Abstract
A significant increase in carbapenemase-producing Klebsiella pneumoniae (CP-Kp) bacteraemias has been observed worldwide. The objective of the present work was to study the risk factors and predictors of mortality of CP-Kp bacteraemias among critically ill patients. During a 4-year period (2012–3015), a matched 1:2 case-control study was conducted. Klebsiella pneumoniae was identified by Vitek 2 technology. Antibiotic susceptibility was performed by the agar disc diffusion method and Etest. The presence of the bla KPC, bla VIM and bla NDM genes was confirmed by polymerase chain reaction (PCR). Epidemiologic data were collected from the intensive care unit (ICU) computerised database. One hundred and thirty-nine patients who developed a CP-Kp bacteraemia were matched with 278 patients. The majority of isolates (128; 92.1%) carried the bla KPC gene, seven carried both bla KPC and bla VIM, three bla VIM and one carried bla NDM. Risk factors for the development of CP-Kp bacteraemia were administration of tigecycline and number of antibiotics administered prior to CP-Kp bacteraemia. Overall, the 30-day mortality was 36.0%. Multivariate analysis revealed septic shock, Simplified Acute Physiology Score II (SAPS II) upon infection onset, adjunctive corticosteroid administration and parenteral nutrition as independent predictors of mortality, while treatment with a combination of appropriate antibiotics was identified as a predictor of good prognosis. Among septic shock patients (n = 74), Sequential Organ Failure Assessment (SOFA) score upon infection onset, adjunctive corticosteroid administration and strain carrying the bla KPC gene were independently associated with mortality, while the administration of combination treatment was identified as a predictor of a good prognosis. The administration of tigecycline predisposes to the induction of bacteraemia. Appropriate antibiotic treatment is associated with better survival, while concomitant corticosteroid treatment is associated with mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Carbapenemase-producing Klebsiella pneumoniae (CP-Kp) constitute a significant public health issue reaching worldwide proportions. During the previous decade, CP-Kp infections reached a high level of endemicity in Greece, while only sporadic occurrence or single hospital outbreaks were reported in other European countries [1, 2]. Since 2010, CP-Kp isolates disseminated to almost all European countries. In 2015, three more countries (Italy, Malta and Turkey) became CP-Kp endemic, while regional and interregional spread is reported from most countries [1, 2].
CP-Kp infections are associated with high morbidity and mortality due to limited therapeutic options, which include only gentamicin, colistin, tigecycline and carbapenems [2–5]. As shown in observational studies, combination treatment has been associated with increased survival, while the optimal regimen has not been identified, probably due to the emergence and increase of isolates’ resistance to the aforementioned therapeutic options [2–5]. Few studies have compared the impact of different carbapenemase type to mortality, with KPC (K. pneumoniae carbapenemase) being associated with reduced survival [6].
The aim of this study was to identify the risk factors for the development of CP-Kp bloodstream infections (BSI) among critically ill patients. We also investigated predictors of mortality in patients with CP-Kp bacteraemia and in the subgroup who developed septic shock.
Materials and methods
This 1:2 case–control study was performed in the general intensive care unit (ICU; 13 beds) of the University General Hospital of Patras, Greece, during a 4-year period (2012–2015).
Cases were considered patients with a CP-Kp BSI, defined as at least one positive blood culture for CP-Kp and clinical symptoms consistent with bacteraemia. Primary or secondary bacteraemia (urinary, respiratory, catheter-related, abdominal, skin and soft tissue infections) was determined in accordance with the US Centers for Disease Control and Prevention (CDC) definitions [7]. Infection was categorised as sepsis or septic shock according to new sepsis definition [8]. The date of collection of the first positive blood culture was defined as infection onset. Appropriate antibiotic treatment was defined as one that included an antibacterial agent with in vitro activity against the infecting isolate, initiated within 72 h from the onset of infection, at an adequate dosage [9, 10]. Combination therapy was defined as the administration of at least two antibacterial agents with in vitro activity against the infecting CP-Kp isolate.
Controls were patients without CP-Kp infection. Three patients that developed infection by carbapenem-susceptible K. pneumoniae were not included in the analysis. Matching parameters for controls included Acute Physiology and Chronic Health Evaluation II (APACHE II) score (±3 points), age (±2 years), as well as days at risk (±4 days; time from admission to BSI for cases and length of ICU stay for controls).
