Abstract
The incubation of eggs plays a key role in avian parental care. To ensure embryo development, incubating parents have to keep their eggs within the appropriate temperature limits. To do so, incubating individuals allocate substantial energy to the thermal demands of their eggs, but they face a trade-off with self-maintenance (own metabolism) because they usually cannot forage while incubating eggs. In species with female-only incubation, males can help their partners by providing them with food on the nest, a behavior which may enable females to spend more time incubating and could, consequently, lead to improved reproductive performance. In the study reported here, we first investigated whether incubation feeding by males affects nest attendance by females in Blue Tits (Cyanistes caeruleus) and subsequently determined how this incubation feeding affects reproductive performance. We found that the female nest attendance tended to increase with increasing amounts of food supplied by their male partner. Thus, males may enable females to incubate more when needed, as suggested by our observation that male incubation feeding was more frequent when the ambient temperature was lower, and especially so when females incubated later in the breeding season (during the study period the ambient temperature decreased rapidly over the breeding season, which is exceptional). Although female nest attendance did not result in a shorter time until the eggs hatched or in higher hatching success, females that attended the nest more produced heavier nestlings. We suggest that the trade-off between self-maintenance and meeting the demands of egg incubation likely tends to be less when females received more assistance from their partner during the egg incubation period, resulting in a higher investment in offspring.
Zusammenfassung
Der Effekt männlicher Partnerfütterung auf weibliche Nestanwesenheit und Reproduktionsleistung bei einem sozial monogamen Vogel Für Vögel ist die Bebrütung der Eier ein ausschlaggebender Faktor der elterlichen Sorge. Um die erfolgreiche Entwicklung der Embryonen zu gewährleisten, müssen die brütenden Eltern die Eier innerhalb kritischer Temperaturgrenzen halten. Erhebliche Energie und Zeit wird deshalb für die thermische Regulation des Geleges aufgewandt. Allerdings muss bei der Aufteilung der Energieressourcen ein Kompromiss zwischen dem Wohl der Nachkommen und der eigenen Selbsterhaltung gefunden werden. Weil es in der Regel nicht möglich ist, während der Bebrütung auszufliegen und auf Nahrungssuche zu gehen, ist die Aufrechterhaltung des eigenen Stoffwechsels kompromittiert. In Arten mit ausschließlich weiblicher Bebrütung können Männchen ihre Partnerinnen unterstützen, indem sie ihnen Nahrung im Nest zur Verfügung stellen (männliche Partnerfütterung). So können Weibchen mehr Zeit brütend im Nest verbringen, und dies könnte folglich zu einer verbesserten Reproduktionsleistung führen. In dieser Studie untersuchten wir, in wie weit männliche Partnerfütterung die weibliche Nestanwesenheit in Blaumeisen (Cyanistes caeruleus) beeinflusst und ob sich dies auf die Reproduktionsleistung auswirkt. Wir fanden, dass Weibchen tatsächlich dazu neigten, mehr zu brüten, wenn sie mehr von ihrem Partner gefüttert wurden. Männchen fütterten häufiger bei niedriger Umgebungstemperatur und insbesondere dann, wenn die Weibchen später begannen zu brüten (die Umgebungstemperatur nahm außergewöhnlicher Weise während der Brutzeit ab). So ermöglichten es die Männchen den Weibchen, mehr zu brüten, wenn dies besonders nötig erschien. Weibliche Nestanwesenheit führte weder zu einer verkürzten Zeit bis zum Schlüpfen, noch zu einem höheren Schlupferfolg. Jedoch produzierten Weibchen mit höherer Nestanwesenheit schwerere Nestlinge. Wir vermuten, dass der Kompromiss zwischen der Selbsterhaltung und den thermischen Ansprüchen der Eier wahrscheinlich erleichtert wird, wenn Weibchen während der Bebrütung mehr von ihrem Partner unterstützt werden, und darum mehr in die Nachkommen investieren können.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Incubation is a key factor of avian parental care and requires a substantial energetic investment by the incubating individuals (e.g. Camfield and Martin 2009; Bulla et al. 2015). To optimize embryo development, the parents have to ensure that the eggs are kept within appropriate temperature limits (Yom-Tov et al. 1978; Reid et al. 1999, 2000; Stein et al. 2010). Consequently, incubation is a crucial part of reproduction in almost all avian species (Matysioková and Remeš 2010); however, incubation is energetically costly, and incubating individuals also face a trade-off between self-maintenance and the thermal needs of the embryos. A common system that has been adopted by several species, possibly as a resolution to this trade-off, is a uniparental incubation system in which only the female incubates while the male provides aid in the form of supplying food (‘male incubation feeding’; Kluijver 1950; Hałupka 1994; Matysioková and Remeš 2010; Stein et al. 2010). The “female nutrition” hypothesis (Royama 1966) is one of the most invoked theories in this context and states that male incubation feeding enables the female to increase the amount of time she can spend incubating (female nest attendance; Royama 1966; Camfield and Martin 2009; Matysioková et al. 2011; Stein et al. 2010; Ibáñez-Álamo and Soler 2012). Hence, in order to understand avian reproductive strategies, it is important to understand the interplay between female nest attendance and male incubation feeding and the underlying factors that drive variation in these behaviours.
