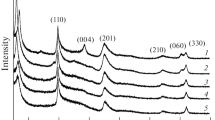
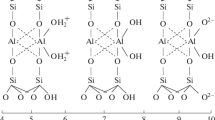
Abstract
Alkylammonium derivatives of montmorillonite (standards SAz-1 and SWy-2, Source Clay Repository (USA)) were prepared by sorption of hexadecyltrimethylammonium, benzyldimethylhexadecylammonium and hexadecylpyridinium cations. Characterization of the prepared alkylammonium montmorillonites by means of X-ray diffraction and infrared spectroscopy proved a presence of alkylammonium cations in the montmorillonites structure. Intercalation as well as ion exchange process is supposed during the alkylammonium cation sorption into the montmorillonite structure. Cyclic voltammetry of the pair [Fe(CN)6]3−/4− on carbon paste electrodes modified with the alkylammonium derivatives exhibited slightly different electrochemical behaviour of the derivatives in comparison with the original montmorillonites. Sorption properties of all alkylammonium derivatives were studied by means of batch sorption experiments (sorption of Cu, Zn, Pb) as well as by means of voltammetry of Cu(II) on carbon paste electrodes modified with the montmorillonites. The metal sorption was decreased on about 50 % in the case of the alkylammonium montmorillonites. Both techniques showed a significant decrease of Cu(II) sorption in the case of montmorillonites modified with the alkylammonium cations.










Similar content being viewed by others
References
Ray SS, Okamoto M (2003) Polymer/layered silicate nanocomposites: a review from preparation to processing. Prog Polym Sci 28:1539–1641
Centi G, Perathoner S (2008) Catalysis by layered materials: a review original. Microporous Mesoporous Mater 107:3–15
Arabi-Bergaya F (2008) Layered clay minerals. Basic research and innovative composite applications. Microporous Mesoporous Mater 107:141–148
Boyd SA, Jaynes WF (1994) In: Mermut AR (ed) ICMS workshop lectures. The Clay Minerals Society, Aurora
Lagaly G (1994) In: Mermut AR (ed) CMS workshop lectures. The Clay Minerals Society, Aurora
He H, Ma Y, Zhu J, Yuan P, Qing Y (2010) Organoclays prepared from montmorillonites with different cation exchange capacity and surfactant configuration. Appl Clay Sci 48:67–72
Liu R, Frost RL, Martens WN, Yuan Y (2008) Synthesis, characterization of mono, di and tri alkyl surfactant intercalated Wyoming montmorillonite for the removal of phenol from aqueous systems. J Colloid Interface Sci 327:287–294
Witthuhn B, Klauth P, Pernyeszi T, Vereecken H, Klumpp E (2006) Organoclays for aquifer bioremediation: adsorption of chlorobenzene on organoclays and its degradation by RHODOCOCCUS B528. Water Air Soil Pollut Focus 6:317–329
Chen D, Zhu JX, Yuan P, Yang SJ, Chen TH, He HP (2008) Preparation and characterization of anion-cation surfactants modified montmorillonite. J Therm Anal Calorim 94:841–848
Anirudhan TS, Ramachandran M (2007) Surfactant-modified bentonite as adsorbent for the removal of humic acid from wastewaters. Appl Clay Sci 35:276–281
Zhu L, Tian S, Shi Y (2005) Adsorption of volatile organic compounds onto porous clay heterostructures based on spent organobentonites. Clay Clay Miner 53:123–136
Majdan M, Sabah E, Bujacka M, Pikus S, Plaska AG (2009) Spectral and equillibrium properties of phenol–HDTMA- and phenol–BDMHDA-bentonite as a response to the molecular arrangements of surfactant cations. J Mol Struct 938:29–34
Changchaivong S, Khaodhiar S (2009) Adsorption of naphthalene and phenanthrene on dodecylpyridinium-modified bentonite. Appl Clay Sci 43:317–332
Li Z, Galus L (2007) Adsorption of dodecyl trimethylammonium and hexadecyl trimethylammonium onto kaolinite—competitive adsorption and chain length effect. Appl Clay Sci 35:250–257
Abou-El-Sherbini K, Hassanien MM (2010) Study of organically-modified montmorillonite clay for the removal of copper(II). J Hazard Mater 184:654–661
Bhattacharyya KG, Gupta KS (2008) Adsorption of a few heavy metals on natural and modified kaolinite and montmorillonite: a review. Adv Colloid Interf 140:114–131
Say R, Birlik E, Erdemgil Z, Denizli A, Ersoz A (2008) Removal of mercury species with dithiocarbamate-anchored polymer/organosmectite composites. J Hazard Mater 150:560–564
Tonle IK, Ngameni E, Walcarius A (2004) From clay- to organoclay-film modified electrodes: tuning chargé selectivity in ion exchange voltammetry. Electrochim Acta 49:3435–3443
Tonle IK, Ngameni E, Njopwouo D, Carteret C, Walcarius A (2003) Functionalization of natural smectite-type clays by grafting with organosilanes: physico-chemical characterization and application to mercury(II) uptake. Phys Chem Chem Phys 5:4951–4961
