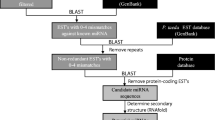
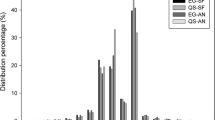
Abstract
MicroRNAs (miRNAs) are small RNAs that regulate genes involved in various aspects of plant development, but their presence and expression patterns in the male gametophytes of gymnosperms have not yet been established. Therefore, this study identified and compared the expression patterns of conserved miRNAs from two stages of the male gametophyte of loblolly pine (Pinus taeda), which are the mature (ungerminated) and germinated pollen. Microarray was used to identify conserved miRNAs that varied in expression between these two stages of the loblolly pine male gametophyte. Forty-seven conserved miRNAs showed significantly different expression levels between mature and germinated loblolly pine pollen. In particular, miRNAs representing 14 and 8 families were up- and down-regulated in germinated loblolly pine pollen, respectively. qRT-PCR was used to validate their expression patterns using representative miRNAs. Target genes and proteins were identified using psRNATarget program. Predicted targets of the 22 miRNA families belong mostly to classes of genes involved in defense/stress response, metabolism, regulation, and signaling. qRT-PCR was also used to validate the expression patterns of representative target genes. This study shows that conserved miRNAs are expressed in mature and germinated loblolly pine pollen. Many of these miRNAs are differentially expressed, which indicates that the two stages of the male gametophyte examined are regulated at the miRNA level. This study also expands our knowledge of the male gametophytes of seed plants by providing insights on some similarities and differences in the types and expression patterns of conserved miRNAs between loblolly pine with those of rice and Arabidopsis.

Similar content being viewed by others
References
Arazi T, Talmor-Neiman M, Stav R, Riese M, Huijser P, Baulcombe DC (2005) Cloning and characterization of microRNAs from moss. Plant J 43:837–848
Axtell MJ, Bartel DP (2005) Antiquity of microRNAs and their targets in land plants. Plant Cell 17:1658–1673
Borges F, Pereira PA, Slotkin RK, Martienssen RA, Becker JD (2011) MicroRNA activity in the Arabidopsis male gametophyte. J Exp Bot 62:1611–1620
Brewbaker JL, Kwack BH (1963) The essential role of calcium ion in pollen germination and pollen tube growth. Am J Bot 50:859–865
Brodersen P, Sakvarelidze-Achard L, Bruun-Rasmussen M, Dunoyer P, Yamamoto YY, Sieburth L, Voinnet O (2008) Widespread translational inhibition by plant miRNAs and siRNAs. Science 320:1185–1190
Carthew RW, Sontheimer EJ (2009) Origins and mechanisms of miRNAs and siRNAs. Cell 136:642–655
Chambers C, Shuai B (2009) Profiling microRNA expression in Arabidopsis pollen using microRNA array and real-time PCR. BMC Plant Biol 9:87
Chen CC (2009) Small RNAs and their roles in plant development. Annu Rev Cell Biol 25:21–44
Chen Y, Chen T, Shen S, Zheng M, Guo Y, Lin J, Baluska F, Samai J (2006) Differential display proteomic analysis of Picea meyeri pollen germination and pollen tube growth after inhibition of actin polymerization by latrunculin B. Plant J 47:174–195
Dai X, Zhao PX (2011) psRNATarget: a plant small RNA target analysis server. Nucleic Acids Res 39:W155–W159
Dai S, Chen T, Chong K, Xue Y, Liu S, Wang T (2007) Proteomics identification of differentially expressed proteins associated with pollen germination and tube growth reveals characteristics of germinated Oryza sativa pollen. Mol Cell Proteomics 6:207–230