The frequency of male incubation feeding often differs between individuals (Royama 1966; Hatchwell et al. 1999; Matysioková et al. 2011; Stein et al. 2010). If male incubation feeding affects female nest attendance, this variation may be explained by variation in the circumstances that affect the need for incubation. For example, environmental factors, such as (seasonal changes in) ambient temperature (Webb 1987; Hatchwell et al. 1999; Camfield and Martin 2009; Matysioková and Remeš 2010) may affect the rate at which the eggs cool down and require females to incubate more. In addition, characteristics of the clutch, such as number of eggs, may be correlated with male incubation feeding: females with a larger clutch may require more energy to incubate the eggs or may spend more time on incubation and less on foraging (de Heij et al. 2007), so that males may need to feed females incubating larger clutches more frequently. Overall, females are expected to adjust nest attendance and males to adjust incubation feeding frequency in such a way that they shorten the incubation period (Hałupka 1994; Hatchwell et al. 1999; Camfield and Martin 2009; Stein et al. 2010), decrease developmental risks and/or maximize hatching and reproductive success (Lyon and Montgomerie 1985; Hatchwell et al. 1999; Martin and Ghalambor 1999; Tulp and Schekkerman 2006; Stein et al. 2010). Therefore, it can be expected that the variation in nest attendance and incubation feeding affects individuals’ reproduction depending on environmental circumstances and characteristics of the clutch.
In the study reported here, we investigated the effect of male incubation feeding on female nest attendance and pairs’ reproductive performance in Blue Tits (Cyanistes caeruleus), a passerine species with female-only incubation and male incubation feeding behaviour. Little is known on the proximate and ultimate components of male incubation feeding in Blue Tits (but see Nilsson and Smith 1988), which stresses the necessity of additional research. The aims of our study were (see also Fig. 1a): (1) to investigate how ambient temperature and life-history traits (i.e. the date of incubation onset and clutch size) relate to male incubation feeding and female nest attendance; (2) to determine whether male incubation feeding leads to higher female nest attendance; (3) to investigate the relation between female nest attendance and her reproductive performance, specifically the duration of the incubation period, hatching success and nestling body mass.
An overall scheme of our expectations (a) and results (b) regarding presumed relationships (i–iii) between environmental variables, life-history traits, incubation patterns and reproductive performance in Blue Tits (Cyanistes caeruleus). Direction of arrows indicate the correlation and effect, solid lines main results of the study, dashed lines the covariances (0 no effect, + positive effect, − negative effect)
Materials and methods
Study area and brood characteristics
This study was conducted during the breeding season (March–June) of 2014 in ‘De Vosbergen’ which is located in the northern part of The Netherlands (53º 08′N, 06º 35′E). The area consists of 54 ha of mixed deciduous and coniferous forest, woodlands and spaces of grassland. In total 209 nest boxes designed for Blue Tits were present in the study population in 2014.