Tonle IK, Ngameni E, Walcarius A (2005) Preconcentration and voltammetric analysis of mercury(II) at a carbon paste electrode modified with natural smectite-type clays grafted with organic chelating groups. Sensors Actuators B Chem 110:195–203
Ngameni E, Tonle IK et al (2006) Permselective and preconcentration properties of a surfactant-intercalated clay modified electrode. Electroanalysis 18:2243–2250
Manisankar P, Selvanathan G, Vedhi C (2006) Determination of pesticides using heteropolyacid montmorillonite clay-modified electrode with surfactant. Talanta 68:686–692
Wagheu JK, Forano C et al (2013) Electrochemical determination of mesotrione at organoclay modified glassy carbon electrodes. Talanta 103:337–343
Fernandez L, Borrás C, Carrero H (2006) Electrochemical behavior of phenol in alkaline media at hydrotalcite-like clay/anionic surfactants/glassy carbon modified electrode. Electrochim Acta 52:872–884
Fernandez M, Fernández L, Borras C, Mostany J, Carrero H (2007) Characterization of surfactant/hydrotalcite-like clay/glassy carbon modified electrodes: oxidation of phenol. Anal Chim Acta 597:245–256
Yang H, Zheng X, Huang W, Wu K (2008) Modification of montmorillonite with cationic surfactant and application in electrochemical determination of 4-chlorophenol. Colloids Surf B 65:281–284
Newton Dias Filho L, Ribeiro do Carmo D (2006) Study of an organically modified clay: selective adsorption of heavy metal ions and voltammetric determination of mercury(II). Talanta 68:919–927
Ngassa GBP, Tonlé IK, Walcarius A, Ngameni E (2014) One-step co-intercalation of cetyltrimethylammonium and thiourea in smectite and application of the organoclay to the sensitive electrochemical detection of Pb(II). Appl Clay Sci 99:297–305
Maghear A et al (2014) Tetrabutylammonium-modified clay film electrodes: characterization and application to the detection of metal ions. Talanta 125:36–44
Marsalek R, Navratilova Z (2011) Comparative study of CTAB adsorption on bituminous coal and clay mineral. Chem Pap 65:77–84
Navratilova Z, Marsalek R (2012) In: Valaskova M, Martynkova S (eds) Clay minerals in nature—their characterization, modification and application. Intech, Croatia
Navratilova Z, Kula P (2000) Cation and anion exchange on clay modified electrodes. J Solid State Electrochem 4:342–347
Navratilova Z, Vaculikova L (2006) Electrodeposition of mercury film on electrodes modified with clay minerals. Chem Pap 60:348–352
Navratilova Z, Kula P (2003) Clay modified electrodes: present applications and prospects. Electroanalysis 15:837–846
Navratilova Z, Wojtowicz P, Vaculikova L, Sugarkova V (2007) Sorption of alkylammonium cations on montmorillonite. Acta Geodynamica Geomaterialia 4:59–65
Volzone G, Rinaldi JO, Ortiga J (2006) Retention of gases by hexadecyltrimethylammonium-montmorillonite clays. J Environ Manag 79:247–252
Lee SY, Cho WJ, Kim KJ, Ahn JH, Lee M (2005) Interaction between cationic surfactants and montmorillonites under nonequilibrium condition. J Colloid Interface Sci 284:667–673
Gammoudi S, Frini-Srasra N, Srasra E (2012) Influence of exchangeable cation of smectite on HDTMA adsorption: equilibrium, kinetic and thermodynamic studies. Appl Clay Sci 69:99–107
Garea SA, Iovu H, Voicu G (2010) The influence of some new montmorillonite modifier agents on the epoxy-montmorillonite nanocomposites structure. Appl Clay Sci 50:469–475
Weiss Z, Kužvart M (2005) Clay minerals. Their nanostructure and application (in Czech). Karolinum, Prague
Zeng Z, Jiang J (2005) Effects of the type and structure of modified clays on adsorption performance. Int J Environ Stud 62:403–414
Gupta SS, Bhattacharyya KG (2006) Adsorption of Ni (II) on clays. J Colloid Interface Sci 295:21–32
Vittal R, Gomathi H, Kj K (2006) Beneficial role of surfactants in electrochemistry and in the modification of electrodes. Adv Colloid Interf 119:55–68
Fitch A (1990) Apparent formal potential shifts in ion exchange voltammetry. J Electroanal Chem 284:237–244
Kalcher K, Grabec I, Raber G, Cai X, Tavcar G, Ogorevc B (1995) The vermiculite modified carbon paste electrode as a model system for preconcentrating mono- and divalent cations. J Electroanal Chem 386:149–156
Bozzini B, D’Urzo L et al (2008) Electrodeposition of Cu from acidic sulphate solutions containing cetyltrimethylammonium bromide (CTAB). J Appl Electrochem 38:1561–1569
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Navrátilová, Z., Mucha, M. Organo-montmorillonites as carbon paste electrode modifiers. J Solid State Electrochem 19, 2013–2022 (2015). https://doi.org/10.1007/s10008-015-2751-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-015-2751-9