Abstract
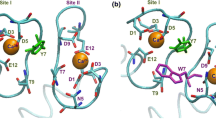
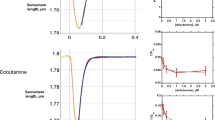
Human cardiac troponin C (HcTnC), a member of the EF hand family of proteins, is a calcium sensor responsible for initiating contraction of the myocardium. Ca2+ binding to the regulatory domain induces a slight change in HcTnC conformation which modifies subsequent interactions in the troponin–tropomyosin–actin complex. Herein, we report a calorimetric study of Ca2+ binding to HcTnC. Isotherms obtained at 25 °C (10 mM 2-morpholinoethanesulfonic acid, 50 mM KCl, pH 7.0) provided thermodynamic parameters for Ca2+ binding to both the high-affinity and the low-affinity domain of HcTnC. Ca2+ binding to the N-domain was shown to be endothermic in 2-morpholinoethanesulfonic acid buffer and allowed us to extract the thermodynamics of Ca2+ binding to the regulatory domain. This pattern stems from changes that occur at the Ca2+ site rather than structural changes of the protein. Molecular dynamics simulations performed on apo and calcium-bound HcTnC1–89 support this claim. The values of the Gibbs free energy for Ca2+ binding to the N-domain in the full-length protein and to the isolated domain (HcTnC1–89) are similar; however, differences in the entropic and enthalpic contributions to the free energy provide supporting evidence for the cooperativity of the C-domain and the N-domain. Thermograms obtained at two additional temperatures (10 and 37 °C) revealed interesting trends in the enthalpies and entropies of binding for both thermodynamic events. This allowed the determination of the change in heat capacity (∆C p ) from a plot of ∆H verses temperature and may provide evidence for positive cooperativity of Ca2+ binding to the C-domain.





Similar content being viewed by others
References
Schaub MC, Heizmann CW (2008) Biochem Biophys Res Commun 369:247–264
Gifford JL, Walsh MP, Vogel HJ (2007) Biochem J 405:199–221
Krebs J, Heizmann CW (2007) Calcium-binding proteins and the EF-hand principle. Elsevier, Amsterdam
Grabarek Z (2006) J Mol Biol 359:509–525
Capozzi F, Casadei F, Luchinat C (2006) J Biol Inorg Chem 11:949–962
Bhattacharya S, Bunick CG, Chazin WJ (2004) Biochim Biophys Acta Mol Cell Res 1742:69–79
Crouch TH, Klee CB (1980) Biochemistry 19:3692–3698
Teleman O, Drakenberg T, Forsen S, Thulin E (1983) Eur J Biochem 134:453–457
Pearlstone JR, Mccubbin WD, Kay CM, Sykes BD, Smillie LB (1992) Biochemistry 31:9703–9708
Linse S, Chazin WJ (1995) Protein Sci 4:1038–1044
Sia SK, Li MX, Spyracopoulos L, Gagne SM, Putkey JA, Sykes BD (1997) Biophys J 72:WP191–WP191
Potter JD, Gergely J (1975) J Biol Chem 250:4628–4633
Leavis PC, Kraft EL (1978) Arch Biochem Biophys 186:411–415
Johnson JD, Potter JD (1978) J Biol Chem 253:3775–3777
Johnson JD, Collins JH, Potter JD (1978) J Biol Chem 253:6451–6458
Johnson JD, Potter JD (1978) J Biol Chem 253:3775–3777
Takahashi K (1976) Biochemistry 15:1171–1180
Cheung JY, Tillotson DL, Yelamarty RV, Scaduto RC (1989) Am J Physiol 256:C1120–C1130
Spyracopoulos L, Li MX, Sia SK, Gagne SM, Chandra M, Solaro RJ, Sykes BD (1997) Biochemistry 36:12138–12146
Gomes AV, Potter JD (2004) Mol Cell Biochem 263:99