The first nest box check was performed at the end of March, following which time occupied nest boxes with nesting material were regularly monitored to determine the date of first egg-laying, clutch size, date of incubation onset, duration of incubation period, hatching date and hatching success (proportion of hatched eggs). Nest boxes were not approached for the first 6 days after the start of egg-laying to minimize disturbance of the nest (note: Blue Tits usually lay >7 eggs). After day 7 (with day 1 being the day the first egg was laid), nest-checks were performed every day until incubation onset was observed. Incubation onset was either indicated by the female being present in the nest incubating the eggs or by the eggs being warm and uncovered during the nest-check. Once incubation onset was determined, nest-checks started on day 11 of incubation to check for hatching (hatching day 1 being the day the first egg hatched). Four days after hatching the nestlings were weighed using a Pesola spring balance. Nestling body mass was found not to be related to time of day when they were weighed (n = 59; P = 0.19), and therefore the original nestling body mass was used in the analyses.
Frequency of incubation feeding
The nest attendance and incubation feeding behaviour inside the nest boxes were recorded with infra-red cameras which were placed directly under the lid of the nest box. Dummy cameras were placed in the nest boxes before egg-laying to allow the birds to become accustomed to the presence of the camera. On day 5 after incubation onset, the dummy cameras were replaced by real cameras, and on day 6 the recording was started during a similar period to control the effect of recording time on ambient temperature. Our criterion for recording was around the middle of the incubation period (day 6, taken as a median representative of the incubation period), but not very early and not very late in the incubation period. The recording was continued for an average [± standard deviation (SD)] of 7.47 ± 1.25 h (n = 63 nest boxes).
The video recordings were analysed with a specially designed program ‘BirdBox’ which enabled faster and more efficient video analyses (for a detailed description of the procedures, see Amininasab et al. 2016). This program scans the events (e.g. male and female entry and behaviour) to distinguish these from other times of recording (no male or female inside the nest-box). We then reviewed all recorded events manually to check for female attendance, observe whether the male entered the nest-box with or without food and identify the prey type. Our analyses of the recordings indicated that males did enter the nest box when the female was also inside attending the eggs and that upon on next-box entry the male always carried food (see also Kluijver 1950 for more details). We did not use variation in prey quality in the analysis as in most cases it was not possible to reliably identify and distinguish the food type.
With these data, BirdBox generates a summary of the outputs, including (1) male incubation feeding (counts per hour) and (2) female nest attendance (minutes per hour). For male incubation feeding, we used similar data as reported in Amininasab et al. (2016), in which we investigated the combined effects of ambient temperature, habitat quality and individual age on incubation behaviour and incubation feeding. However, in the present study we used incubation feeding behaviour in order to link this behaviour directly with female nest attendance and other parameters of reproductive performance. From these data, male feeding per hour of female incubation was calculated as the number of feeds divided by the total time the female was incubating.
We also recorded the incubation behaviour of females in ten nest boxes using conventional cameras placed approximately 5 m in front of the nest boxes (~2 h per nest box) with the aim to record male incubation feeding around the nest box as opposed to inside the box (see Kluijver 1950). However, we did not record any events of male incubation feeding outside the nest box. For 15 nest boxes, we also followed the female further away with binoculars, but it was not possible to record whether males fed females far away from the nest box due to difficulties in the monitoring of birds in the dense vegetation (see Nilsson and Smith 1988; Pearse et al. 2004; Matysioková and Remeš 2010).
Ambient temperature
To estimate the ambient temperature during the incubation period, we used hourly measurements of the online weather archive of Eelde Airport, Groningen (1.6 km from our study area). The average temperature from the beginning until the end of the recording session was calculated per nest box.
