouhant E (1998) Selection of fruits for oviposition by the chestnut weevil, Curculio elephas. Entomol Exp Appl 86:71–78
Dunphy GB, Rutherford TA, Webster JM (1985) Growth and virulence of Steinernema glaseri influenced by different subspecies of Xenorhabdus nematophilus. J Nematol 17:476–482
Ertan E, Seferoglu G (2003) The comparison of the biochemical characteristics of chestnut at fruit ripening and after traditional storage periods. Biosci Res Bull 19:139–149
Gaugler R, Wang Y, Campbell JF (1994) Aggressive and evasive behaviors in Popillia japonica (Coleoptera: Scarabaeidae) larvae: defenses against entomopathogenic nematode attack. J Invertebr Pathol 64:193–199
Gulcu B, Hazir S (2012) An alternative storage method for entomopathogenic nematodes. Turk J Zool 36:562–565
Han RC, Ehlers RU (2000) Pathogenicity, development, and reproduction of Heterorhabditis bacteriophora and Steinernema carpocapsae under axenic in vivo conditions. J Invertebr Pathol 75:55–58
Hazir S, Kaya HK, Stock SP, Keskin N (2003) Entomopathogenic nematodes (Steinernematidae and Heterorhabditidae) for biological control of soil pests. Turk J Biol 27:181–202
Karagoz, M., Gulcu, B. and Hazir, S. 2007. Manas larvalarının (Coleoptera: Scarabeidae) biyolojik mücadelesinde entomopatojenik nematodların test edilmesi. I. Entomopatojenler ve Mikrobiyal Mücadele Sempozyumu, 21–24 Haziran, Trabzon
Karagoz M, Gulcu B, Hazir S, Kaya HK (2009) Laboratory evaluation of Turkish entomopathogenic nematodes for suppression of the chestnut pests, Curculio elephas (Coleoptera: Curculionidae) and Cydia splendana (Lepidoptera: Tortricidae). Biocontrol Sci Techn 19:755–768
Karimi, J., Rezapanah, M., Monfared, F. and Mirsaeidi, H. 2010. Biological control potential of an entomopathogenic preparation of Heterorhabditis bacteriophora on the white grub Polyphylla adspersa. Society for Invertebrate Pathology meeting, 11–15 July, Trabzon Turkey
Kaya HK (1984) Nematode parasites of bark beetle. In: Nickle WR (ed) Plant and insect nematodes. Marcel Dekker, New York, pp 727–754
Kaya HK, Gaugler R (1993) Entomopathogenic nematodes. Annu Rev Entomol 38:181–206
Kaya HK, Stock SP (1997) Techniques in insect nematology. In: Lacey LA (ed) Manual of techniques in ınsect pathology. Academic, London, pp 281–324
Kepenekci I, Gokce A, Gaugler R (2004) Virulence of three species of entomopathogenic nematodes to the chestnut weevil, Curculio elephas (Coleoptera: Curculionidae). Nematropica 34:199–204
Kondo E (1989) Studies on the infectivity and propagation of entomogenous nematodes, Steinernema spp. (Rhabditida: Steinernematidae) in the common cutworm, Spodoptera litura (Lepidoptera: Noctuidae). Bull Fac Agric 67:1–87
Koppenhöfer AM, Grewal PS (2005) Compatibility and interactions with agrochemicals and other biological control agents. In: Grewal PS, Ehlers RU, Shapiro-Ilan DI (eds) Nematodes as biocontrol agents. CABI, Wallingford, pp 363–381
Koppenhöfer AM, Kaya HK (1997) Additive and synergistic interactions between entomopathogenic nematodes and Bacillus thuringiensis for scarab grub control. Biol Control 8:131–137
Koppenhöfer AM, Kaya HK, Shanmugan S, Wood GL (1995) Interspecific competition between steinernematid nematodes within an insect host. J Invertebr Pathol 66:99–103
Koppenhöfer AM, Choo HY, Kaya HK, Lee DW, Gelernter WD (1999) Increased field and greenhouse efficacy against scarab grubs with a combination of an entomopathogenic nematode and Bacillus thuringiensis. Biol Control 14:37–44
Koppenhöfer AM, Grewal PS, Kaya HK (2000) Synergism of imidacloprid and entomopathogenic nematodes against white grubs: the mechanism. Entomol Exp Appl 94:283–293
Sankar M, Prasad JS, Padmakumari AP, Katti G, Divya K (2009) Combined application of two entomopathogenic nematodes, Heterorhabditis indica and Steinernema asiaticum to control the rice leaf folder, Cnaphalocrosis medinalis (Goen.). J Biopestic 2:135–140
Shapiro-Ilan DI, Jackson M, Reilly CC, Hotchkiss MW (2004) Effects of combining an entomopathogenic fungi or bacterium with entomopathogenic nematodes on mortality of Curculio caryae (Coleoptera: Curculionidae). Biol Control 30:119–126
Shapiro-Ilan DI, Cottrell TE, Wood BW (2011) Effects of combining microbial and chemical insecticides on mortality of the pecan weevil (Coleoptera: Curculionidae). J Econ Entomol 104:14–20
Speranza S (1999) Chestnut pests in central Italy. Acta Hort 494:417–423
SPSS. 2004. SPSS v.13.0 for windows. SPSS, Chicago
Thurston GS, Kaya HK, Burlando TM, Harrison RE (1993) Milky disease bacterium as a stressor to increase susceptibility of scarabaeid larvae to an entomopathogenic nematode. J Invertebr Pathol 61:167–172
Thurston GS, Kaya HK, Gaugler R (1994) Characterizing the enhanced susceptibility of milky disease-infected scarabaeid grubs to entomopathogenic nematodes. Biol Control 4:67–73
Unlu I, Ehlers R-U, Susurluk A (2007) Additional data and first record of the entomopathogenic nematode Steinernema weiseri from Turkey. Nematology 9:739–741
Acknowledgments
We thank Dr. Albrecht M. Koppenhöfer, Rutgers University, New Brunswick, NJ, for comments on an earlier version of the manuscript and Adnan Menderes University for support of this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R.-U. Ehlers.
Rights and permissions
About this article
Cite this article
Demir, S., Karagoz, M., Hazir, S. et al. Evaluation of entomopathogenic nematodes and their combined application against Curculio elephas and Polyphylla fullo larvae. J Pest Sci 88, 163–170 (2015). https://doi.org/10.1007/s10340-014-0571-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-014-0571-9