The ICU’s computerised database (Criticus™, University of Patras, Greece) and patients’ chart reviews were used in order to collect epidemiologic data. Parameters assessed included demographic characteristics (age, sex), co-morbidities, severity scores of illness on admission and upon onset of infection [APACHE II, SAPS II (Simplified Acute Physiology Score II) and SOFA (Sequential Organ Failure Assessment) score], prior surgery, length of hospitalisation, presence of invasive catheters, tracheotomy, prior BSI, type of antibiotic administration, corticosteroid administration and enteral or parenteral nutrition.
In the event of appearance of fever (≥38.0°C) or clinical presentation suggestive of BSI, two blood sets from peripheral sites and one from the central venous line, as well as cultures from the suspected source (bronchial secretions, urine, pleural or peritoneal fluid, pus, catheter tip etc.) were obtained and sent to the microbiology laboratory. Klebsiella pneumoniae isolates were identified using the Vitek 2 Advanced Expert System (bioMérieux, Marcy l’Etoile, France) and antibiotic susceptibility was performed by the agar disc diffusion method (Kirby–Bauer). Susceptibility to carbapenems, colistin and tigecycline was determined by the Etest (AB Biodisk). The results were interpreted according to the European Committee on Antimicrobial Susceptibility Testing (EUCAST) guidelines [11]. The presence of the bla KPC, bla VIM and bla NDM genes was confirmed by polymerase chain reaction (PCR) [12].
Data analyses were performed by SPSS version 23.0 (SPSS, Chicago, IL) software. Categorical variables were analysed by using Fisher’s exact test and continuous variables with the Mann–Whitney U-test. Backward stepwise multiple logistic regression analysis used all those variables from the univariate analysis with a p-value < 0.1. Factors contributing to multicollinearity were excluded from the multivariate analysis. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated to evaluate the strength of any association. All statistical tests were two-tailed and p < 0.05 was considered statistically significant.
Results
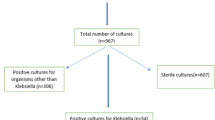
Among the 1390 patients admitted to the ICU during the study period, 139 (10.0%) developed a CP-Kp bacteraemia within an average of 17.5 days, and were matched to 278 non-bacteraemic patients. The incidence was 10 infections per 1000 patient days. All isolates were resistant to carbapenems [all exhibiting minimum inhibitory concentration (MIC) >32 mg/L], while 84 (60.4%), 62 (44.6%) and 39 (28.1%) were resistant to gentamicin, colistin and tigecycline, respectively. The majority of isolates (128; 92.1%) carried the bla KPC gene, seven carried both bla KPC and bla VIM, three bla VIM and one carried bla NDM. Primary BSI predominated (88; 63.3%), followed by 46 (33.1%) catheter-related, four abdominal (2.9%) and one ventilator-associated pneumonia (0.7%).
Univariate analysis for CP-Kp BSI risk factors is shown in Table 1. Risk factors independently associated with the development of CP-Kp were administration of tigecycline (p 0.010; OR 2.6, 95% CI 1.3–5.5) and number of administered antibiotics prior to CP-Kp bacteraemia (p 0.004; OR 1.3, 95% CI 1.1–1.5).
The overall 30-day mortality was 36.0% (50 patients). Table 2 shows the univariate analysis of predictors of mortality of CP-Kp BSI. Multivariate analysis revealed septic shock (p 0.001; OR 6.5, 95% CI 2.2–19.5), SAPS II upon onset of infection (p <0.001; OR 1.1, 95% CI 1.0–1.2), corticoid administration during treatment of bacteraemia (p 0.027; OR 3.1, 95% CI 1.1–8.6) and parenteral nutrition (p 0.048; OR 2.8, 95% CI 1.0–7.7) as independent predictors of mortality, while combination antibiotic treatment (p 0.014; OR 0.237, 95% CI 0.075–0.750) was identified as a predictor of good prognosis. Mortality among the 74 patients with septic shock was 58.1%. Moreover, multivariate analysis identified SOFA score upon onset of infection (p 0.023; OR 1.4, 95% CI 1.0–1.8), corticosteroid administration as adjunctive treatment (p 0.025; OR 6.0, 95% CI 1.3–28.3) and strain carrying only the bla KPC gene as compared to other types of carbapenemases (p 0.041; OR 22.6, 95% CI 1.1–448.4) as predictors of mortality among patients with CP-Kp, while administration of a combination treatment (p 0.029; OR 0.430, 95% CI 0.201–0.918) was identified as a predictor of a good prognosis.