Statistical analyses
The program R (version 3.1.0.; R Development Core Team 2014) was used for the statistical analysis. We tested for several correlations, as indicated in Fig. 1a and Table 1. To test for correlations between male incubation feeding (response variable) and ambient temperature, clutch size and date of onset of incubation (independent variables), we used a generalized linear model with Poisson distribution (offset = total female incubating time). To test whether male incubation feeding predicted female nest attendance, we used a linear model, with male incubation feeding frequency as an independent variable and female nest attendance as the response variable, including ambient temperature, clutch size and date of onset of incubation as independent variables. The relationship between female nest attendance and incubation period (total number of days of incubation—response variable) was analysed with a generalized linear model with Poisson distribution while also including the above-mentioned covariates. The correlation between female nest attendance and hatching success was tested with a generalized linear mixed model with a binominal distribution and logit link function (cbind) and the above-mentioned covariates. The frequency of male incubation feeding was not included as a direct predictor for duration of incubation and hatching success because incubation feeding could influence these variables through its effect on female nest attendance (Matysioková and Remeš 2010). Finally, the relationships between male incubation feeding and female nest attendance, female nest attendance and nestling body mass were analysed using linear models. The average nestling body mass per brood was used as the response variable. In all models, we started with the model that included all independent variables, then subsequently removed the least significant variable in each step to find the minimal adequate model (where all variables had a significant effect; P < 0.05). Due to the potential presence of collinearity between independent variables, we additionally tested for all singular correlations without other covariates, using Spearman rank correlations (see Table 1).
Ethical statement
All procedures were performed according to the animal experimentation standards of the University of Groningen (DEC number 6367). Handling times and visits to nest boxes were kept to a minimum in order to minimize disturbance to the birds.
Results
The average clutch size was 11.06 eggs (SD 1.71, range 8–15 eggs; n = 63), and the first and last date of onset of incubation was 16 April and 02 May, respectively. The average ambient temperature during the recordings throughout the breeding season was 15.07 °C (SD 2.18, range 9.57–19.30 °C) and unexpectedly declined with the date of incubation onset (r = −0.62, P < 0.001; Table 1). On average, males fed the females 1.84 times h−1 of incubation (SD 1.89, range 0–7.52 times h−1 of incubation; n = 63), and females incubated 43.93 min per hour (SD 3.76, range 35.13–51.36 min per hour; n = 63). Average hatching success (proportion hatched) was 0.88 (SD 0.16, range 0.33–1; n = 62; one brood was abandoned before hatching), and the average incubation period was 13.45 days (SD 1.30, range 11 – 18 days; n = 62). Average nestling body mass (4 days old) was 3.24 g per brood (SD 0.52, range 1.66–4.26 g per brood; n = 59; in 3 nest-boxes nestlings died before weighing).
An overall overview of the observed relationships between environmental variables, life-history traits, incubation patterns and reproductive performance is presented in Fig. 1b, and the results will be discussed in detail below.
Male incubation feeding in relation to incubation onset, ambient temperature and clutch size
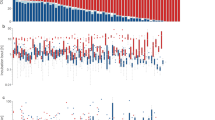
The frequency of male incubation feeding significantly increased throughout the breeding season (Table 2; Fig. 2). There was no significant effect of ambient temperature on male incubation feeding in the model with co-variates (Table 2), whereas ambient temperature was negatively correlated with male incubation feeding (Table 1). However, this might have been the effect of both ambient temperature and male incubation feeding being negatively correlated with date of incubation onset. It is therefore very difficult to separate the combined effects of ambient temperature and date of incubation onset on male incubation feeding. Clutch size was not associated with male incubation feeding frequency (Table 2).
Female nest attendance in relation to male incubation feeding
When all co-variates were included in the model, only ambient temperature negatively influenced female nest attendance (Table 3; Fig. 3b), with no effect on clutch size, date of incubation onset and frequency of male incubation feeding. However, in the model excluding the co-variates, a higher male incubation feeding frequency was associated with higher female nest attendance [marginally significant; estimate ± standard error (SE) 0.49 ± 0.24, t = 1.99, P = 0.051, n = 63; Fig. 3a]. Again, this difference may be the result of male incubation feeding rate being significantly correlated with ambient temperature.
Female nest attendance tends to a increase with increasing frequency of male incubation feeding per female attentive hour in Blue Tits [estimate ± standard error (SE) 0.49 ± 0.24, t = 1.99, P = 0.051, n = 63] and b decrease with increasing ambient temperature (see Table 2)
Reproductive performance in relation to female nest attendance
Female nest attendance was not related to the incubation period (estimate ± SE −0.006 ± −0.009, z = −0.71, P = 0.48) or hatching success (estimate ± SE 0.003 ± 0.05, t = 0.06, P = 0.95), and these results were similar when the co-variates were included in the model (Tables 4, 5). We found, however, a positive correlation between female nest attendance and average nestling body mass (Fig. 4).
