Abstract
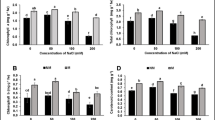
Salinity is a major stress that adversely affects plant growth and crop production. Understanding the cellular responses and molecular mechanisms by which plants perceive and adopt salinity stress is of fundamental importance. In this work, some of the cellular signaling events including cell death, reactive oxygen species (ROS) generation, and the behaviors of organelles were analyzed in a salt-tolerant species (Keyuan-1) of peppermint (Mentha × piperita L.) under NaCl treatment. Our results showed that 200 mM NaCl treatment elicited a distinct progress of cell death with chromatin condensation and caspase-3-like activation and a dramatic burst of ROS which was required for the execution of cell death. The major ROS accumulation occurred in the mitochondria and chloroplasts, which were the sources of ROS production under NaCl stress. Moreover, mitochondrial activity and photosynthetic capacity also exhibited the obvious decrease in the ROS-dependent manner under 200 mM NaCl stress. Furthermore, the activities of superoxide dismutase (SOD), ascorbate peroxidase (APX), glutathione reductase (GR), and dehydroascorbate reductase (DHAR) as well as the contents of ascorbate and glutathione changed in the concentration-dependent manner under NaCl stress. Altogether, our data showed the execution of programmed cell death (PCD), the ROS dynamics, and the behaviors of organelles especially mitochondria and chloroplasts in the cellular responses of peppermint to NaCl stress which can be used for the tolerance screening, and contributed to the understanding of the cellular responses and molecular mechanisms of peppermint to salinity stress, providing the theoretic basis for the further development and utilization of peppermint in saline areas.








Similar content being viewed by others
References
Affenzeller MJ, Dareshouri A, Andosch A, Lutz C, Lutz-Meindl U (2009) Salt stress-induced cell death in the unicellular green alga Micrasterias denticulata. J Exp Bot 60:939–954
Alscher RG, Erturk N, Heath LS (2002) Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J Exp Bot 53:1331–1341
Anderson JV, Chevone BI, Hess JL (1992) Seasonal variation in the antioxidant system of eastern white pine needles. Plant Physiol 98:501–508
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Ashtamker C, Kiss V, Sagi M, Davydov O, Fluhr R (2007) Diverse subcellular locations of cryptogein-induced reactive oxygen species production in tobacco bright yellow-2 cells. Plant Physiol 143:1817–1826
Baier M, Dietz KJ (2005) Chloroplasts as source and target of cellular redox regulation: a discussion on chloroplast redox signals in the context of plant physiology. J Exp Bot 416:1449–1462
Baker NR, Rosenqvist E (2004) Applications of chlorophyll fluorescence can improve crop production strategies: an examination of future possibilities. J Exp Bot 55:1607–1621
Behn H, Albert A, Marx F, Noga G, Ulbrich A (2010) Ultraviolet-B and photosynthetically active radiation interactively affect yield and pattern of monoterpenes in leaves of peppermint (Mentha × piperita L.). J Agric Food Chem 58:7361–7367
Chaerle L, Van Der Straeten D (2000) Imaging techniques and the early detection of plant stress. Trends Plant Sci 5:495–500
Chen Q, Vazquez EJ, Moghaddas S, Hoppel CL (2003) Production of reactive oxygen species by mitochondria. J Biol Chem 278:36027–36031
Clark RJ, Menary RC (1980) Environmental effects on peppermint, I. Effect of day length, photon flux density, night temperature and day temperature on the yield and composition of peppermint oil. Australian J Plant Physiol 7:685–692
Flowers TJ, Yeo AR (1995) Breeding for salinity resistance in crop plants—where next? Australian J Plant Physiol 22:875–884
Gao XH, Ren ZH, Zhao YX, Zhang H (2003) Overexpression of SOD2 increases salt tolerance of Arabidopsis. Plant Physiol 133:1873–1881
Garnier L, Simon-Plas F, Thuleau P (2006) Cadmium affects tobacco cells by a series of three waves of reactive oxygen species that contribute to cytotoxicity. Plant Cell Environ 29:1956–1969
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Annu Rev Plant Physiol Plant Mol Biol 51:463–499
Hiroko PI, Mercy D, Hsiu-Chuan Y, Shigeaki S, Kazuo T, Takeshi N, Masahiro H, Yasutoshi K, Toshihiko O, Hideyuki JM (2007) Evidence of ROS generation by mitochondria in cells with impaired electron transport chain and mitochondrial DNA damage. Mitochondrion 7:106–118
Huh GH, Damsz B, Matsumoto TK, Reddy MP, Rus AM, Ibeas JI, Narasimhan ML, Bressan RA, Hasegawa PM (2002) Salt causes ion disequilibrium-induced programmed cell death in yeast and plants. The Plant J 29:649–659
Jeffrey SA (2006) The role of the mitochondrial permeability transition in cell death. Mitochondrion 6:225–234
