Abstract
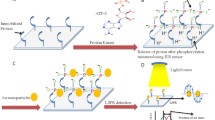
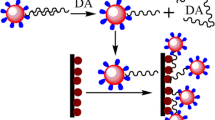
We report on an electrochemical biosensor for the determination of the activity of dipeptidyl peptidase-IV (DPP-IV), and on a method for screening the effect of its inhibitors. An enzyme substrate (Fc-peptide) was immobilized on the surface of a gold electrode, and double signal amplification was accomplished via an additional layer consisting of phenyl rings and gold nanoparticles. The activity of DPP-IV was determined at levels as low as 39 nU·mL−1 and over a linear detection range as wide as from 0.5 μU·mL−1 to 2.5 mU·mL−1. The inhibitory effects of diprotin A and the His-Leu dipeptide on the activity of DPP-IV also were tested and gave IC50 values of 93.5 and 95.5 μM, respectively. The assay is rapid, precise and selective. It may be extended to other peptidases and, possibly, proteases and their inhibitors.

An electrochemical biosensor for the determination of dipeptidyl peptidase-IV activity and the screening of its inhibitors was established. The substrate was immobilized on a gold electrode surface, and double signal amplification was accomplished via phenyl ring layer and gold nanoparticles.





Similar content being viewed by others
References
Juillerat-Jeanneret L (2014) Dipeptidyl peptidase IV and its inhibitors: therapeutics for type 2 diabetes and what else? J Med Chem 57(6):2197–2212. doi:10.1021/jm400658e
Shimodaira M, Muroya Y, Kumagai N, Tsuzawa K, Honda K (2013) Effects of short-term intensive glycemic control on insulin, glucagon, and glucagon-like peptide-1 secretion in patients with Type 2 diabetes. J Endocrinol Investig 36(9):734–738. doi:10.3275/8934
Holz G (2004) New insights concerning the glucose-dependent insulin secretagogue action of glucagon-like peptide-1 in pancreatic beta-cells. Horm Metab Res 36(11–12):787–794. doi:10.1055/s-2004-826165
Durinx C, Lambeir AM, Bosmans E, Falmagne JB, Berghmans R, Haemers A, Scharpe S, De Meester I (2000) Molecular characterization of dipeptidyl peptidase activity in serum–soluble CD26/dipeptidyl peptidase IV is responsible for the release of X-Pro dipeptides. Eur J Biochem 267(17):5608–5613. doi:10.1046/j.1432-1327.2000.01634.x
Shane R, Wilk S, Bodnar RJ (1999) Modulation of endomorphin-2-induced analgesia by dipeptidyl peptidase IV. Brain Res 815(2):278–286. doi:10.1016/s0006-8993(98)01121-4
Aertgeerts K, Ye S, Shi LH, Prasad SG, Witmer D, Chi E, Sang BC, Wijnands RA, Webb DR, Swanson RV (2004) N-linked glycosylation of dipeptidyl peptidase IV (CD26): effects on enzyme activity, homodimer formation, and adenosine deaminase binding. Protein Sci 13(1):145–154. doi:10.1110/ps.03352504
Hama T, Okada M, Kojima K, Kato T, Matsuyama M, Nagatsu T (1982) Purification of dipeptidyl-aminopeptidase IV from human kidney by anti dipeptidyl-aminopeptidase IV affinity chromatography. Mol Cell Biochem 43(1):35–42
Kim YB, Kopcho LM, Kirby MS, Hamann LG, Weigelt CA, Metzler WJ, Marcinkeviciene J (2006) Mechanism of Gly-Pro-pNA cleavage catalyzed by dipeptidyl peptidase-IV and its inhibition by saxagliptin (BMS-477118). Arch Biochem Biophys 445(1):9–18. doi:10.1016/j.abb.2005.11.010
Swisher LZ, Syed LU, Prior AM, Madiyar FR, Carlson KR, Nguyen TA, Hua DH, Li J (2013) Electrochemical protease biosensor based on enhanced AC voltammetry using carbon nanofiber nanoelectrode arrays. J Phys Chem C 117(8):4268–4277. doi:10.1021/jp312031u
Zhang Y, Guo G, Qian Q, Cui D (2012) Chloroplasts-mediated biosynthesis of nanoscale Au-Ag alloy for 2-butanone assay based on electrochemical sensor. Nanoscale Res Lett 7:475–478. doi:10.1186/1556-276x-7-475
Syed LU, Liu J, Prior AM, Hua DH, Li J (2011) Enhanced electron transfer rates by AC voltammetry for ferrocenes attached to the end of embedded carbon nanofiber nanoelectrode arrays. Electroanalysis 23(7):1709–1717. doi:10.1002/elan.201100088
Mahmoud KA, Hrapovic S, Luong JHT (2008) Picomolar detection of protease using peptide/single walled carbon nanotube/gold nanoparticle-modified electrode. ACS Nano 2(5):1051–1057. doi:10.1021/nn8000774
Rónai AZ, Király K, Szebeni A, Szemenyei E, Prohászka Z, Darula Z, Tóth G, Till I, Szalay B, Kató E, Barna I (2009) Immunoreactive endomorphin 2 is generated extracellularly in rat isolated L4,5 dorsal root ganglia by DPP-IV. Regul Pept 157(1–3):1–2. doi:10.1016/j.regpep.2009.06.006
Corgier BP, Marquette CA, Blum LJ (2005) Diazonium-protein adducts for graphite electrode microarrays modification: direct and addressed electrochemical immobilization. J Am Chem Soc 127(51):18328–18332. doi:10.1021/ja056946w