Figure 1 shows the Kaplan–Meier curves of the survival probability of patients with CP-Kp BSI according to administration of corticosteroid and number of active antibiotics among patients with septic shock. Even though the administration of cortisone did not alter survival among patients that received one active or no active antibiotic (35.3% vs. 55.6%; p 0.800), its administration among patients receiving two or more active antibiotics had a deleterious effect on survival (21.4% vs. 71.4%; p 0.034). No difference among age, comorbidities, severity scores upon admission or upon onset of infection was detected among patients with septic shock who received or did not receive corticosteroids.
Kaplan–Meier curves of survival probability of patients with carbapenemase-producing Klebsiella pneumoniae (CP-Kp) bloodstream infection (BSI) according to administration of corticosteroids among patients with septic shock who received (a) one active or no active antibiotic or (b) a combination of active antibiotics
Discussion
We observe a high incidence of CP-Kp BSI (10 infections per 1000 patient days), with 10% of all patients admitted into the ICU during the study period being infected by a CP-Kp isolate, confirming the endemicity of such isolates in Greek hospitals. This high incidence can be explained by the high rectal colonisation observed in a previous study from the same ICU [3].
In accordance to previous studies, the present investigation confirms that the total number of administered antibiotics is associated with CP-Kp infection [13, 14]. Moreover, it is the first study which identifies the use of tigecycline as a risk factor for CP-Kp BSI development. This is peculiar, since tigecycline is usually used extensively to treat such infections. A possible explanation may be that almost one-third of isolates were resistant to tigecycline.
In contrast to a previous Italian multicentre study which showed that prior BSI by other pathogens was associated with the development of CP-Kp, no such association was found in the present study [15]. There are two main differences among the present and the aforementioned study; first, non-fermenting Gram-negatives, mainly Acinetobacter baumannii and Pseudomonas aeruginosa, predominated in the present study and second, in the Italian study, only patients colonised by CP-Kp were included, while in the present study, no rectal cultures were obtained during the study period in order to determine the status of colonisation [15].
The all-cause 30-day mortality was 36.0%, which is comparable to other studies [3, 5, 6, 16]. As previously shown, SAPS II or SOFA score upon CP-Kp infection onset were associated with 30-day mortality [3, 17]. In our study, SAPS II was independently associated with mortality of all CP-Kp infections; the SOFA score was better in predicting mortality in the subgroup of patients with septic shock.
The development of septic shock is another factor independently associated with mortality [3]. The new definitions proposed by the Third International Consensus were used to define septic shock [8]. Septic shock mortality was 58.1%, which was higher than reported in a similar study (39.6%) conducted at a university hospital in Rome, Italy [4]. In that study, an important predictor of mortality was the resistance of infecting isolates to colistin (51.3%) [4]. In our study, even though resistance to colistin (44.6%) was comparable to that of the aforementioned study, no difference among surviving and deceased patients concerning resistance to any antibiotic (colistin, tigecycline or gentamicin) was observed.
CP-Kp isolates carrying the bla KPC gene have mainly disseminated into Greek hospitals, while other types of carbapenemase-producing strains have been isolated to a lesser degree [2, 6, 12, 14]. The type of carbapenemase gene carriage by the infecting isolate did not have any effect on mortality. However, in the subgroup of septic shock, the carriage of only bla KPC was associated with reduced survival, as compared to the carriage of bla NDM or co-carriage of bla KPC and bla VIM. This is probably due to the higher rates of resistance against the three principal antibiotics (colistin, tigecycline and gentamicin) used for the treatment of such infections [12]. KPC-producing isolates, as compared to isolates carrying other carbapenemases, have been rarely shown to be associated with higher mortality [6, 16]. A need for multicentre studies, including different types of carbapenemases, is needed in order to extrapolate reliable conclusions concerning the propensity of KPC-producing isolates to provoke higher mortality rates.
