Abstract
To give further insight into the influence of the structural modifications of enones and dienones on their antiproliferative properties, 25 derivatives of enones: (E)-2-benzylidene-1-cyclohexanones, (E)-2-benzylidene-1-tetralones, (E)-2-benzylidene-1-indanone, and dienones: (E,E)-2,5- or 2,6-dibenzylidene-1-cyclanones, (E,E)-3,5-dibenzylidene-4-piperidones were synthesized using a newly developed “one-pot” synthetic method. Due to the fact that all of them have the same aryl substituents (phenyl or 4-chlorophenyl) in the arylidene moiety, it is possible to compare the relevant contribution of the single-point structural modifications (type of ring or N-substitution) on their potency on the basis of their IC 50 values. Their antiproliferative activity was evaluated against the following four human adherent cancer cell lines: HeLa, A431, A2780, and MCF7. The cytotoxicity screen has revealed that the dibenzylidene dienones in general dominate the monobenzylidene enones in this respect. The nitrogen-containing heterocyclic dienones at the same time displayed higher inhibitory properties toward these human carcinoma cell lines compared with their homocyclic dienone analogs. One of the eight newly prepared 4-piperidone derivatives, N-(γ-oxobutyl)-(E,E)-3,5-bis(p-chlorobenzylidene)-4-piperidone is as potent a cell growth inhibitor (IC 50 of 0.438–1.409 μM) as the N-methyl-(E,E)-3,5-bis(p-chlorobenzylidene)-4-piperidone (IC 50 of 0.447–1.048 μM), one of the most active among the previously described compounds in this series. Catalytic hydrogen-transfer isomerization of compounds with two exocyclic benzylidene double bonds to derivatives with endocyclic double bonds results in the complete loss of antiproliferative activity. The structural modifications and 50 % inhibitory concentration (IC 50) values resulted in correlations which can promote the design of more potent derivatives of the 4-piperidone dienones.
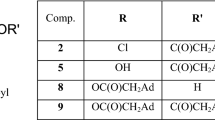
Graphical abstract



Similar content being viewed by others
References
Dimmock JR, Kandepu NM, Nazarali AJ, Kowalchuk TP, Motaganahalli N, Quail JW, Mykytiuk PA, Audette GF, Prasad L, Perjési P, Allen TM, Santos CL, Szydlowsky J, De Clerck E, Balzarini J (1999) J Med Chem 42:1358
Dimmock JR, Zello GA, Oloo EO, Quail JW, Kraatz H, Perjési P, Aradi F, Takács-Novák K, Allen TM, Santos CL, Balzarini J, De Clerck E, Stables JP (2002) J Med Chem 45:3103
Perjési P, Das U, De Clerck E, Balzarini J, Kawase M, Sakagami H, Stables JP, Lóránd T, Rózmer Zs, Dimmock JR (2008) Eur J Med Chem 43:839
Selvendiran K, Ahmed S, Dayton A, Kuppusamy ML, Tazi M, Bratasz A, Tong L, Rivera BK, Kálai T, Hideg K, Kuppusamy P (2010) Free Radic Biol Med 48:1228
Modzelewska A, Pettit C, Achanta G, Davidson NE, Huang P, Khan SR (2006) Bioorg Med Chem 14:3491
Karthikeyan NS, Sathiyanarayanan KI, Aravindan PG, Giridharan P (2011) Med Chem Res 20:81
Lagisetti P, Vilekar P, Sahoo K, Anant S, Awasthi V (2010) Bioorg Med Chem 18:6109
Jha A, Mukherjee C, Prasad AK, Parmar VS, De Clerck E, Balzarini J, Stables JP, Manavathu EK, Shrivastav A, Sharma RK, Nienaber KH, Zello GA, Dimmock JR (2007) Bioorg Med Chem 15:5854
Pati HN, Das U, Das S, Bandy B, De Clerck E, Balzarini J, Kawase M, Sakagami H, Quail JW, Stables JP, Dimmock JR (2009) Eur J Med Chem 44:54
Pati HN, Das U, Quail JW, Kawase M, Sakagami H, Dimmock JR (2008) Eur J Med Chem 43:1