Discussion
Our results suggest that the frequency of male incubation feeding increases throughout the breeding season and that males do not change the intensity of incubation feeding based on clutch size. Males also feed incubating females less when ambient temperatures are higher, i.e. circumstances in which female nest attendance decreases and females are likely to obtain sufficient food themselves. Female nest attendance did, however, not result in a shorter incubation period or higher hatching success, although females which exhibited higher nest attendance produced heavier nestlings. These results, their implications and potential complications are discussed in the following sections.
Male incubation feeding in relation to ambient temperature, incubation onset and clutch size
Our results show that male incubation feeding is significantly correlated with date of incubation onset, such that females which incubated later in the season were fed more often. However, the study period was exception in that the ambient temperature decreased over the breeding season whereas it normally increases. Therefore, our interpretation and separation of the effect of ambient temperature or/and date of incubation onset on the frequency of male incubation feeding are complicated, and a combination of the following alternatives should be considered:
-
1.
Earlier in the breeding season there may be more fertile females in the population (e.g. Vedder et al. 2012) so that males may spend more time visiting potential extra-pair partners instead of spending time feeding their incubating female (Nisbet 1973; Tobias and Seddon 2002; Pearse et al. 2004; Tryjanowski and Hromada 2005).
-
2.
Male incubation feeding may also increase over the season because females which incubate earlier in the season may have access to sufficient food in the territory, thereby reducing the need of male feeding (Perrins 1970; Martin 1987). Indeed, if food availability is predicted by temperature, males may use ambient temperature as a cue to estimate the female’s need of food, which could explain our result that males feed incubating females more when ambient temperatures are lower.
-
3.
Food availability may actually be higher later in the season, which may also explain why males can feed their female partner later.
-
4.
Later incubating females may also be fed more as an adaptive mechanism to hatch their brood sooner due to more nest attendance, with the aim to ensure hatching coincides with the food peak (Martin 1987; Visser et al. 2006). Studies on Pied Flycatchers (Ficedula hypoleuca) (Lifjeld et al. 1987; Smith et al. 1989) and Bewick’s Wrens (Thryomanes bewickii) (Pearse et al. 2004) obtained similar outcomes.
Therefore, taken altogether, we suggest that male incubation feeding may increase over the season depending on social and/or environmental circumstances, although with our single-year results and the unexpected ambient temperature profile over the breeding season it is difficult to separate the effect of ambient temperature from the date of incubation onset on male incubation feeding. Therefore, future studies are needed to disentangle the effects of these factors on male incubation feeding in years in which the incubation onset is positively correlated with ambient temperature.
We expected a higher incubation cost, more female nest attendance and higher frequency of incubation feeding in larger clutches (Martin and Wiebe 2000). However, in our study male incubation feeding was not associated with clutch size, similar to observations on the Great Tits (Parus major; Matysioková and Remeš 2010). In another experimental study on the Great Tits, clutch size enlargement had no effect on female energy expenditure (de Heij et al. 2008). However, the absence of an observed effect of clutch size on male incubation feeding may be related to aspects of female behaviour or physiology (e.g. females in better condition laying larger clutches), a possibility which requires more empirical study.
In addition to the above-mentioned parameters, other potential factors (e.g. the quality of prey, the quality of males) may have an influence on the frequency of male incubation feeding. This possibility makes it difficult to separate the effect of different parameters on male incubation feeding and to distinguish between the actual mechanisms. Therefore, further studies which take these potential factors into account are needed to separate the observed effects on male incubation feeding from food- and male-quality effects.
Female nest attendance in relation to male incubation feeding
The results of our study suggest that male incubation feeding likely enabled the female to spend more time incubating. However, support for this suggestion comes from the model that excluded other covariates and is based on a result which only borders statistical significance (P = 0.051). When the ambient temperature was included in the model, the date of incubation onset and clutch size led to different results—namely, not male incubation feeding frequency, but ambient temperature was associated with the time females spent incubating. This latter association would imply that ambient temperature influences female metabolism and the need to search for food. While a male may determine the hunger state of an incubating female by ambient temperature per se, males and females have been found to communicate using specified calls that may trigger males to search for additional food to support the incubating female (see Kluijver 1950 for details).