As consistently shown in observational studies, combination antibiotic treatment is associated with better survival as compared to monotherapy [2–5, 16]. No combination of antibiotics has shown clear superiority. Only 27.3% received a combination of active antibiotics, which is lower than that from previous studies. This is mainly due to the high resistance of all isolates to carbapenems (MIC to meropenem >32 mg/L), excluding in that way a potent antibiotic category that shows clear superiority when used as combination therapy [2, 16]. Moreover, infecting isolates showed high resistance rates against gentamicin, colistin and tigecycline, rendering the use of a combination treatment not always feasible.
As proposed by the Surviving Sepsis Campaign guidelines, corticosteroid should be added in the treatment of septic shock during the duration of inotropic administration [17]. An alarming finding of the present study is that the use of corticosteroids as adjunctive therapy in infections caused by CP-Kp has deleterious effects. This is clearly shown in the subgroup of patients with septic shock. Corticosteroid use among septic patients who received one active or no active antibiotic did not influence mortality, while their use in patients who received a combination of active antibiotics exhibited a catastrophic effect (21.4% vs. 71.4%; p 0.034). The effect of adjunctive corticosteroids in the treatment of septic shock due to CP-Kp should be validated through randomised controlled trials.
The study has several limitations, including its retrospective form and the relatively small number of CP-Kp. The predominance of KPC-producing isolates limits the importance of our finding that these isolates are associated with higher mortality rate among patients who developed septic shock.
In conclusion, the number of antibiotics administered and the prior administration of tigecycline is associated with CP-Kp BSI among critically ill patients. Either severity score (SAPS II or SOFA) upon infection onset can be used for mortality prediction. The development of septic shock is associated with higher mortality and the concomitant use of corticosteroids may have a deleterious effect on the survival of patients with CP-Kp bacteraemia. The use of a proper combination antibiotic regimen, when feasible, is associated with better survival.
References
European Centre for Disease Prevention and Control (ECDC) (2016) Rapid risk assessment: carbapenem-resistant Enterobacteriaceae. ECDC, Stockholm
Tzouvelekis LS, Markogiannakis A, Psichogiou M, Tassios PT, Daikos GL (2012) Carbapenemases in Klebsiella pneumoniae and other Enterobacteriaceae: an evolving crisis of global dimensions. Clin Microbiol Rev 25:682–707
Papadimitriou-Olivgeris M, Marangos M, Christofidou M, Fligou F, Bartzavali C, Panteli ES, Vamvakopoulou S, Filos KS, Anastassiou ED (2014) Risk factors for infection and predictors of mortality among patients with KPC-producing Klebsiella pneumoniae bloodstream infections in the intensive care unit. Scand J Infect Dis 46:642–648
Falcone M, Russo A, Iacovelli A, Restuccia G, Ceccarelli G, Giordano A, Farcomeni A, Morelli A, Venditti M (2016) Predictors of outcome in ICU patients with septic shock caused by Klebsiella pneumoniae carbapenemase-producing K. pneumoniae. Clin Microbiol Infect 22:444–450
Tumbarello M, Trecarichi EM, De Rosa FG, Giannella M, Giacobbe DR, Bassetti M, Losito AR, Bartoletti M, Del Bono V, Corcione S, Maiuro G, Tedeschi S, Celani L, Cardellino CS, Spanu T, Marchese A, Ambretti S, Cauda R, Viscoli C, Viale P; ISGRI-SITA (Italian Study Group on Resistant Infections of the Società Italiana Terapia Antinfettiva) (2015) Infections caused by KPC-producing Klebsiella pneumoniae: differences in therapy and mortality in a multicentre study. J Antimicrob Chemother 70:2133–2143
Mouloudi E, Protonotariou E, Zagorianou A, Iosifidis E, Karapanagiotou A, Giasnetsova T, Tsioka A, Roilides E, Sofianou D, Gritsi-Gerogianni N (2010) Bloodstream infections caused by metallo-beta-lactamase/Klebsiella pneumoniae carbapenemase-producing K. pneumoniae among intensive care unit patients in Greece: risk factors for infection and impact of type of resistance on outcomes. Infect Control Hosp Epidemiol 31:1250–1256