Dolgosheina EV, Morin RD, Aksay G, Sahinalp SC, Magrini V, Mardis ER, Mattsson J, Unrau PJ (2008) Conifers have a unique small RNA silencing signature. RNA 14:1508–1515
Fernando DD, Owens JN, von Aderkas P, Takaso T (1997) In vitro pollen tube growth and penetration of female gametophyte in Douglas fir (Pseudotsuga menziesii). Sex Plant Reprod 10:209–216
Fernando DD, Quinn CR, Brenner ED, Owens JN (2010) Male gametophyte development and evolution in extant gymnosperms. Int J Plant Dev Bio 4:47–63
Floyd SK, Bowman JL (2004) Gene regulation: ancient microRNA target sequences in plants. Nature 428:485–486
Fujioka T, Kaneko F, Kazama T, Suwabe K, Suzuki G, Makino A, Mae T, Endo M, Kawagishi-Kobayashi M, Watanabe M (2008) Identification of small RNAs in late developmental stage of rice anthers. Genes Genet Syst 83:281–284
Gifford EM, Foster AS (1989) Morphology and evolution of vascular plants. W. H. Freedman and Company, New York
Grant-Downton R (2010) Through a generation darkly: small RNAs in the gametophyte. Biochem Soc Trans 38:617–621
Grant-Downton R, Le Trionnaire G, Schmid R, Rodriguez-Enriquez J, Hafidh S, Mehdi S, Twell D, Dickinson H (2009) MicroRNA and tasiRNA diversity in mature pollen of Arabidopsis thaliana. BMC Genomics 10:643
Jones-Rhoades MW, Bartel DP, Bartel B (2006) MicroRNAs and their regulatory roles in plants. Annu Rev Plant Biol 57:19–53
Kidner CA, Martienssen RA (2005) The developmental role of microRNA in plants. Curr Opin Plant Biol 8:38–44
Le Trionnaire G, Twell D (2010) Small RNAs in angiosperm gametophytes: from epigenetics to gamete development. Gene Dev 24:1081–1085
Le Trionnaire G, Grant-Downton RT, Kourmpetli S, Dickinson HG, Twell D (2011) Small RNA activity and function in angiosperm gametophytes. J Exp Bot 62:1601–1610
Lelandais-Briere C, Sorin C, Declerck M, Benslimane A, Crespi M, Hartmann C (2010) Small RNA diversity in plants and its impact in development. Curr Genomics 11:14–23
Lu SF, Sun YH, Shi R, Clark C, Li LG, Chiang VL (2005) Novel and mechanical stress-responsive microRNAs in Populus trichocarpa that are absent from Arabidopsis. Plant Cell 17:2186–2203
Lu SF, Sun YH, Amerson H, Chiang VL (2007) MicroRNAs in loblolly pine (Pinus taeda L.) and their association with fusiform rust gall development. Plant J 51:1077–1098
Mallory AC, Vaucheret H (2006) Functions of microRNAs and related small RNAs in plants. Nat Genet 38:S31–S36
Morin RD, Askay G, Dolgosheina D, Ebhardt HA, Magrinio V, Mardiso ER, Cenk Sahinalp SC, Unrau PJ (2008) Comparative analysis of the small RNA transcriptomes of Pinus contorta and Oryza sativa. Genome Res 18:571–584
Nystedt et al (2013) The Norway spruce genome sequence and conifer genome evolution. doi:10.1038/nature12211
Oh TJ, Wartell RM, Cairney J, Pullman GS (2008) Evidence for stage-specific modulation of specific microRNAs (miRNAs) and miRNA processing components in zygotic embryo and female gametophyte of loblolly pine (Pinus taeda). New Phytol 179:67–80
Peng H, Cun J, Ai TB, Tong YA, Zhang R, Zhao MM, Chen F, Wang SH (2012) MicroRNA profiles and their control of male gametophyte development in rice. Plant Mol Biol 80:85–102
Qiu D, Xiaoping P, Wilson IW, Li F, Liu M, Teng W, Zhang B (2009) High-throughput sequencing technology reveals that the taxoid elicit methyl jasmonate regulated microRNA expression in Chinese yew (Taxus chinensis). Gene 432:37–44
