Abstract
Background
New biodegradable synthetic and biologic hernia implants have been promoted for rapid integration and tissue reinforcement in challenging repairs, e.g. at the hiatus or in contaminated wound fields. Interestingly, experimental data to support or falsify this assumption is scarce.
Methods
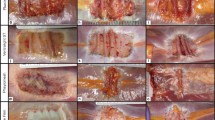
Synthetic (BioA®) and biologic implants (porcine and bovine collagen matrices Strattice® and Veritas®) have been tested in experimental onlay hernia repair in rats in observation periods of 30 and 60 days. The key outcome parameters were mesh integration and reinforcement of the tissue at the implant site over sutured and sealed defects as well as comparison to native abdominal wall. Macroscopic assessment, biomechanical analysis and histology with haematoxylin/eosin staining, collagen staining and van Willebrand factor staining for detection of neovascularization were performed.
Results
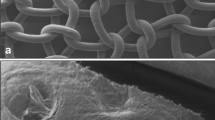
BioA® was well integrated. Although the matrices were already fragmented at 60 days follow-up, hernia sites treated with synthetic scaffolds showed a significantly enhanced tissue deflection and resistance to burst force when compared to the native abdominal wall. In porcine and bovine matrices, tissue integration and shrinkage were significantly inferior to BioA®. Histology revealed a lack of fibroblast ingrowth through mesh interstices in biologic samples, whereas BioA® was tightly connected to the underlying tissue by reticular collagen fibres.
Conclusions
Strattice® and Veritas® yielded reduced tissue integration and significant shrinkage, prohibiting further biomechanical tests. The synthetic BioA® provides little inherent strength but reticular collagen remodelling led to an augmentation of the scar due to significantly higher burst force resistance in comparison to native tissue.









Similar content being viewed by others
References
Cavallo JA, Greco SC, Liu J, Frisella MM, Deeken CR, Matthews BD (2015) Remodeling characteristics and biomechanical properties of a crosslinked versus a non-crosslinked porcine dermis scaffolds in a porcine model of ventral hernia repair. Hernia 19(2):207–218
De Castro Brás LE, Shurey S, Sibbons PD (2012) Evaluation of crosslinked and non-crosslinked biologic prostheses for abdominal hernia repair. Hernia 16:77–89
Deeken CR, Melman L, Jenkins ED, Greco SC, Frisella MM, Matthews BD (2011) Histologic and biomechanical evaluation of crosslinked and non-crosslinked biologic meshes in a porcine model of ventral incisional hernia repair. J Am Coll Surg 212:880–888
De Silva GS, Krpata DM, Gao Y, Criss CN, Anderson JM, Soltanian HT, Rosen MJ, Novitsky YW (2014) Lack of identifiable biologic behavior in a series of porcine mesh explants. Surgery 156(1):183–189
Fernandez-Moure JS, Van Eps JL, Menn ZK, Cabrera FJ, Tasciotti E, Weiner BK, Ellsworth WA IV (2015) Platelet rich plasma enhances tissue incorporation of biologic mesh. J Surg Res. pii: S0022-4804(15)00715-5. doi:10.1016/j.jss.2015.06.034 [Epub ahead of print]
Gruber-Blum S, Petter-Puchner AH, Mika K, Brand J, Redl H, Öhlinger W et al (2010) A comparison of a bovine albumin/glutaraldehyde glue versus fibrin sealant for hernia mesh fixation in experimental onlay and IPOM repair in rats. Surg End 24:3086–3094
Hood K, Millikan K, Pittman T, Zelhart M, Secemsky B, Rajan M et al (2013) Abdominal wall reconstruction: a case series of ventral hernia repair using the component separation technique with biologic mesh. Am J Surg 205:322–327 (discussion 327–328)
Leppäniemi A, Tukiainen E (2013) Reconstruction of complex abdominal wall defects. Scand J Surg 102:14–19
Melman L, Jenkins ED, Hamilton NA, Bender LC, Brodt MD, Deeken CR et al (2011) Early biocompatibility of crosslinked and non-crosslinked biologic meshes in a porcine model of ventral hernia repair. Hernia 15:157–164
Peeters E, van Barneveld KWY, Schreinemacher MH, De Hertogh G, Ozog Y, Bouvy N et al (2013) One-year outcome of biological and synthetic bioabsorbable meshes for augmentation of large abdominal wall defects in a rabbit model. J Surg Res 180:274–283
Petter-Puchner A, Fortelny RH, Mittermayr R, Walder N, Ohlinger W, Redl H (2006) Adverse effects of porcine small intestine submucosa implants in experimental ventral hernia repair. Surg End 20:942–946
Petter-Puchner AH, Fortelny RH, Silic K, Brand J, Gruber-Blum S, Redl H (2011) Biologic hernia implants in experimental intraperitoneal onlay mesh plasty repair: the impact of proprietary collagen processing methods and fibrin sealant application on tissue integration. Surg End 25:3245–3252
Petter-Puchner AH, Fortelny RH, Walder N, Mittermayr R, Öhlinger W, van Griensven M et al (2008) Adverse effects associated with the use of porcine cross-linked collagen implants in an experimental model of incisional hernia repair. J Surg Res 145:105–110
Petter-Puchner AH, Fortelny RH, Walder N, Morales-Conde S, Gruber-Blum S, Öhlinger W et al (2010) Small intestine submucosa (SIS) implants in experimental IPOM repair. J Surg Res 161:264–271
Powell BS, Wandrey D, Voeller GR (2013) A technique for placement of a bioabsorbable prosthesis with fibrin glue fixation for reinforcement of the crural closure during hiatal hernia repair. Hernia 17:81–84
Rich L, Whittaker P (2005) Collagen and picrosirius red staining: a polarized light assessment of fibrillar hue and spatial distribution. Braz J Morphol Sci 22:97–104
Rosen MJ, Denoto G, Itani KMF, Butler C, Vargo D, Smiell J et al (2013) Evaluation of surgical outcomes of retro-rectus versus intraperitoneal reinforcement with bio-prosthetic mesh in the repair of contaminated ventral hernias. Hernia 17:31–35
Shah BC, Tiwari MM, Goede MR, Eichler MJ, Hollins RR, McBride CL et al (2011) Not all biologics are equal! Hernia 15:165–171
Shaikh FM, Kennedy TE, Kavanagh EG, Grace PA (2012) Initial experience of double-layer tension free reconstruction of abdominal wall defects with porcine acellular dermal collagen implant and polypropylene mesh. Ir J Med Sci 181:205–209
Sutton PA, Evans JP, Uzair S, Varghese JV (2013) The use of Gore Bio-A in the management of the open abdomen. BMJ Case Rep. doi:10.1136/bcr-2012-008064
Acknowledgements
The authors thank K. Brenner for animal care, K. Kropik for technical support, W. Öhlinger for the histological analysis, I. Jung for statistical analysis and J. Ferguson for review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Simone Gruber-Blum, Julian Brand, Claudia Keibl, René H. Fortelny, Heinz Redl, Franz Mayer and Alexander H. Petter-Puchner declare that they have no conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed.
Human and animal rights
This article does not contain any studies with human participants performed by any of the authors.
Informed consent
None.
Rights and permissions
About this article
Cite this article
Gruber-Blum, S., Brand, J., Keibl, C. et al. Abdominal wall reinforcement: biologic vs. degradable synthetic devices. Hernia 21, 305–315 (2017). https://doi.org/10.1007/s10029-016-1556-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10029-016-1556-9