Abstract
Background
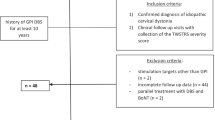
Dystonia has been treated well using deep brain stimulation at the globus pallidus internus (GPi DBS). Dystonia can be categorized as two basic types of movement, phasic-type and tonic-type. Cervical dystonia is the most common type of focal dystonia, and sequential differences in clinical outcomes between phasic-type and tonic-type cervical dystonia have not been reported.
Methods
This study included a retrospective cohort of 30 patients with primary cervical dystonia who underwent GPi DBS. Age, disease duration, dystonia direction, movement types, employment status, relevant life events, and neuropsychological examinations were analyzed with respect to clinical outcomes following GPi DBS.
Results
The only significant factor affecting clinical outcomes was movement type (phasic or tonic). Sequential changes in clinical outcomes showed significant differences between phasic- and tonic-type cervical dystonia. A delayed benefit was found in both phasic- and tonic-type dystonia.
Conclusions
The clinical outcome of phasic-type cervical dystonia is more favorable than that of tonic-type cervical dystonia following GPi DBS.




Similar content being viewed by others
References
Argyelan M, Carbon M, Niethammer M, Ulug AM, Voss HU, Bressman SB, Dhawan V, Eidelberg D (2009) Cerebellothalamocortical connectivity regulates penetrance in dystonia. J Neurosci 29:9740–9747
Barow E, Neumann WJ, Brucke C, Huebl J, Horn A, Brown P, Krauss JK, Schneider GH, Kuhn AA (2014) Deep brain stimulation suppresses pallidal low-frequency activity in patients with phasic dystonic movements. Brain 137:3012–3024
Brown P, Oliviero A, Mazzone P, Insola A, Tonali P, Di Lazzaro V (2001) Dopamine dependency of oscillations between subthalamic nucleus and pallidum in Parkinson’s disease. J Neurosci 21:1033–1038
Byl NN, Merzenich MM, Jenkins WM (1996) A primate genesis model of focal dystonia and repetitive strain injury: I learning-induced dedifferentiation of the representation of the hand in the primary somatosensory cortex in adult monkeys. Neurology 47:508–520
Chiken S, Nambu A (2014) Disrupting neuronal transmission: mechanism of DBS? Front Syst Neurosci 8:33
Chung M, Han I, Chung SS, Jang DK, Huh R (2015) Effectiveness of selective peripheral denervation in combination with pallidal deep brain stimulation for the treatment of cervical dystonia. Acta Neurochir (Wien) 157:435–442
Comella C, Bhatia K (2015) An international survey of patients with cervical dystonia. J Neurol 262:837–848
de Hemptinne C, Ryapolova-Webb ES, Air EL, Garcia PA, Miller KJ, Ojemann JG, Ostrem JL, Galifianakis NB, Starr PA (2013) Exaggerated phase-amplitude coupling in the primary motor cortex in Parkinson disease. Proc Natl Acad Sci U S A 110:4780–4785
Eltahawy HA, Saint-Cyr J, Poon YY, Moro E, Lang AE, Lozano AM (2004) Pallidal deep brain stimulation in cervical dystonia: clinical outcome in four cases. Can J Neurol Sci 31:328–332
Frucht SJ (2014) “Closing the loop” in cervical dystonia: a new clinical phenomenon. Tremor Other Hyperkinet Mov (N Y) 4
Grips E, Blahak C, Capelle HH, Bazner H, Weigel R, Sedlaczek O, Krauss JK, Wohrle JC (2007) Patterns of reoccurrence of segmental dystonia after discontinuation of deep brain stimulation. J Neurol Neurosurg Psychiatry 78:318–320
Hamani C, Moro E, Zadikoff C, Poon YY, Lozano AM (2008) Location of active contacts in patients with primary dystonia treated with globus pallidus deep brain stimulation. Neurosurgery 62:217–225
Hashimoto T, Elder CM, Okun MS, Patrick SK, Vitek JL (2003) Stimulation of the subthalamic nucleus changes the firing pattern of pallidal neurons. J Neurosci 23:1916–1923