Singh H (1978) Embryology of gymnosperms. Gebruder Borntraegar, Berlin
Wan LC, Wang F, Guo X, Lu S, Qiu Z, Zhao Y, Zhang H, Lin J (2012a) Identification and characterization of small non-coding RNAs from Chinese fir by high throughput sequencing. BMC Plant Biol 12:146
Wan LC, Zhang H, Lu S, Zhang L, Qiu Z, Zhao Y, Zeng QY, Lin J (2012b) Transcriptome-wide identification and characterization of miRNAs from Pinus densata. BMC Biol 13:132
Wei LQ, Xu WY, Deng ZY, Su Z, Xue Y, Wang T (2010) Genome-scale analysis and comparison of gene expression profiles in developing and germinated pollen in Oryza sativa. BMC Genomics 11:338
Wei LQ, Yan LF, Wang T (2011) Deep sequencing on genome-wide scale reveals the unique composition and expression patterns of microRNAs in developing pollen of Oryza sativa. Genome Biol 12:R53
Williams J (2012) Pollen tube growth rates and the diversification of flowering plant reproductive cycles. Int J Plant Sci 173:649–661
Yakovlev IA, Fossdal CG, Johnsen O (2010) MicroRNAs, the epigenetic memory and climatic adaptation in Norway spruce. New Phytol 187:1154–1169
Zhan S, Lukens L (2010) Identification of novel miRNAs and miRNA dependent developmental shifts of gene expression in Arabidopsis thaliana. PLoS One 5:e10157
Zhang Y (2005) miRU: an automated plant miRNA target prediction server. Nucleic Acids Res 33:W701–W704
Zhang BH, Pan XP, Cannon CH, Cobb GP, Anderson TA (2006a) Conservation and divergence of plant microRNA genes. Plant J 46:243–259
Zhang B, Pan X, Anderson TA (2006b) Identification of 188 conserved maize microRNAs and their targets. FEBS Lett 580:3753–3762
Zhang S, Zhou J, Han S, Yang W, Li W, Wei H, Li X, Qi L (2010) Four abiotic stress-induced miRNA families differentially regulated in the embryonic and non-embryonic callus tissues of Larix leptolepis. Biochem Biophs Res Commun 398:355–360
Zhang J, Zhang S, Han S, Wu T, Li X, Li W, Qi L (2012) Genome-wide identification of microRNAs in larch and stage-specific modulation of 11 conserved microRNAs and their targets during somatic embryogenesis. Planta 236:647–657
Zhang J, Wu T, Li L, Han S, Li X, Zhang S, Qi L (2013) Dynamic expression of small RNA populations in larch (Larix leptolepis). Planta 237:89–101
Zhao C-Z, Xia H, Frazier TP, Yao YY, Bi YP, Li AQ, Li M-, Li CS, Zhang BH, Wang XJ (2010) Deep sequencing identifies novel and conserved microRNAs in peanuts (Arachis hypogaea L.). BMC Plant Biol 10:3–14
Zhou L, Liu Y, Liu Z, Kong D, Duan M, Luo L (2010) Genome-wide identification and analysis of drought-responsive microRNAs in Oryza sativa. J Exp Bot 61:4157–4168
Zhu Y, Zhao P, Wu X, Wang W, Scali M, Cresti M (2011) Proteomic identification of differentially expressed proteins in mature and germinated maize pollen. Acta Physiol Plant 33:1467–1474
Zou J, Song L, Zhang W, Wang Y, Ruan S, Wu W-H (2009) Comparative proteomic analysis of Arabidopsis mature pollen and germinated pollen. J Int Plant Biol 51:438–455
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Venkatesan Sundaresan.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Quinn, C.R., Iriyama, R. & Fernando, D.D. Expression patterns of conserved microRNAs in the male gametophyte of loblolly pine (Pinus taeda). Plant Reprod 27, 69–78 (2014). https://doi.org/10.1007/s00497-014-0241-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-014-0241-3