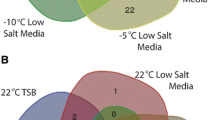
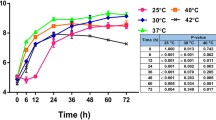
Abstract
It is crucial to examine the physiological processes of psychrophiles at temperatures below 4°C, particularly to facilitate extrapolation of laboratory results to in situ activity. Using two dimensional electrophoresis, we examined patterns of protein abundance during growth at 16, 4, and −4°C of the eurypsychrophile Psychrobacter cryohalolentis K5 and report the first identification of cold inducible proteins (CIPs) present during growth at subzero temperatures. Growth temperature substantially reprogrammed the proteome; the relative abundance of 303 of the 618 protein spots detected (∼31% of the proteins at each growth temperature) varied significantly with temperature. Five CIPs were detected specifically at −4°C; their identities (AtpF, EF-Ts, TolC, Pcryo_1988, and FecA) suggested specific stress on energy production, protein synthesis, and transport during growth at subzero temperatures. The need for continual relief of low-temperature stress on these cellular processes was confirmed via identification of 22 additional CIPs whose abundance increased during growth at −4°C (relative to higher temperatures). Our data suggested that iron may be limiting during growth at subzero temperatures and that a cold-adapted allele was employed at −4°C for transport of iron. In summary, these data suggest that low-temperature stresses continue to intensify as growth temperatures decrease to −4°C.





Similar content being viewed by others
Abbreviations
- CIP:
-
Cold inducible protein
- CAP:
-
Cold acclimation protein
- CSP:
-
Cold shock protein
References
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Anderson NG, Anderson NL (1978a) Analytical techniques for cell fractions. XXI. Two-dimensional analysis of serum and tissue proteins multiple isoelectric focusing. Anal Biochem 85:331–340
Anderson NL, Anderson NG (1978b) Analytical techniques for cell fractions. XXI. Two-dimensional analysis of serum and tissue proteins multiple gradient slab-gel electrophoresis. Anal Biochem 85:341−354
Antelmann H, Schmid R, Hecker M (1997) The NAD synthetase NadE (OutB) of Bacillus subtilis is a sigma(B)-dependent general stress protein. FEMS Microbiol Lett 153:405–409
Araki T (1991) The effect of temperature shifts on protein synthesis by the psychrophilic bacterium Vibrio sp. strain ANT-300. J Gen Microbiol 137:817–826
Bakermans C, Nealson KH (2004) Relationship of critical temperature to macromolecular synthesis and growth yield in “Psychrobacter cryopegella”. J Bacteriol 186:2340–2345
Bakermans C, Tsapin AI, Souza-Egipsy V, Gilichinsky DA, Nealson KH (2003) Reproduction and metabolism at −10°C of bacteria isolated from Siberian permafrost. Environ Microbiol 5:321–326
Bakermans C, Ayala-del-Río HL, Ponder MA, Vishnivetskaya T, Gilichinsky D, Thomashow MF, Tiedje JM (2006) Psychrobacter cryohalolentis sp. nov. and Psychrobacter arcticus sp. nov. isolated from Siberian permafrost. Int J Syst Evol Microbiol 56:1285–1291
Bayles DO, Annous BA, Wilkinson BJ (1996) Cold stress proteins in Listeria monocytogenes in response to temperature downshock and growth at low temperatures. Appl Environ Microbiol 62:1116–1119
Berger F, Normand P, Potier P (1997) CapA, a cspA-like gene that encodes a cold acclimation protein in the psychrotrophic bacterium Arthrobacter globiformis SI55. J Bacteriol 179:5670–5676
Berger F, Morellet N, Menu F, Potier P (1996) Cold shock and cold acclimation proteins in the psychrotrophic bacterium Arthrobacter globiformis SI55. J Bacteriol 178:2999–3007
Broeze RJ, Solomon CJ, Pope DH (1978) Effects of low-temperature on in vivo and in vitro protein-synthesis in Escherichia coli and Pseudomonas fluorescens. J Bacteriol 134:861–874