The different results of our analyses due to exclusion and inclusion of the covariates may be due to the underlying correlations of ambient temperature and the date of incubation onset with male incubation feeding; these potential correlations make it impossible to separate the relationships. Hałupka (1994) also found a similar pattern in Meadow Pipits (Anthus pratensis) with male incubation feeding frequency and female nest attendance both co-varying with ambient temperature. These results suggest that males likely enable females to spend more time incubating, especially when eggs need to be incubated longer, whereas males do not need to feed females under circumstances when there is sufficient food. As such, the results of our study provide partial support to the female nutrition hypothesis (Arcese and Smith 1988; Moreno 1989; Smith et al. 1989; Hatchwell et al. 1999; Klatt et al. 2008; but see Jawor and Breitwisch 2006; Matysioková and Remeš 2010; Boulton et al. 2010).
However, with respect to this hypothesis, the interpretation our data requires care due to the bordering statistical significance and statistical co-variance between male incubation feeding and ambient temperature. Male incubation feeding may favour the female mental state independently of energetic state. For example, assisted females may pay more attention to nest maintenance, thereby influencing nest insulation capacities. Furthermore, other parameters may also affect female nest attendance, such as nest design which also influences insulating capacity during incubation (Deeming 2016; Cruz et al. 2016). However, one single-day measure may not reflect breeding behaviour (male incubation feeding and female nest attendance) during a 1-month period. To better analyse these factors, future studies are needed in which behaviours are monitored during the entire incubation period for each pair in order to clarify the causal effect and the mechanism with greater accuracy.
Reproductive performance in relation to female nest attendance
We predicted that an increase in female nest attendance associated with male feedings would shorten the overall incubation period and would increase hatching success due to the reduction of developmental risks. Our results do not support these predictions. Our findings are similar to those reported by Hatchwell et al. (1999) and Pearse et al. (2004) who found no significant link between female nest attendance and incubation period or/and hatching success (e.g. Matysioková and Remeš 2010; Stein et al. 2010). However, several other studies have documented that food supplementation can reduce incubation period or/and improve hatching success (Lyon and Montgomerie 1985; Nilsson and Smith 1988). The inconsistency between the results of these earlier studies and our study remains unexplained, but several potential reasons were put forward by Matysioková and Remeš (2010). First, female nest attendance and subsequent hatching success may be related to unmeasured differences in the quality of food brought by the male to the next or to females using food to provide energy for self-maintenance rather than for incubation. These explanations seem unlikely to explain the results in our study, as females that were fed more tended to incubate longer. Second, and a more likely explanation, hatching success may also be affected by factors other than incubation, such as inbreeding (Hemmings et al. 2012; Kingma et al. 2013). Third, we could not record potential incubation feeding further away from the nest box due to the dense vegetation around the nest and monitoring limitations (see Nilsson and Smith 1988; Pearse et al. 2004; Matysioková and Remeš 2010). However, incubation feeding far away from the nest may be an important factor determining a female’s decision and subsequently may have an influence on incubation period length and hatching success. These potential explanations for the lack of correlation between female nest attendance and incubation period or hatching success need to be considered in future studies aimed at assessing the potential evolutionary forces driving female nest attendance and male incubation feeding.
Although we did not find an effect on hatching success and incubation duration, a higher female nest attendance resulted in higher average nestling body mass. The immediate link between these variables is not immediately clear, and it has to be noted that between incubation and hatching there may be other factors (e.g. parental body quality, parental feeding rates, ambient temperature and egg mass) that influence the growth of the nestlings. Similarly, females that have reduced costs of incubation may save energy, which may enable them to invest more energy in raising the offspring. A somewhat similar explanation is that females who leave the nest less often and are better foragers (e.g. more efficient, higher body quality or in better quality territories) may be able to also feed their brood at a higher rate.
Generally, it is difficult to demonstrate a causal effect of male incubation feeding on female nest attendance and other parameters of reproductive performance. Experimental studies should be conducted to separate the effects and avoid the underlying correlations of explanatory variables. For example, one should experimentally provide additional food to the female, while manipulating the ambient temperature to demonstrate a causal effect between male incubation feeding frequency, female nest attendance and outcomes of reproductive performance. Such an approach may reveal further insights into the patterns of avian incubation behaviour and reproductive performance.