Das U, Alcorn J, Shrivastav A, Sharma RK, De Clerck E, Balzarini J, Dimmock JR (2007) Eur J Med Chem 42:71
Das U, Sakagami H, Chu Q, Wang Q, Kawase M, Selvakumar P, Sharma RK, Dimmock JR (2010) Bioorg Med Chem Lett 20:912
Dimmock JR, Padmanilayam MP, Puthucode RN, Nazarali AJ, Motaganahalli NL, Zello GA, Quail JW, Oloo EO, Kraatz H, Prisciak JA, Allen TM, Santos CL, Balzarini J, De Clerck E, Manavathu E (2001) J Med Chem 44:586
Das S, Das U, Varela-Ramirez A, Lema C, Aguilera RJ, Balzarini J, De Clerck E, Dimmock SG, Gorecki DKJ, Dimmock JR (2011) ChemMedChem 6:1892
Dimmock JR, Smith LM, Smith PJ (1980) Can J Chem 58:984
Baluja G, Municio AM, Vega S (1964) Chem Ind 2053
Sun A, Lu YJ, Hu H, Shoji M, Liotta DC, Snyder JP (2009) Bioorg Med Chem Lett 19:6627
Dimmock JR, Kumar P, Chen M, Quail JW, Yang J, Allen TM, Kao GY (1995) Pharmazie 50:449
Dimmock JR, Raghavan SK, Logan BM, Bigam GE (1980) Eur J Med Chem 18:248
Mutus B, Wagner JD, Talpas CJ, Dimmock JR, Phillips OA, Reid RS (1989) Anal Biochem 177:237
Benvenuto JA, Connor TH, Monteith DK, Laidlow JA, Adams SC, Matney TS, Theiss JC (1993) J Pharm Sci 82:988
Dimmock JR, Arora VK, Wonko SL, Hamon NW, Quail JW, Jia Z, Warrington RC, Fang WD, Lee JS (1990) Drug Res Deliv 6:183
Perjési P, Földesi A, Szabó D, Zschunke A, Mak M (1987) Chem Ber 120:1449
Popkov SV, Kovalenko LV, Bobylev MM, Molchanov OY, Krimer MZ, Tashchi VP, Putsykin YG (1997) Pestic Sci 49:125
Sohár P, Csámpai A, Perjési P (2003) Arkivoc (v):114
Lóránd T, Szabó D, Földesi A, Párkányi L, Kálmán A, Neszmélyi A (1985) J Chem Soc Perkin 1:481
George H, Roth HJ (1971) Tetrahedron Lett 12:4057
Frey H, Behmann G, Kaupp G (1987) Chem Ber 120:387
Huber I, Fülöp F, Dombi Gy, Bernáth G, Hermecz I, Mészáros Z (1987) J Chem Soc Perkin 1:909
Fülöp F, Huber I, Dombi Gy, Bernáth G (1987) Tetrahedron 43:1157
Leonard NJ, Locke DM (1955) J Am Chem Soc 77:1852
Batt DG, Maynard GD, Petraitis JJ, Shaw JE, Galbraith W, Harris RR (1990) J Med Chem 33:360
Hassner A, Cromwell NH, Davis SJ (1957) J Am Chem Soc 79:230
El-Rayyes NR, Al-Jawhary A (1986) J Heterocycl Chem 23:135
Yi WB, Cai C (2005) J Fluorine Chem 126:1553
McElvain SM, Rorig K (1948) J Am Chem Soc 70:1820
Kumar N, Sun G, Ni N, Chen W, Molina ADC, Wang B, Rawat DS (2013) Chem Biol Interface 3:164
Rozmer Z, Perjési P (2013) J Planar Chromatogr 26:284
Pirger Z, Lubics A, Reglodi D, László Z, Márk L, Kiss T (2010) Neuropeptides 44:475
Brubel R, Reglodi D, Jámbor E, Koppán M, Biró Z, Kiss P, Matkovits A, Farkas J, Lubics A, Várnagy A, Bódis J, Bay C, Veszprémi B, Tamás A, Márk L (2011) J Mass Spectr 46:189
Mosmann T (1983) J Immunol Methods 65:55
ChemAxon (2012) Marvin Suite vers. 5.9.4 (http://www.chemaxon.com)
Acknowledgments
This work was supported by the University of Pécs, Faculty of Medicine Research Funds PTE ÁOK-KA-34039-12/10-11 and PTE ÁOK-KA-2013/20.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is considered to be part XI in the series of (E)-2-benzylidenebenzocyclanones. For part X, see Ref. [38].
Rights and permissions
About this article
Cite this article
Huber, I., Zupkó, I., Kovács, I.J. et al. Synthesis and antiproliferative activity of cyclic arylidene ketones: a direct comparison of monobenzylidene and dibenzylidene derivatives. Monatsh Chem 146, 973–981 (2015). https://doi.org/10.1007/s00706-015-1426-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-015-1426-7