Abstract
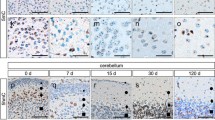
The epigenome is of fundamental importance for development and ageing. The discovery of 5-hydroxymethylcytosine (5hmC), a further base modification of cytosine beyond 5-methylcytosine, might be of high relevance in understanding the complexity of the human brain, as 5hmC is found in great extent in brain tissue. The aim of this study was to investigate the quantity of 5hmC containing nuclei by immunohistochemistry in human and murine brains at several developmental stages. We performed immunohistochemical stainings on frontal cortex, white matter and cerebellar cortex of 15 healthy controls. Three cases each were assigned to five age groups (foetus, adolescent, adult, elderly, aged). Additionally, cortex and cerebellum of 15 mice sacrificed between day 0 and 120 after birth were investigated. We found marked alterations of 5hmC amount during ageing. In human cortex there was an increase of 5hmC of 50 %, in white matter we found an increase of even 200 % during ageing. In the cerebellum both internal granular cell layer and molecular cell layer showed a significant increase of 5hmC till adulthood. Purkinje cell nuclei showed constantly positive signals for 5hmC. These data were paralleled in murine brains. Co-labelling of 5hmC and markers for mature and immature cells in murine cerebellar cortex at the age of 7 days revealed that 5hmC was found in mature but not in immature cells. In conclusion, the findings described in this study emphasise the importance of 5hmC in brain development and ageing and will help to better understand the complexity and plasticity of the brain.




Similar content being viewed by others
References
Bradley-Whitman MA, Lovell MA (2013) Epigenetic changes in the progression of Alzheimer’s disease. Mech Ageing Dev 134:486–495. doi:10.1016/j.mad.2013.08.005
Branco MR, Ficz G, Reik W (2012) Uncovering the role of 5-hydroxymethylcytosine in the epigenome. Nat Rev Genet 13:7–13. doi:10.1038/nrg3080
Clancy B, Kersh B, Hyde J, Darlington RB, Anand KJ, Finlay BL (2007) Web-based method for translating neurodevelopment from laboratory species to humans. Neuroinformatics 5:79–94
Condliffe D et al (2014) Cross-region reduction in 5-hydroxymethylcytosine in Alzheimer’s disease brain. Neurobiol Aging 35:1850–1854. doi:10.1016/j.neurobiolaging.2014.02.002
Ficz G et al (2011) Dynamic regulation of 5-hydroxymethylcytosine in mouse ES cells and during differentiation. Nature 473:398–402. doi:10.1038/nature10008
He YF et al (2011) Tet-mediated formation of 5-carboxylcytosine and its excision by TDG in mammalian DNA. Science 333:1303–1307. doi:10.1126/science.1210944
Ito S, D’Alessio AC, Taranova OV, Hong K, Sowers LC, Zhang Y (2010) Role of Tet proteins in 5mC to 5hmC conversion. ES-cell self-renewal and inner cell mass specification Nature 466:1129–1133. doi:10.1038/nature09303
Ito S et al (2011) Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science 333:1300–1303. doi:10.1126/science.1210597
James SJ, Shpyleva S, Melnyk S, Pavliv O, Pogribny IP (2014) Elevated 5-hydroxymethylcytosine in the Engrailed-2 (EN-2) promoter is associated with increased gene expression and decreased MeCP2 binding in autism cerebellum. Transl Psychiatry 4:e460. doi:10.1038/tp.2014.87
Jin SG, Wu X, Li AX, Pfeifer GP (2011) Genomic mapping of 5-hydroxymethylcytosine in the human brain. Nucleic Acids Res 39:5015–5024. doi:10.1093/nar/gkr120
Kraus TF et al (2012) Low values of 5-hydroxymethylcytosine (5hmC), the “sixth base,” are associated with anaplasia in human brain tumors. Int J Cancer 131:1577–1590. doi:10.1002/ijc.27429
Kriaucionis S, Heintz N (2009) The nuclear DNA base 5-hydroxymethylcytosine is present in Purkinje neurons and the brain. Science 324:929–930. doi:10.1126/science.1169786
Mellen M, Ayata P, Dewell S, Kriaucionis S, Heintz N (2012) MeCP2 binds to 5hmC enriched within active genes and accessible chromatin in the nervous system. Cell 151:1417–1430. doi:10.1016/j.cell.2012.11.022
Munzel M, Globisch D, Carell T (2011) 5-Hydroxymethylcytosine, the sixth base of the genome. Angew Chem 50:6460–6468. doi:10.1002/anie.201101547
Pelvig DP, Pakkenberg H, Stark AK, Pakkenberg B (2008) Neocortical glial cell numbers in human brains. Neurobiol Aging 29:1754–1762. doi:10.1016/j.neurobiolaging.2007.04.013
Pfaffeneder T et al (2011) The discovery of 5-formylcytosine in embryonic stem cell DNA. Angew Chem 50:7008–7012. doi:10.1002/anie.201103899
Spruijt CG et al (2013) Dynamic readers for 5-(hydroxy)methylcytosine and its oxidized derivatives. Cell 152:1146–1159. doi:10.1016/j.cell.2013.02.004
Szulwach KE et al (2011a) Integrating 5-hydroxymethylcytosine into the epigenomic landscape of human embryonic stem cells. PLoS Genet 7:e1002154. doi:10.1371/journal.pgen.1002154
Szulwach KE et al (2011b) 5-hmC-mediated epigenetic dynamics during postnatal neurodevelopment and aging. Nat Neurosci 14:1607–1616. doi:10.1038/nn.2959
Tahiliani M et al (2009) Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by MLL partner TET1. Science 324:930–935. doi:10.1126/science.1170116
van den Hove DL, Chouliaras L, Rutten BP (2012) The role of 5-hydroxymethylcytosine in aging and Alzheimer’s disease: current status and prospects for future studies. Curr Alzheimer Res 9:545–549
Villar-Menendez I et al (2013) Increased 5-methylcytosine and decreased 5-hydroxymethylcytosine levels are associated with reduced striatal A2AR levels in Huntington’s disease. Neuromolecular Med 15:295–309. doi:10.1007/s12017-013-8219-0
Wang F et al (2013) Genome-wide loss of 5-hmC is a novel epigenetic feature of Huntington’s disease. Hum Mol Genet 22:3641–3653. doi:10.1093/hmg/ddt214
Zhubi A, Chen Y, Dong E, Cook EH, Guidotti A, Grayson DR (2014) Increased binding of MeCP2 to the GAD1 and RELN promoters may be mediated by an enrichment of 5-hmC in autism spectrum disorder (ASD) cerebellum. Transl Psychiatry 4:e349. doi:10.1038/tp.2013.123
Acknowledgments
We thank Dr. Thomas Arzberger, Department for Psychiatry and Psychotherapy/Centre for Neuropathology and Prion Research, Ludwig-Maximilians-University Munich, Germany for critically reading the manuscript.
Conflict of interest
There is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kraus, T.F.J., Guibourt, V. & Kretzschmar, H.A. 5-Hydroxymethylcytosine, the “Sixth Base”, during brain development and ageing. J Neural Transm 122, 1035–1043 (2015). https://doi.org/10.1007/s00702-014-1346-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-014-1346-4