Olejnik P, Palys B, Kowalczyk A, Nowicka AM (2012) Orientation of laccase on charged surfaces. Mediatorless oxygen reduction on amino- and carboxyl-ended ethylphenyl groups. J Phys Chem 116(49):25911–25918. doi:10.1021/jp3098654
Gutiérrez-Sánchez C, Pita M, Vaz-Domínguez C, Shleev S, De Lacey AL (2012) Gold nanoparticles as electronic bridges for laccase-based biocathodes. J Am Chem Soc 134(41):17212–17220. doi:10.1021/ja307308j
Zhang Y, Zheng J, Gao G, Kong Y, Zhi X, Wang K, Zhang X, Cui D (2011) Biosynthesis of gold nanoparticles using chloroplasts. Int J Nanomedicine 6:2899–2906. doi:10.2147/ijn.s24785
Kim JH, Kim JW, Chung BH (2011) Enzymatic tailoring for precise control of plasmonic resonance absorbance of gold nanoparticle assemblies. J Colloid Interface Sci 360(2):335–340. doi:10.1016/j.jcis.2011.05.008
Lyskawa J, Belanger D (2006) Direct modification of a gold electrode with aminophenyl groups by electrochemical reduction of in situ generated aminophenyl monodiazonium cations. Chem Mater 18(20):4755–4763. doi:10.1021/cm060752d
Chira A, Covaci OI, Radu GL (2012) A comparative study of gold electrodes modification methods with aromatic compounds based on diazonium and thiol chemistry. Sci Bull B Chem Mater Sci 74(1):183–192
Mallesha M, Manjunatha R, Nethravathi C, Suresh GS, Rajamathi M, Melo JS, Venkatesha TV (2011) Functionalized-graphene modified graphite electrode for the selective determination of dopamine in presence of uric acid and ascorbic acid. Bioelectrochemistry 81(2):104–108. doi:10.1016/j.bioelechem.2011.03.004
Kang X, Mai Z, Zou X, Cai P, Mo J (2007) A sensitive nonenzymatic glucose sensor in alkaline media with a copper nanocluster/multiwall carbon nano tube-modified glassy carbon electrode. Anal Biochem 363(1):143–150. doi:10.1016/j.ab.2007.01.003
Deng C, Chen J, Nie L, Nie Z, Yao S (2009) Sensitive bifunctional aptamer-based electrochemical biosensor for small molecules and protein. Anal Chem 81(24):9972–9978. doi:10.1021/ac901727z
Hu C, Yang DP, Wang Z, Yu L, Zhang J, Jia N (2013) Improved EIS performance of an electrochemical cytosensor using three-dimensional architecture Au@BSA as sensing layer. Anal Chem 85(10):5200–5206. doi:10.1021/ac400556q
Frasconi M, Mazzei F (2012) Electrochemically controlled assembly and logic gates operations of gold nanoparticle arrays. Langmuir 28(6):3322–3331. doi:10.1021/la203985n
Song MJ, Lee SK, Lee JY, Kim JH, Lim DS (2012) Electrochemical sensor based on Au nanoparticles decorated boron-doped diamond electrode using ferrocene-tagged aptamer for proton detection. J Electroanal Chem 677–680:139–144. doi:10.1016/j.jelechem.2012.05.019
Liu G, Wang J, Wunschel DS, Lin Y (2006) Electrochemical proteolytic beacon for detection of matrix metalloproteinase activities. J Am Chem Soc 128(38):12382–12383. doi:10.1021/ja0626638
Miao L, Lei JY, Jin J, Xu ZH (2009) Establishment and application of screening method for DPP IV inhibitors in vito. Chin Pharmacol Bull 25(3):411–414
Jiang HX, Lv JX, Cao Y, Pang JX (2012) Construction of screening model for dipeptidyl peptidase IV in vitro and active inhibitory estimation of related compounds. Chin J Exp Tradit Med Form 18:210–214
Cao Y, Wang J, Xu Y, Li G (2010) Combination of enzyme catalysis and electrocatalysis for biosensor fabrication: application to assay the activity of indoleamine 2,3-dioxygensae. Biosens Bioelectron 26(1):87–91. doi:10.1016/j.bios.2010.05.019
Hu C, Yang DP, Wang Z, Huang P, Wang X, Chen D, Cui D, Yang M, Jia N (2013) Bio-mimetically synthesized Ag@BSA microspheres as a novel electrochemical biosensing interface for sensitive detection of tumor cells. Biosens Bioelectron 41:656–662. doi:10.1016/j.bios.2012.09.035
Bharti SK, Krishnan S, Kumar A, Rajak KK, Murari K, Bharti BK, Gupta AK (2012) Antihyperglycemic activity with DPP-IV inhibition of alkaloids from seed extract of Castanospermum australe: investigation by experimental validation and molecular docking. Phytomedicine 20(1):24–31. doi:10.1016/j.phymed.2012.09.009
Nongonierma AB, FitzGerald RJ (2013) Dipeptidyl peptidase IV inhibitory and antioxidative properties of milk protein-derived dipeptides and hydrolysates. Peptides 39:157–163. doi:10.1016/j.peptides.2012.11.016
Acknowledgments
This work is supported by the National Natural Science Foundation of China (Grant Nos. 31101354 and 21235003).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 137 kb)
Rights and permissions
About this article
Cite this article
Zhang, J., Liu, Y., Lv, J. et al. Dipeptidyl peptidase-IV activity assay and inhibitor screening using a gold nanoparticle-modified gold electrode with an immobilized enzyme substrate. Microchim Acta 182, 281–288 (2015). https://doi.org/10.1007/s00604-014-1329-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-014-1329-z