Budde I, Steil L, Scharf C, Volker U, Bremer E (2006) Adaptation of Bacillus subtilis to growth at low temperature: a combined transcriptomic and proteomic appraisal. Microbiology 152:831–853
Cavicchioli R, Thomas T, Curmi PMG (2000) Cold stress response in Archaea. Extremophiles 4:321–331
Cloutier J, Prevost D, Nadeau P, Antoun H (1992) Heat and cold shock protein synthesis in Arctic and temperate strains of Rhizobia. Appl Environ Microbiol 58:2846–2853
Colucci MS, Inniss WE (1996) Ethylene glycol utilization, cold and ethylene glycol shock and acclimation proteins in a psychrotrophic bacterium. Curr Microbiol 32:179–182
Drouin P, Prevost D, Antoun H (2000) Physiological adaptation to low temperatures of strains of Rhizobium leguminosarum bv. viciae associated with Lathyrus spp. FEMS Microbiol Ecol 32:111–120
Farris M, Grant A, Richardson TB, O’Connor CD (1998) BipA: a tyrosine-phosphorylated GTPase that mediates interactions between enteropathogenic Escherichia coli (EPEC) and epithelial cells. Mol Microbiol 28:265–279
Fedorov R, Meshcheryakov V, Gongadze G, Fomenkova N, Nevskaya N, Selmer M et al (2001) Structure of ribosomal protein TL5 complexed with RNA provides new insights into the CTC family of stress proteins. Acta Crystallogr D Biol Crystallogr 57:968–976
Feller G, Gerday C (2003) Psychrophilic enzymes: hot topics in cold adaptation. Nature Rev Microbiol 1:200–208
Gardan R, Duche O, Leroy-Setrin S, Labadie J (2003) Role of Ctc from Listeria monocytogenes in osmotolerance. Appl Environ Microbiol 69:154–161
Georlette D, Blaise V, Collins T, D’Amico S, Gratia E, Hoyoux A et al (2004) Some like it cold: biocatalysis at low temperatures. FEMS Microbiol Rev 28:25–42
Giometti CS, Gemmell MA, Tollaksen SL, Taylor J (1991a) Quantitation of human leukocyte proteins after silver staining: a study with two-dimensional electrophoresis. Electrophoresis 12:536–543
Goodchild A, Saunders NFW, Ertan H, Raftery M, Guilhaus M, Curmi PMG, Cavicchioli R (2004) A proteomic determination of cold adaptation in the Antarctic archaeon, Methanococcoides burtonii. Mol Microbiol 53:309–321
Goodchild A, Raftery M, Saunders NFW, Guilhaus M, Cavicchioli R (2005) Cold adaptation of the Antarctic archaeon, Methanococcoides burtonii assessed by proteomics using ICAT. J Proteome Res 4:473–480
Grammann K, Volke A, Kunte HJ (2002) New type of osmoregulated solute transporter identified in halophilic members of the Bacteria domain: TRAP transporter TeaABC mediates uptake of ectoine and hydroxyectoine in Halomonas elongata DSM 2581T. J Bacteriol 184:3078–3085
Grant AJ, Farris M, Alefounder P, Williams PH, Woodward MJ, O’Connor CD (2003) Co-ordination of pathogenicity island expression by the BipA GTPase in enteropathogenic Escherichia coli (EPEC). Mol Microbiol 48:507–521
Graumann P, Marahiel MA (1996) Some like it cold: response of microorganisms to cold shock. Arch Microbiol 166:293–300
Graumann P, Schroder K, Schmid R, Marahiel M (1996) Cold shock stress-induced proteins in Bacillus subtilis. J Bacteriol 178:4611–4619
Gygi SP, Rochon Y, Franza BR, Aebersold R (1999) Correlation between protein and mRNA abundance in yeast. Mol Cell Biol 19:1720–1730
Hamel R, Appanna VD, Viswanatha T, Puiseux-Dao S (2004) Overexpression of isocitrate lyase is an important strategy in the survival of Pseudomonas fluorescens exposed to aluminum. Biochem Biophys Res Commun 317:1189–1194
He HJ, Gordon R, Gow JA (2001) The effect of temperature on the fatty acids and isozymes of a psychrotrophic and two mesophilic species of Xenorhabdus, a bacterial symbiont of entomopathogenic nematodes. Can J Microbiol 47:382–391
Hebraud M, Potier P (1999) Cold shock response and low temperature adaptation in psychrotrophic bacteria. J Mol Microbiol Biotech 1:211–219