Hirschmann J, Hartmann CJ, Butz M, Hoogenboom N, Ozkurt TE, Elben S, Vesper J, Wojtecki L, Schnitzler A (2013) A direct relationship between oscillatory subthalamic nucleus-cortex coupling and rest tremor in Parkinson’s disease. Brain 136:3659–3670
Huh R, Han IB, Chung M, Chung S (2010) Comparison of treatment results between selective peripheral denervation and deep brain stimulation in patients with cervical dystonia. Stereotact Funct Neurosurg 88:234–238
Hung SW, Hamani C, Lozano AM, Poon YY, Piboolnurak P, Miyasaki JM, Lang AE, Dostrovsky JO, Hutchison WD, Moro E (2007) Long-term outcome of bilateral pallidal deep brain stimulation for primary cervical dystonia. Neurology 68:457–459
Isaias IU, Volkmann J, Kupsch A, Burgunder JM, Ostrem JL, Alterman RL, Mehdorn HM, Schonecker T, Krauss JK, Starr P, Reese R, Kuhn AA, Schupbach WM, Tagliati M (2011) Factors predicting protracted improvement after pallidal DBS for primary dystonia: the role of age and disease duration. J Neurol 258:1469–1476
Jankovic J (2009) Treatment of hyperkinetic movement disorders. Lancet Neurol 8:844–856
Jankovic J, Tsui J, Bergeron C (2007) Prevalence of cervical dystonia and spasmodic torticollis in the United States general population. Parkinsonism Relat Disord 13:411–416
Johnson MD, Miocinovic S, McIntyre CC, Vitek JL (2008) Mechanisms and targets of deep brain stimulation in movement disorders. Neurotherapeutics 5:294–308
Karas PJ, Mikell CB, Christian E, Liker MA, Sheth SA (2013) Deep brain stimulation: a mechanistic and clinical update. Neurosurg Focus 35, E1
Kiss ZH, Doig-Beyaert K, Eliasziw M, Tsui J, Haffenden A, Suchowersky O (2007) The Canadian multicentre study of deep brain stimulation for cervical dystonia. Brain 130:2879–2886
Koch G, Porcacchia P, Ponzo V, Carrillo F, Caceres-Redondo MT, Brusa L, Desiato MT, Arciprete F, Di Lorenzo F, Pisani A, Caltagirone C, Palomar FJ, Mir P (2014) Effects of two weeks of cerebellar theta burst stimulation in cervical dystonia patients. Brain Stimul 7:564–572
Krauss JK (2010) Surgical treatment of dystonia. Eur J Neurol 17(Suppl 1):97–101
Krauss JK, Yianni J, Loher TJ, Aziz TZ (2004) Deep brain stimulation for dystonia. J Clin Neurophysiol 21:18–30
Kupsch A, Benecke R, Muller J, Trottenberg T, Schneider GH, Poewe W, Eisner W, Wolters A, Muller JU, Deuschl G, Pinsker MO, Skogseid IM, Roeste GK, Vollmer-Haase J, Brentrup A, Krause M, Tronnier V, Schnitzler A, Voges J, Nikkhah G, Vesper J, Naumann M, Volkmann J (2006) Pallidal deep-brain stimulation in primary generalized or segmental dystonia. N Engl J Med 355:1978–1990
Li Q, Qian ZM, Arbuthnott GW, Ke Y, Yung WH (2014) Cortical effects of deep brain stimulation: implications for pathogenesis and treatment of Parkinson disease. JAMA Neurol 71:100–103
Liu X, Griffin IC, Parkin SG, Miall RC, Rowe JG, Gregory RP, Scott RB, Aziz TZ, Stein JF (2002) Involvement of the medial pallidum in focal myoclonic dystonia: a clinical and neurophysiological case study. Mov Disord 17:346–353
Liu X, Wang S, Yianni J, Nandi D, Bain PG, Gregory R, Stein JF, Aziz TZ (2008) The sensory and motor representation of synchronized oscillations in the globus pallidus in patients with primary dystonia. Brain 131:1562–1573
Miocinovic S, Somayajula S, Chitnis S, Vitek JL (2013) History, applications, and mechanisms of deep brain stimulation. JAMA Neurol 70:163–171
Moro E, Piboolnurak P, Arenovich T, Hung SW, Poon YY, Lozano AM (2009) Pallidal stimulation in cervical dystonia: clinical implications of acute changes in stimulation parameters. Eur J Neurol 16:506–512
Munts AG, Koehler PJ (2010) How psychogenic is dystonia? views from past to present. Brain 133:1552–1564