Horan TC, Andrus M, Dudeck MA (2008) CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 36:309–332
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche JD, Coopersmith CM, Hotchkiss RS, Levy MM, Marshall JC, Martin GS, Opal SM, Rubenfeld GD, van der Poll T, Vincent JL, Angus DC (2016) The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 315:801–810
Plachouras D, Karvanen M, Friberg LE, Papadomichelakis E, Antoniadou A, Tsangaris I, Karaiskos I, Poulakou G, Kontopidou F, Armaganidis A, Cars O, Giamarellou H (2009) Population pharmacokinetic analysis of colistin methanesulfonate and colistin after intravenous administration in critically ill patients with infections caused by gram-negative bacteria. Antimicrob Agents Chemother 53:3430–3436
De Pascale G, Montini L, Pennisi M, Bernini V, Maviglia R, Bello G, Spanu T, Tumbarello M, Antonelli M (2014) High dose tigecycline in critically ill patients with severe infections due to multidrug-resistant bacteria. Crit Care 18:R90
The European Committee on Antimicrobial Susceptibility Testing (EUCAST) (2016) Breakpoint tables for interpretation of MICs and zone diameters. Version 6.0. Home page at: http://www.eucast.org
Spyropoulou A, Papadimitriou-Olivgeris M, Bartzavali C, Vamvakopoulou S, Marangos M, Spiliopoulou I, Anastassiou ED, Christofidou M (2016) A ten-year surveillance study of carbapenemase-producing Klebsiella pneumoniae in a tertiary care Greek university hospital: predominance of KPC- over VIM- or NDM-producing isolates. J Med Microbiol 65:240–246
Borer A, Saidel-Odes L, Eskira S, Nativ R, Riesenberg K, Livshiz-Riven I, Schlaeffer F, Sherf M, Peled N (2012) Risk factors for developing clinical infection with carbapenem-resistant Klebsiella pneumoniae in hospital patients initially only colonized with carbapenem-resistant K pneumoniae. Am J Infect Control 40:421–425
Daikos GL, Vryonis E, Psichogiou M, Tzouvelekis LS, Liatis S, Petrikkos P, Kosmidis C, Tassios PT, Bamias G, Skoutelis A (2010) Risk factors for bloodstream infection with Klebsiella pneumoniae producing VIM-1 metallo-beta-lactamase. J Antimicrob Chemother 65:784–788
Giacobbe DR, Del Bono V, Bruzzi P, Corcione S, Giannella M, Marchese A, Magnasco L, Maraolo AE, Pagani N, Saffioti C, Ambretti S, Cardellino CS, Coppo E, De Rosa FG, Viale P, Viscoli C; ISGRI-SITA (Italian Study Group on Resistant Infections of the Società Italiana Terapia Antinfettiva) (2016) Previous bloodstream infections due to other pathogens as predictors of carbapenem-resistant Klebsiella pneumoniae bacteraemia in colonized patients: results from a retrospective multicentre study. Eur J Clin Microbiol Infect Dis
Daikos GL, Tsaousi S, Tzouvelekis LS, Anyfantis I, Psichogiou M, Argyropoulou A, Stefanou I, Sypsa V, Miriagou V, Nepka M, Georgiadou S, Markogiannakis A, Goukos D, Skoutelis A (2014) Carbapenemase-producing Klebsiella pneumoniae bloodstream infections: lowering mortality by antibiotic combination schemes and the role of carbapenems. Antimicrob Agents Chemother 58:2322–2328
Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, Sevransky JE, Sprung CL, Douglas IS, Jaeschke R, Osborn TM, Nunnally ME, Townsend SR, Reinhart K, Kleinpell RM, Angus DC, Deutschman CS, Machado FR, Rubenfeld GD, Webb SA, Beale RJ, Vincent JL, Moreno R; Surviving Sepsis Campaign Guidelines Committee including the Pediatric Subgroup (2013) Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med 41:580–637
Acknowledgements
A part of this work was submitted for presentation at the 27th European Congress of Clinical Microbiology and Infectious Diseases (ECCMID), 22–25 April 2017, Vienna, Austria.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Hospital Ethics Committee (HEC no.: 571), which waived the need for informed consent.
Rights and permissions
About this article
Cite this article
Papadimitriou-Olivgeris, M., Fligou, F., Bartzavali, C. et al. Carbapenemase-producing Klebsiella pneumoniae bloodstream infection in critically ill patients: risk factors and predictors of mortality. Eur J Clin Microbiol Infect Dis 36, 1125–1131 (2017). https://doi.org/10.1007/s10096-017-2899-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-017-2899-6