Hebraud M, Dubois E, Potier P, Labadie J (1994) Effect of growth temperature on the protein levels in a psychrotrophic bacterium, Pseudomonas fragi. J Bacteriol 176:4017–4024
Hecker M, Volker U (1998) Non-specific, general and multiple stress resistance of growth-restricted Bacillus subtilis cells by the expression of the sigma B regulon. Mol Microbiol 29:1129–1136
Herendeen S, van Bogelen RA, Neidhardt FC (1979) Levels of major proteins of Escherichia coli during growth at different temperatures. J Bacteriol 139:185–194
Homma T, Iwahashi H, Komatsu Y (2003) Yeast gene expression during growth at low temperature. Cryobiology 46:230–237
Ishii A, Ochiai T, Imagawa S, Fukunaga N, Sasaki S, Minowa O et al (1987) Isozymes of isocitrate dehydrogenase from an obligately psychrophilic bacterium, Vibrio sp strain Abe-1-purification, and modulation of activities by growth-conditions. J Biochem 102:1489–1498
Jiang W, Hou Y, Inouye M (1997) CspA, the major cold-shock protein of Escherichia coli, is an RNA chaperone. J Biol Chem 272:196–202
Jones PG, Inouye M (1996) RbfA, a 30S ribosomal binding factor, is a cold-shock protein whose absence triggers the cold-shock response. Mol Microbiol 21:1207–1218
Jones PG, van Bogelen RA, Neidhardt FC (1987) Induction of proteins in response to low temperature in Escherichia coli. J Bacteriol 169:2092–2095
Kaan T, Homuth G, Mader U, Bandow J, Schweder T (2002) Genome-wide transcriptional profiling of the Bacillus subtilis cold-shock response. Microb 148:3441–3455
Kiss E, Huguet T, Poinsot V, Batut J (2004) The typA gene is required for stress adaptation as well as for symbiosis of Sinorhizobium meliloti 1021 with certain Medicago truncatula lines. Mol Plant Microbe Interact 17:235–244
Kolker E, Purvine S, Galperin MY, Stolyar S, Goodlett DR, Nesvizhskii AI et al (2003) Initial proteome analysis of model microorganism Haemophilus influenzae strain Rd KW20. J Bacteriol 185:4593–4602
Lesniak J, Barton WA, Nikolov DB (2003) Structural and functional features of the Escherichia coli hydroperoxide resistance protein OsmC. Protein Sci 12:2838–2843
Liu S, Graham JE, Bigelow L, Morse PD, Wilkinson BJ (2002) Identification of Listeria monocytogenes genes expressed in response to growth at low temperature. Appl Environ Microbiol 68:1697–1705
Nedwell DB (1999) Effect of low temperature on microbial growth: lowered affinity for substrates limits growth at low temperature. FEMS Microb Ecol 30:101–111
O’Farrell PH (1975) High-resolution two-dimensional electrophoresis of proteins. J Biol Chem 250:4007−4021
Panoff J-M, Legrand S, Thammavongs B, Boutibonnes P (1994) The cold shock response in Lactococcus lactis subsp. lactis. Curr Microbiol 29:213–216
Panoff J-M, Corroler D, Thammavongs B, Boutibonnes P (1997) Differentiation between cold shock proteins and cold acclimation proteins in a mesophilic gram-positive bacterium, Enterococcus faecalis JH2–2. J Bacteriol 179:4451–4454
Pavlovic M, Hormann S, Vogel RF, Ehrmann MA (2005) Transcriptional response reveals translation machinery as target for high pressure in Lactobacillus sanfranciscensis. Arch Microbiol 184:11–17
Phadtare S, Inouye M (2004) Genome-wide transcriptional analysis of the cold shock response in wild-type and cold-sensitive, quadruple-csp-deletion strains of Escherichia coli. J Bacteriol 186:7007–7014
Ponder MA, Gilmour SJ, Bergholz PW, Mindock CA, Hollingsworth R, Thomashow MF, Tiedje JM (2005) Characterization of potential stress responses in ancient Siberian permafrost psychroactive bacteria. FEMS Microbiol Ecol 53:103–115
Pruss BM, Francis KP, von Stetten F, Scherer S (1999) Correlation of 16S ribosomal DNA signature sequences with temperature-dependent growth rates of mesophilic and psychrotolerant strains of the Bacillus cereus group. J Bacteriol 181:2624–2630