Conclusions
The results of our study show that females which incubate later in the breeding season (exceptionally lower ambient temperature) are likely to be fed more by their partners than females which start incubating earlier, which is likely the effect of female nest attendance being higher in lower ambient temperature. However, due to our study period being unusual in that the temperature decreased—rather than increased—over the breeding season, it is difficult to separate the effect of ambient temperature from the date of incubation onset. We suggest that the frequency of male incubation feeding probably increases female nest attendance. Finally, we found that female nest attendance was not correlated with the duration of the incubation period and hatching success, although it may result in the production of heavier offspring. Generally, our results suggest that male incubation rate is somewhat adjusted to the needs of the incubating female, rather than facilitating an overall improvement of reproduction.
References
Amininasab SM, Kingma SA, Birker M, Hildenbrandt H, Komdeur J (2016) The effect of ambient temperature, habitat quality and individual age on incubation behaviour and incubation feeding in a socially monogamous songbird. Behav Ecol Sociobiol 70:1591–1600
Arcese P, Smith JNM (1988) Effects of population density and supplemental food on reproduction in song sparrows. J Anim Ecol 57:119–136
Boulton RL, Richard Y, Armstrong DP (2010) The effect of male incubation feeding, food and temperature on the incubation behaviour of New Zealand Robins. Ethology 116:490–497
Bulla M, Cresswell W, Rutten AL, Valcu M, Kempenaers B (2015) Biparental incubation-scheduling: no experimental evidence for major energetic constraints. Behav Ecol 26:30–37
Camfield AF, Martin K (2009) The influence of ambient temperature on horned lark incubation behaviour in an Alpine environment. Behaviour 146:1615–1633
Cruz A, Alvarez E, Barba E (2016) Nest insulating capacity during incubation and after fledging are related. Avian Biol Res 9:22–27
De Heij ME, van der Graaf AJ, Hafner D, Tinbergen JM (2007) Metabolic rate of nocturnal incubation in female Great Tits (Parus major) in relation to clutch size measured in a natural environment. J Exp Biol 210:2006–2012
De Heij ME, Ubels R, Visser GH, Tinbergen JM (2008) Female great tits (Parus major) do not increase their daily energy expenditure when incubating enlarged clutches. J Avian Biol 39:121–126
Deeming DC (2016) Perspectives on nest construction and function. Avian Biol Res 5:191–192
Hałupka K (1994) Incubation feeding in meadow pipit (Anthus pratensis) affects female time budget. J Avian Biol 25:251–253
Hatchwell BJ, Fowlie MK, Ross DJ, Russell AF (1999) Incubation behaviour of Long-tailed Tits: why do males provision incubating females? Condor 101:681–686
Hemmings NL, Slate J, Birkhead TR (2012) Inbreeding causes early death in a passerine bird. Nat Commun 3:863
Ibáñez-Álamo JD, Soler M (2012) Predator-induced female behaviour in the absence of male incubation feeding: an experimental study. Behav Ecol Sociobiol 66:1067–1073
Jawor JM, Breitwisch R (2006) Is mate provisioning predicted by ornamentation? A test with Northern cardinals (Cardinalis cardinalis). Ethology 112:888–895
Kingma SA, Hall ML, Peters A (2013) Breeding synchronization facilitates extrapair mating for inbreeding avoidance. Behav Ecol 24:1390–1397
Klatt PH, Stutchbury BJM, Evans ML (2008) Incubation feeding by male scarlet tanagers: a mate removal experiment. J Field Ornithol 79:1–10
Kluijver HN (1950) Daily routines of the Great Tit, Parus m. major L. Ardea 38:99–135
Lifjeld JT, Slagsvold T, Stenmark G (1987) Allocation of incubation feeding in a polygynous mating system: a study on Pied Flycatchers (Ficedula hypoleuca). Anim Behav 35:1663–1669
Lyon BE, Montgomerie RD (1985) Incubation feeding in snow buntings: female manipulation or indirect male parental care? Behav Ecol Sociobiol 17:279–284
Martin TE (1987) Food as a limit on breeding birds: a life-history perspective. Annu Rev Ecol Evol Syst 18:453–487