Murrow RW (2014) Penfield’s prediction: a mechanism for deep brain stimulation. Front Neurol 5:213
Perlmutter JS, Mink JW, Bastian AJ, Zackowski K, Hershey T, Miyawaki E, Koller W, Videen TO (2002) Blood flow responses to deep brain stimulation of thalamus. Neurology 58:1388–1394
Quartarone A, Hallett M (2013) Emerging concepts in the physiological basis of dystonia. Mov Disord 28:958–967
Quartarone A, Pisani A (2011) Abnormal plasticity in dystonia: disruption of synaptic homeostasis. Neurobiol Dis 42:162–170
Richardson SP (2015) Enhanced dorsal premotor-motor inhibition in cervical dystonia. Clin Neurophysiol 126:1387–1391
Sadnicka A, Kimmich O, Pisarek C, Ruge D, Galea J, Kassavetis P, Parees I, Saifee T, Molloy A, Bradley D, O’Riordan S, Zrinzo L, Hariz M, Bhatia KP, Limousin P, Foltynie T, Rothwell JC, Hutchinson M, Edwards MJ (2013) Pallidal stimulation for cervical dystonia does not correct abnormal temporal discrimination. Mov Disord 28:1874–1877
Sako W, Morigaki R, Mizobuchi Y, Tsuzuki T, Ima H, Ushio Y, Nagahiro S, Kaji R, Goto S (2011) Bilateral pallidal deep brain stimulation in primary Meige syndrome. Parkinsonism Relat Disord 17:123–125
Shaikh AG, Mewes K, Jinnah HA, DeLong MR, Gross RE, Triche S, Freeman A, Factor SA (2014) Globus pallidus deep brain stimulation for adult-onset axial dystonia. Parkinsonism Relat Disord 20:1279–1282
Sharott A, Grosse P, Kuhn AA, Salih F, Engel AK, Kupsch A, Schneider GH, Krauss JK, Brown P (2008) Is the synchronization between pallidal and muscle activity in primary dystonia due to peripheral afferance or a motor drive? Brain 131:473–484
Shimamoto SA, Ryapolova-Webb ES, Ostrem JL, Galifianakis NB, Miller KJ, Starr PA (2013) Subthalamic nucleus neurons are synchronized to primary motor cortex local field potentials in Parkinson’s disease. J Neurosci 33:7220–7233
Silberstein P, Kuhn AA, Kupsch A, Trottenberg T, Krauss JK, Wohrle JC, Mazzone P, Insola A, Di Lazzaro V, Oliviero A, Aziz T, Brown P (2003) Patterning of globus pallidus local field potentials differs between Parkinson’s disease and dystonia. Brain 126:2597–2608
Svetel M, Pekmezovic T, Jovic J, Ivanovic N, Dragasevic N, Maric J, Kostic VS (2007) Spread of primary dystonia in relation to initially affected region. J Neurol 254:879–883
Svetel M, Pekmezovic T, Tomic A, Kresojevic N, Kostic VS (2015) The spread of primary late-onset focal dystonia in a long-term follow-up study. Clin Neurol Neurosurg 132:41–43
Tolleson C, Pallavaram S, Li C, Fang J, Phibbs F, Konrad P, Hedera P, D’Haese PF, Dawant BM, Davis TL (2015) The optimal pallidal target in deep brain stimulation for dystonia: a study using a functional atlas based on nonlinear image registration. Stereotact Funct Neurosurg 93:17–24
Vidailhet M, Grabli D, Roze E (2009) Pathophysiology of dystonia. Curr Opin Neurol 22:406–413
Vidailhet M, Vercueil L, Houeto JL, Krystkowiak P, Benabid AL, Cornu P, Lagrange C, Tezenas du Montcel S, Dormont D, Grand S, Blond S, Detante O, Pillon B, Ardouin C, Agid Y, Destee A, Pollak P (2005) Bilateral deep-brain stimulation of the globus pallidus in primary generalized dystonia. N Engl J Med 352:459–467
Volkmann J, Mueller J, Deuschl G, Kuhn AA, Krauss JK, Poewe W, Timmermann L, Falk D, Kupsch A, Kivi A, Schneider GH, Schnitzler A, Sudmeyer M, Voges J, Wolters A, Wittstock M, Muller JU, Hering S, Eisner W, Vesper J, Prokop T, Pinsker M, Schrader C, Kloss M, Kiening K, Boetzel K, Mehrkens J, Skogseid IM, Ramm-Pettersen J, Kemmler G, Bhatia KP, Vitek JL, Benecke R (2014) Pallidal neurostimulation in patients with medication-refractory cervical dystonia: a randomised, sham-controlled trial. Lancet Neurol 13:875–884
