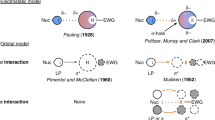
Abstract
Halogen bonding (XB) is a type of noncovalent interaction between a halogen atom X in one molecule and a negative site in another. X can be chlorine, bromine or iodine. The strength of the interaction increases in the order Cl<Br<I. After a brief review of experimental evidence relating to halogen bonding, we present an explanation for its occurrence in terms of a region of positive electrostatic potential that is present on the outermost portions of some covalently-bonded halogen atoms. The existence and magnitude of this positive region, which we call the σ-hole, depends upon the relative electron-attracting powers of X and the remainder of its molecule, as well as the degree of sp hybridization of the s unshared electrons of X. The high electronegativity of fluorine and its tendency to undergo significant sp hybridization account for its failure to halogen bond. Some computed XB interaction energies are presented and discussed. Mention is also made of the importance of halogen bonding in biological systems and processes, and in crystal engineering.

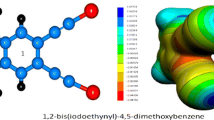
The computed B3PW91/6-31G(d,p) electrostatic potential, in kcal mol−1, on the 0.001 electrons/bohr3 surface of NC–C≡C–Cl. The chlorine atom is at the right. The color ranges are: red, more positive than 15; yellow between 7 and 15; green, between 0 and 7; blue, between −10 and 0; purple, more positive than −10.






Similar content being viewed by others
References
Guthrie F (1863) J Chem Soc 16:239–244
Remsen I, Norris JF (1896) Am Chem J 18:90–96
Mulliken RS (1952) J Am Chem Soc 74:811–824
Flurry RL Jr (1969) J Phys Chem 69:1927–1933
Flurry RL Jr (1969) J Phys Chem 73:2111–2117
Bent HA (1968) Chem Rev 68:587–648
Hassel O (1970) Science 170:497–502
Dumas J-M, Peurichard H, Gomel MJ (1978) Chem Res (S) 54–57
Dumas J-M, Geron C, Peurichard H, Gomel M (1976) Bull Soc Chim Fr 720–722
Dumas J-M, Kern M, Janier-Dubry JL (1976) Bull Soc Chim Fr 1785–1787
Murray-Rust P, Motherwell WDS (1979) J Am Chem Soc 101:4374–4376
Murray-Rust P, Stallings WC, Monti CT, Preston RK, Glusker JP (1983) J Am Chem Soc 105:3206–3214
Ramasubbu N, Parthasarathy R, Murray-Rust P (1986) J Am Chem Soc 108:4308–4314
Bernard-Houplain M-C, Sandorfy C (1973) Can J Chem 51:1075–1083
Bernard-Houplain M-C, Sandorfy C (1973) Can J Chem 3640–3647
Di Paolo T, Sandorfy C (1974) Chem Phys Lett 26:466–469
Di Paolo T, Sandorfy C (1974) Can J Chem 52:3612–3622
Brinck T, Murray JS, Politzer P (1992) Int J Quantum Chem, Quantum Biol Symp 19:57–64
Murray JS, Paulsen K, Politzer P (1994) Proc Indian Acad Sci, Chem Sci 106:267–275
Stewart RF (1972) J Chem Phys 57:1664–1668
Politzer P, Truhlar DG (eds) (1981) Chemical applications of atomic and molecular electrostatic potentials. Plenum, New York
Politzer P, Laurence PR, Jayasuriya K (1985) Environ Health Perspect 61:191–202
Murray JS, Politzer P (1998) J Mol Struct, Theochem 425:107–114
Politzer P, Murray JS (1999) Trends Chem Phys 7:157–165
Politzer P, Murray JS, Peralta-Inga Z (2001) Int J Quantum Chem 85:676–684
Bader RFW, Carroll MT, Cheeseman JR, Chang C (1987) J Am Chem Soc 109:7968–7979
Hagelin H, Brinck T, Berthelot M, Murray JS, Politzer P (1995) Can J Chem 73:483–488
Weinstein H, Politzer P, Srebrenik S (1975) Theor Chim Acta 38:159–163
Politzer P, Murray JS (2002) Theor Chem Acc 108:134–142
Scrocco E, Tomasi J (1973) Top Curr Chem 42:95–170
Politzer P, Daiker KC (1981) In: Deb BM (ed) The force concept in chemistry (Ch 6). Van Nostrand, New York
Politzer P, Murray JS (1991) In: Lipkowitz KB, Boyd DB (eds) Reviews in computational chemistry Ch 7 Vol 2. VCH, New York
Politzer P, Harris RR (1970) J Am Chem Soc 92:6451–6454 (and references cited)
Auffinger P, Hays FA, Westhof E, Shing Ho P (2004) Proc Nat Acad Sci 101:16789–16794
Clark T, Hennemann M, Murray JS, Politzer P (2006) J Mol Model DOI 10.1007/s00894-006-0130-2
Kutzelnigg W (1984) Angew Chem 96:262–269
Kutzelnigg W (1984) Angew Chem, Int Ed Engl 23:272–275
Nyburg SC, Wong-Ng W (1979) Proc Roy Soc (London) A 367:29–45
Price SL, Stone AJ, Lucas J, Rowland RS, Thornley AE (1994) J Am Chem Soc 116:4910–4918
Lommerse JPM, Stone AJ, Taylor R, Allen FH (1996) J Am Chem Soc 118:3108–3116
Valerio G, Raos G, Meille SV, Metrangolo P, Resnati G (2000) J Phys Chem, A 104:1617–1620
Romaniello P, Lelj F (2002) J Phys Chem, A 106:9114–9119
Larsen DW, Allred AL (1965) J Phys Chem 69:2400–2401
Corradi E, Meille SV, Messina MT, Metrangolo P, Resnati G (2000) Angew Chem, Int Ed Engl 39:1782–1786
Metrangolo P, Neukirch H, Pilati T, Resnati G (2005) Acc Chem Res 38:386–395
Cody V, Murray-Rust P (1984) J Mol Struct 112:189–199
De Moliner E, Brown NR, Johnson LN (2003) Eur J Biochem 270:3174–3181
Metrangolo P, Pilati T, Resnati G, Stevenazzi A (2003) Curr Opin Colloid Interface Sci 8:215–222
Thallapally PK, Desiraju GR, Bagien-Bencher M, Masse R, Bourgogne C, Nicoud JF (2002) Chem Commun 1052–1053
Imakubo T, Tajima N, Tamura M, Kato R, Nishio Y, Kajita K (2003) Synth Met 133–134:181–183. DOI 10.1007/s00894-006-0130-2
Metrangolo P, Neukirch H, Pilati T, Resnati G (2005) Acc Chem Res 38:393–394
Acknowledgment
We would like to express our gratitude to Professor Jaroslav Burda, who very kindly provided facilities at the Charles University in Prague so that we could prepare this paper while most of us were evacuees from Hurricane Katrina, which hit New Orleans on August 29, 2005.
Author information
Authors and Affiliations
Corresponding author
Additional information
Proceedings of “Modeling Interactions in Biomolecules II”, Prague, September 5th–9th, 2005.
Rights and permissions
About this article
Cite this article
Politzer, P., Lane, P., Concha, M.C. et al. An overview of halogen bonding. J Mol Model 13, 305–311 (2007). https://doi.org/10.1007/s00894-006-0154-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-006-0154-7