Ramagli LS, Rodriguez LV (1985) Quantitation of microgram amounts of protein in two-dimensional polyacrylamide gel electrophoresis sample buffer. Electrophoresis 6:559–563
Roberts ME, Inniss WE (1992) The synthesis of cold shock proteins and cold acclimation proteins in the psychrophilic bacterium Aquaspirillum arcticum. Curr Microbiol 25:275–278
Russell N, Hamamoto T (1998) Psychrophiles. In: Horikoshi K, Grant WD (eds) Extremophiles: microbial life in extreme environments. Wiley, New York, pp 25–45
Sardesai N, Babu C (2000) Cold stress induces switchover of respiratory pathway to lactate glycolysis in psychrotrophic Rhizobium strains. Folia Microbiol (Praha) 45:177–182
Sardesai N, Babu CR (2001) Poly-beta-hydroxybutyrate metabolism is affected by changes in respiratory enzymatic activities due to cold stress in two psychrotrophic strains of Rhizobium. Curr Microbiol 42:53–58
Scherer S, Neuhaus K (2002) Life at low temperatures. In: Dworkin M (ed) The prokaryotes: an evolving electronic resource for the microbiological community, 3rd edn, release 3.9. Springer, Berlin Heidelberg, New York. http://link.springer-ny.com/link/service/books/10125/
Schmalisch M, Langbein I, Stulke J (2002) The general stress protein Ctc of Bacillus subtilis is a ribosomal protein. J Mol Microbiol Biotech 4:495–501
Smirnova GV, Zakirova ON, Oktyabrskii ON (2001) The role of antioxidant systems in the cold stress response of Escherichia coli. Microbiology 70:45–50
von Stetten F, Francis KP, Lechner S, Neuhaus K, Scherer S (1998) Rapid discrimination of psychrotolerant and mesophilic strains of the Bacillus cereus group by PCR targeting of 16S rDNA. J Microbiol Meth 34:99–106
Strocchi M, Ferrer M, Timmis KN, Golyshin PN (2006) Low temperature-induced systems failure in Escherichia coli: insights from rescue by cold-adapted chaperones. Proteomics 6:193–206
Summers ML, Denton MC, McDermott TR (1999) Genes coding for phosphotransacetylase and acetate kinase in Sinorhizobium meliloti are in an operon that is inducible by phosphate stress and controlled by PhoB. J Bacteriol 181:2217–2224
Weinberg MV, Schut GJ, Brehm S, Datta S, Adams MWW (2005) Cold shock of a hyperthermophilic archaeon: Pyrococcus furiosus exhibits multiple responses to a suboptimal growth temperature with a key role for membrane-bound glycoproteins. J Bacteriol 187:336–348
Welsh DT (2000) Ecological significance of compatible solute accumulation by micro-organisms: from single cells to global climate. FEMS Microbiol Rev 24:263–290
Whyte LG, Inniss WE (1992) Cold shock proteins and cold acclimation proteins in a psychrotrophic bacterium. Can J Microbiol 38:1281–1285
Yamanka K, Fang L, Inouye M (1998) The CspA family of Escherichia coli: multiple gene duplication for stress adaptation. Mol Microbiol 27:247–255
Acknowledgments
This work was supported through membership in the NASA Astrobiology Institute. C. Bakermans was supported by a National Academy of Sciences National Research Council (Postdoctoral Associateship 0385260). The 2DE gel work was performed at Argonne National Laboratory under funding from the US Department of Energy, Office of Biological and Environmental Research Microbial Genome Program under Contract W-31-109-ENG-38. Thanks to T. Khare (ANL) and B. Phinney (UC Davis) for work on preliminary studies.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K. Horikoshi.
Rights and permissions
About this article
Cite this article
Bakermans, C., Tollaksen, S.L., Giometti, C.S. et al. Proteomic analysis of Psychrobacter cryohalolentis K5 during growth at subzero temperatures. Extremophiles 11, 343–354 (2007). https://doi.org/10.1007/s00792-006-0042-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-006-0042-1