Walsh RA, Sidiropoulos C, Lozano AM, Hodaie M, Poon YY, Fallis M, Moro E (2013) Bilateral pallidal stimulation in cervical dystonia: blinded evidence of benefit beyond 5 years. Brain 136:761–769
Weinberger M, Hutchison WD, Alavi M, Hodaie M, Lozano AM, Moro E, Dostrovsky JO (2012) Oscillatory activity in the globus pallidus internus: comparison between Parkinson’s disease and dystonia. Clin Neurophysiol 123:358–368
Witt JL, Moro E, Ash RS, Hamani C, Starr PA, Lozano AM, Hodaie M, Poon YY, Markun LC, Ostrem JL (2013) Predictive factors of outcome in primary cervical dystonia following pallidal deep brain stimulation. Mov Disord 28:1451–1455
Yianni J, Bain PG, Gregory RP, Nandi D, Joint C, Scott RB, Stein JF, Aziz TZ (2003) Post-operative progress of dystonia patients following globus pallidus internus deep brain stimulation. Eur J Neurol 10:239–247
Yianni J, Wang SY, Liu X, Bain PG, Nandi D, Gregory R, Joint C, Stein JF, Aziz TZ (2006) A dominant bursting electromyograph pattern in dystonic conditions predicts an early response to pallidal stimulation. J Clin Neurosci 13:738–746
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Financial disclosures
Moonyoung Chung has no financial disclosure.
Ryoong Huh has no financial disclosure.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Additional information
Comments
In this interesting study, the authors retrospectively review the clinical data of 30 patients affected by primary cervical dystonia who underwent globus pallidus internus (GPi) deep brain stimulation (DBS). This study provides some important information on this topic. The only significant factor affecting clinical outcomes was the dystonia type (phasic or tonic). A benefit was actually found for both types of cervical dystonia, but the clinical outcome of phasic-type cervical dystonia is more favorable. Intriguingly, a delayed benefit was found in both phasic- and tonic-type dystonia. This study has the merit of being conducted in a large and homogenous patient group and to demonstrate the persistence of benefit and even a delayed improvement in both dystonia types. Furthermore, it provides interesting considerations about the mechanisms underlying DBS neuromodulation. It has been suggested that DBS causes a “depolarization block” by high-frequency stimulation of neurons above their maximum firing rate inducing continuous depolarization and conduction block. Nevertheless, this interpretation does not appear completely satisfactory. Indeed, several stimulation-induced side effects, such as visual flushing, tetanic muscle contraction, and paresthesia, seemingly originates from neural activations rather than from neural block. Other possible explanations should therefore be considered. For instance, the effects of DBS on phasic-types dystonia could be the result of desynchronization of a pathologic low-frequency oscillatory activity of the central motor network. Furthermore, the distinctive feature of a delayed benefit of GPi DBS in dystonia patients suggests that this procedure can modulate neural plasticity in a manner that deserves further studies.
Alfredo Conti
Messina, Italy
Some of data of this study have been presented in 55th congress of Korean Society of Neurosurgeon at Oct. 17, 2015.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 23 kb)
Rights and permissions
About this article
Cite this article
Chung, M., Huh, R. Different clinical course of pallidal deep brain stimulation for phasic- and tonic-type cervical dystonia. Acta Neurochir 158, 171–180 (2016). https://doi.org/10.1007/s00701-015-2646-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-015-2646-7