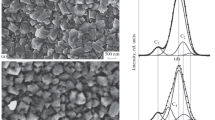
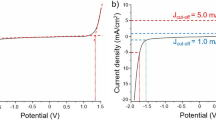
Abstract
Two important mechanisms for electron transfer processes at boron-doped diamond electrodes involving the oxidation of tetramethylphenylenediamine (TMPD) dissolved in aqueous solution and the oxidation of tetrahexylphenylenediamine (THPD) deposited in the form of microdroplets and immersed into aqueous eletrolyte solution are reported. For TMPD, the first oxidation step in aqueous solution follows the equation:
Remarkably slow heterogeneous kinetics at a H-plasma-treated boron-doped diamond electrode are observed, consistent with a process following a pathway more complex than outer-sphere electron transfer. At the same boron-doped diamond electrode surface a deposit of THPD undergoes facile oxidation following the equation:
This oxidation and re-reduction of the deposited liquid material occurs at the triple interface organic droplet|diamond|aqueous electrolyte and is therefore an example of a facile high-current-density process at boron-doped diamond electrodes due to good electrical contact between the deposit and the diamond surface.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 3 February 2000 / Accepted: 18 February 2000
Rights and permissions
About this article
Cite this article
Marken, F., Compton, R., Goeting, C. et al. Fast electrochemical triple-interface processes at boron-doped diamond electrodes. J Solid State Electrochem 5, 88–93 (2001). https://doi.org/10.1007/s100080000122
Issue Date:
DOI: https://doi.org/10.1007/s100080000122