Kholova J, Sairam RK, Meena RC (2010) Osmolytes and metal ions accumulation, oxidative stress and antioxidant enzymes activity as determinants of salinity stress tolerance in maize genotypes. Acta Physiol Plant 32:477–486
Kroemer G (1999) Mitochondrial control of apoptosis: an overview. Biochem Soc Symp 66:1–15
Li Z, Xing D (2011) Mechanistic study of mitochondria-dependent programmed cell death induced by aluminum phytotoxicity using fluorescence techniques. J Exp Bot 62:331–343
Li Z, Xing FQ, Xing D (2012) Characterization of target site of aluminum phytotoxicity in photosynthetic electron transport by fluorescence techniques in tobacco leaves. Plant Cell Physiol. doi:10.1093/pcp/PCS076
Li Z, Wu XQ, Wei YL, Li JS, Yang HT (2014a) Effects of NaCl stress on the growth and antioxidant system of peppermint (Mentha piperita L.). Guangdong Agricul Sci 41:46–51
Li Z, Wu XQ, Zhao XY, Wei YL, Li JS, Yang HT (2014b) Impacts of NaCl stress on photosynthesis and PSII photochemical activity of peppermint (Mentha piperita L.). Shandong Sci 27:56–62
Lin J, Wang Y, Wang G (2005) Salt stress-induced programmed cell death via Ca2+-mediated mitochondrial permeability transition in tobacco protoplasts. Plant Growth Regul 45:243–250
Mitsuya S, Kawasaki M, Taniguchi M, Miyake H (2003) Light dependency of salinity-induced chloroplast degradation. Plant Prod Sci 6:219–223
Moller IM, Rasmusson AG (2005) Isolation of mitochondria. In: Taiz L, Zeiger E, eds. Plant physiology, 4th edn. Chapter 11
Omoto E, Nagao H, Taniguchi M, Miyake H (2013) Localization of reactive oxygen species and change of antioxidant capacities in mesophyll and bundle sheath chloroplasts of maize under salinity. Physiol Plantarum 149:1–12
Pennell RI, Lamb C (1997) Programmed cell death in plants. The Plant Cell 9:1157–1168
Romero-Puertas MC, Rodriguez-Serrano M, Corpas FJ, Gomez M, Delrio LA, Sandalio LM (2004) Cadmium-induced subcellular accumulation of O2 − and H2O2 in pea leaves. Plant Cell Environ 27:1122–1134
Santoro MV, Nievas F, Zygadlo J, Giordano W, Banchio E (2013) Effects of growth regulators on biomass and the production of secondary metabolites in Peppermint (Mentha piperita) micropropagated in vitro. Am J Plant Sci 4:49–55
Shabala S (2009) Salinity and programmed cell death: unraveling mechanisms for ion specific signaling. J Exp Bot 60:709–712
Shabala S, Cuin TA (2008) Potassium transport and plant salt tolerance. Physiol Plantarum 133:651–669
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 135:1–9
Tanaka A, Tanaka R (2006) Chlorophyll metabolism. Curr Opin Plant Biol 9:248–255
Tsanko SG, Jacques H (2005) Hydrogen peroxide as a signal controlling plant programmed cell death. J Cell Biol 168:17–20
Vitaly DS, Elena ML, Dmitry BK, Elena VD, Yana VM, Mikhail VG (2003) Participation of chloroplasts in plant apoptosis. Biosci Report 3:103–117
Wang J, Bao JT, Li XR, Liu YB (2010) Salt stress induced apoptotic-like changes in Thellungiella halophila. Sci Cold Arid Reg 2:31–42
Yamaguchi T, Blumwald E (2005) Developing salt-tolerant crop plants: challenges and opportunities. Trends Plant Sci 10:615–620
Yamane K, Bunai K, Kakeshita H (2004) Protein traffic for secretion and related machinery of Bacillus subtilis. Biosci Biotechnol Biochem 68:2007–2023
Yao N, Tada Y, Sakamoto M, Nakayashiki H, Park P, Tosa Y, Mayama S (2002) Mitochondrial oxidative burst involved in apoptotic response in oats. The Plant J 30:567–579
Yao N, Bartholomew JE, James M, Greenberg JT (2004) The mitochondrion: an organelle commonly involved in programmed cell death in Arabidopsis thaliana. The Plant J 40:596–610
Zhang LR, Xing D (2008) Rapid determination of the damage to photosynthesis caused by salt and osmotic stresses using delayed fluorescence of chloroplast. Photochem Photobiol Sci 7:352–360
Zhang X, Zhou S, Fu Y, Su Z, Wang X, Sun C (2006) Identification of a drought tolerant introgression line derived from Dongxiang common wild rice (O. rufipogon Griff.). Plant Mol Biol 62:247–259
Acknowledgments
This research is supported by the Programs for International S&T Cooperation Program of China (2011DFA30990; 2007DFA30630) and the Youth Science Funds of Shandong Academy of Sciences (2013QN012).
Conflict of interest
The authors declared that they have no conflicts of interest to this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Néstor Carrillo
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 14018 kb)
Rights and permissions
About this article
Cite this article
Li, Z., Yang, H., Wu, X. et al. Some aspects of salinity responses in peppermint (Mentha × piperita L.) to NaCl treatment. Protoplasma 252, 885–899 (2015). https://doi.org/10.1007/s00709-014-0728-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-014-0728-7