Abstract
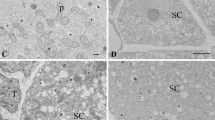
Potassium antimonate was used to locate loosely bound calcium in the stigma and style of tobacco. The tobacco stigma is wet and covered by a thick layer of glycoprotein exudate at anthesis. The exudate contains abundant vesicles, which are densely labeled with calcium precipitates. When pollen grains arrive at the stigma, become hydrated, and as the pollen swells, Ca2+ precipitates accumulate at the aperture. Calcium precipitates that accumulate in pollen cytoplasm are initially concentrated within small vacuoles, but as germination proceeds these appear to fuse, forming prominent, densely labeled vesicles that preferentially accumulate near the proximal region of the growing tube. Although the stigma has abundant particles, few calcium precipitates are observed in the transmitting tissue from anthesis to 11 h after pollination. However, at 22 h after pollination, accumulation of calcium increases distally from the stigmatic interface with the transmitting tissue through the length of the style to the ovary. An examination of flowering plants with differing floral biology will be needed to understand the role of loosely bound calcium accumulation and its relationship to tissue-level changes in calcium uptake, maintenance of other calcium pools, including [Ca2+]cyt, and in pollen and style maturation during the progamic phase.







Similar content being viewed by others
References
Bednarska E (1989) The effect of exogenous Ca2+ ions on pollen grain germination and pollen tube growth: investigations with 45Ca2+ together with verapamil, La3+ and ruthenium red. Sex Plant Reprod 2:53–58
Brewbaker JL, Kwack BH (1963) The essential role of calcium ion in pollen germination and pollen tube growth. Am J Bot 50:859–865
Elleman CJ, Dickinson HG (1999) Commonalities between pollen/stigma and host/pathogen interaction: calcium accumulation during stigmatic penetration by Brassica oleracea pollen tubes. Sex Plant Reprod 12:194–202
Feijó JA, Malhó R, Obermeyer G (1995) Ion dynamics and its possible role during in vitro pollen germination and tube growth. Protoplasma 187:155–167
Ge LL, Tian HQ, Russell SD (2007) Calcium function and distribution during fertilization process of angiosperms. Am J Bot 94:1046–1060
Glenk HO, Eager W, Schimmer O (1971) Can Ca2+ ions act as a chemotropic factor in Oenothera fertilization? In: Heslop-Harrison J (ed) Pollen development and physiology. Appleton-Century-Crofts, New York, pp 255–261
Holdaway-Clarke TL, Feijo JA, Hackett GR, Kunkel JG, Hepler PK (1997) Pollen tube growth and the intracellular cytosolic calcium gradient oscillate in phase while extracellular calcium influx is delayed. Plant Cell 9:1999–2010
Iwano M, Shiba H, Miwa T, Che FS, Takayama S, Nagai T, Miyawaki A, Isogai A (2004) Ca2+ dynamics in a pollen grain and papilla cell during pollination of Arabidopsis. Plant Physiol 136:3562–3571
Lenartowska M, Bednarska E, Butowt R (1997) Ca2+ in the pistil of Petunia hybrida Hort. during growth of the pollen tube: cytochemical and radiographic studies. Acta Biol Crac Ser Bot 39:79–89
Mascarenhas JP (1975) The biochemistry of angiosperm pollen development. Bot Rev 41:259–341
Mascarenhas JP, Machlis L (1962) Chemotropic response of Antirrhinum majus pollen to calcium. Nature 196:292–293
Nobiling R, Reiss HD (1987) Quantitative analysis of calcium gradients and activity in growing pollen tubes of Lilium longiflorum. Protoplasma 139:20–24
Obermeyer G, Weisenseel MH (1991) Calcium channel blocker and calmodulin antagonists affect the gradient of free calcium ions in lily pollen tubes. Eur J Cell Biol 56:319–327
Pierson ES, Miller DD, Callaham DA, Shipley A, Rivers BA, Cresti M, Hepler PK (1994) Pollen tube growth is coupled to the extracellular calcium ion flux and the intracellular gradient: effect of BAPTA-type buffers and hypertonic media. Plant Cell 6:1815–1828
Polito VS (1983) Membrane-associated calcium during pollen grain germination: a microfluorometric analysis. Protoplasma 117:226–232
Qiu Y, Liu R, Xie C, Russell SD, Tian HQ (2008) Calcium changes during megasporogenesis and megaspore degeneration in lettuce (Lactuca sativa L.). Sex Plant Reprod 21:197–204
Reiss HD, Herth W (1978) Visualization of Ca2+ gradient in growing pollen tubes of Lilium longiflorum with chlorotetracycline fluorescence. Protoplasma 97:373–377
Tian HQ, Russell SD (1997) Calcium distribution in fertilized and unfertilized ovules and embryo sacs of Nicotiana tabacum L. Planta 202:93–105
Tian HQ, Kuang A, Musgrave ME, Russell SD (1998) Calcium distribution in fertile and sterile anthers of photoperiod-sensitive genic male-sterile rice. Planta 204:183–192
Yu FL, Zhao J, Liang SP, Yang HY (1999) Ultracytochemical localization of calcium in gynoecium and embryo sac of rice. Acta Bot Sin 41:125–129
Zhang JS, Yang HY, Zhu L, Tong H (1995) Ultracytochemical localization of calcium in the stigma, style and micropyle of sunflower. Acta Bot Sin 37:691–696
Zhang JS, Yang HY, Zhu L, Tong H (1997) Ultracytochemical localization of calcium in the pollen tube track of cotton gynoecium. Acta Bot Sin 39:121–125
Zhao J, Yang HY, Lord EM (2004) Calcium levels increase in the lily stylar transmitting tract after pollination. Sex Plant Reprod 16:259–263
Acknowledgment
This work was supported by the National Natural Science Foundation of China (No. 30670126).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Cresti.
Rights and permissions
About this article
Cite this article
Ge, L.L., Xie, C.T., Tian, H.Q. et al. Distribution of calcium in the stigma and style of tobacco during pollen germination and tube elongation. Sex Plant Reprod 22, 87–96 (2009). https://doi.org/10.1007/s00497-009-0094-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-009-0094-3