Martin TE, Ghalambor CK (1999) Males feeding females during incubation. I. Required by microclimate or constrained by nest predation? Am Nat 153:131–139
Martin K, Wiebe KL (2000) The use of incubation behaviour to adjust avian reproductive costs after egg laying. Behav Ecol Sociobiol 48:463–470
Matysioková B, Remeš V (2010) Incubation feeding and nest attentiveness in a socially monogamous songbird: role of feather colouration, territory quality and ambient environment. Ethology 116:596–607
Matysioková B, Cockburn A, Remeš V (2011) Male incubation feeding in songbirds responds differently to nest predation risk across hemispheres. Anim Behav 82:1347–1356
Moreno J (1989) Energetic constraints on uniparental incubation in the Northern wheatear (Oenanthe oenanthe L.). Ardea 77:107–115
Nilsson JA, Smith HG (1988) Incubation feeding as a tactic for early hatching. Anim Behav 36:641–647
Nisbet ICT (1973) Courtship-feeding, egg size and breeding success in common terns (Sterna hirundo). Nature 241:141–142
Pearse AT, Cavitt JF, Cully JF (2004) Effects of food supplementation on female nest attentiveness and incubation mate feeding in two sympatric wren species. Wilson Bull 16:23–30
Perrins CM (1970) The timing of birds’ breeding seasons. Ibis 112:242–255
R Development Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reid JM, Monaghan P, Ruxton GD (1999) The effect of clutch cooling rate on starling (Sturnus Vulgaris) incubation strategy. Anim Behav 58:1161–1167
Reid JM, Monaghan P, Ruxton GD (2000) The consequences of clutch size for incubation conditions and hatching success in starlings. Funct Ecol 14:560–565
Royama T (1966) A re-interpretation of courtship feeding. Bird Study 13:116–129
Smith HG, Kiillander H, Hultman J, Sanzén B (1989) Female nutritional state affects the rate of male incubation feeding in the pied flycatcher (Ficedula hypoleuca). Behav Ecol Sociobiol 24:417–420
Stein LR, Oh KP, Badyaev AV (2010) Fitness consequences of male provisioning of incubating females in a desert passerine bird. J Ornithol 151:227–233
Tobias JA, Seddon N (2002) Female begging in European robins: do neighbors eavesdrop for extra pair copulations? Behav Ecol 13:637–642
Tryjanowski P, Hromada M (2005) Do males of the great gray shrike (Lanius excubitor) trade food for extra pair copulations? Anim Behav 69:529–533
Tulp I, Schekkerman H (2006) Time allocation between feeding and incubation in uniparental Arctic-breeding shorebirds: energy reserves provide leeway in a tight schedule. J Avian Biol 3:207–218
Vedder O, Magrath MJL, Niehoff DL, van der Velde M, Komdeur J (2012) Declining extra-pair paternity with laying order associated with initial incubation behavior, but independent of final clutch size in the Blue Tit. Behav Ecol Sociobiol 66:603–612
Visser ME, Holleman LJM, Gienapp P (2006) Shifts in caterpillar biomass phenology due to climate change and its impact on the breeding biology of an insectivorous bird. Oecologia 147:164–172
Webb DR (1987) Thermal tolerance of avian embryos: a review. The Condor 89(June):874–898
Yom-Tov Y, Ar A, Mendelssohn H (1978) Incubation behaviour of the dead sparrow. Condor 80:340–343
Acknowledgements
We thank the Kraus-Groeneveld Stichting for permission to work on the De Vosbergen estate. We would like to thank the team of field-workers for their indispensable help in the field. We thank Peter Korsten for valuable general advice during the project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The research was financially supported by a Grant from The Netherlands Organisation for Scientific Research (NWO-ALW 821.01.008) and other grants funded to JK.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Communicated by F. Bairlein.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Amininasab, S.M., Birker, M., Kingma, S.A. et al. The effect of male incubation feeding on female nest attendance and reproductive performance in a socially monogamous bird. J Ornithol 158, 687–696 (2017). https://doi.org/10.1007/s10336-016-1427-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-016-1427-2