Dweck D, Hus N, Potter JD (2008) J Biol Chem 283:33119–33128
Kimura A (2011) Circ J 75:1756–1765
Jacoby D, McKenna WJ (2012) Eur Heart J 33:163–296
Frey N, Luedde M, Katus HA (2012) Nat Rev Cardiol 9:91–100
Wilcox DE (2008) Inorg Chim Acta 361:857–867
Grossoehme NE, Spuches AM, Wilcox DE (2010) J Biol Inorg Chem 15:1183–1191
Sacco C, Skowronsky RA, Gade S, Kenney AM, Spuches AM (2012) J Biol Inorg Chem 17:531–541
Kometani K, Yamada K (1983) Biochem Biophys Res Commun 114:162–167
Yamada K (1999) Mol Cell Biochem 190:39–45
Yamada K (2003) Adv Exp Med Biol 538:203–213
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) J Chem Phys 79:926–935
Toukmaji A, Sagui C, Board J, Darden T (2000) J Chem Phys 113:10913–10927
Essmann U, Perera L, Berkowitz ML, Darden T, Lee H, Pedersen LG (1995) J Chem Phys 103:8577–8593
Ryckaert JP, Ciccotti G, Berendsen HJC (1977) J Comput Phys 23:327–341
Case DA, Cheatham TE, Darden T, Gohlke H, Luo R, Merz KM, Onufriev A, Simmerling C, Wang B, Woods RJ (2005) J Comput Chem 26:1668–1688
Christensen T, Gooden DM, Kung JE, Toone EJ (2003) J Am Chem Soc 125:7357–7366
Martell AE, Smith RM (2004) NIST Critically Selected Stability Constants of Metal Complexes Database, version 8.0. National Institute of Standards and Technology, Gaithersburg
Yamada K (1978) Biochim Biophys Acta Protein Struct 535:342–347
Li MX, Chandra M, Pearlstone JR, Racher KI, Trigogonzalez G, Borgford T, Kay CM, Smillie LB (1994) Biochemistry 33:917–925
Tanokura M, Yamada K (1993) J Biol Chem 268:7090–7092
Tanokura M, Yamada K (1984) J Biochem 95:643–649
Tanokura M, Yamada K (1987) Biochemistry 26:7668–7674
Imaizumi M, Tanokura M, Yamada K (1987) J Biol Chem 262:7963–7966
Tanokura M, Yamada K (1983) J Biochem 94:607–609
Gilli R, Lafitte D, Lopez C, Kilhoffer M-, Makarov A, Briand C, Haiech J (1998) Biochemistry 37:5450–5456
Smith D (1977) J Chem Educ 54(19):540–542
Sturtevant JM (1977) Proc Natl Acad Sci USA 74:2236–2240
Varughese JF, Chalovich JM, Li Y (2010) J Biomol Struct Dyn 28:159–173
Varughese JF, Li Y (2011) J Biomol Struct Dyn 29:123–135
Varughese JF, Baxley T, Chalovich JM, Li Y (2011) J Phys Chem B 115:2392–2400
Wang JM, Morin P, Wang W, Kollman PA (2001) J Am Chem Soc 123:5221–5230
Wang W, Kollman PA (2000) J Mol Biol 303:567–582
Kollman PA, Massova I, Reyes C, Kuhn B, Huo SH, Chong L, Lee M, Lee T, Duan Y, Wang W, Donini O, Cieplak P, Srinivasan J, Case DA, Cheatham TE (2000) Acc Chem Res 33:889–897
Acknowledgments
We thank Nick Grossoehme, Dean Wilcox, Andrew Morehead, and Colin Burns for their useful comments and advice. We also give special thanks to Jason King and the Toone laboratory for allowing us to use their calorimeter when ours was being repaired. We thank East Carolina University for the start-up funds for this work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Skowronsky, R.A., Schroeter, M., Baxley, T. et al. Thermodynamics and molecular dynamics simulations of calcium binding to the regulatory site of human cardiac troponin C: evidence for communication with the structural calcium binding sites. J Biol Inorg Chem 18, 49–58 (2013). https://doi.org/10.1007/s00775-012-0948-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-012-0948-2