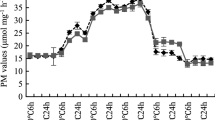
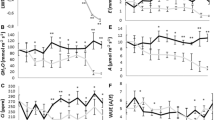
Abstract
The jaborandi is a natural source of alkaloids used in the treatment of important diseases and, therefore, relevant for the pharmaceutical industry. However, the lack of information on the jaborandi ecophysiological responses under different climatic conditions is the major limitation for its expansion in Brazil. Thus, we investigated the effects of different environmental conditions on the ecophysiology of jaborandi seedlings, combining several ranges of mean air temperature (T), vapour pressure deficit (VPD) and water availability. For this, jaborandi seedlings were cultivated in four different environments: cold and humid (Temp: 21.1 °C, VPD: 0.31 kPa), hot and humid (Temp: 26.8 °C, VPD: 0.34 kPa), hot and dry (Temp: 26.3 °C, VPD: 1.09 kPa) and cold and dry (Temp: 20.8 °C, VPD: 0.84 kPa); all the environments had two water availability levels: well-watered (control: C) and water-stressed (45% of the maximum substrate water holding capacity). Growth and fluorescence parameters, crop water stress index and enzymatic antioxidant activity were evaluated. Our results revealed that the seedlings reduced their growth when subjected to water restriction in all treatments, resulting in a lower total dry mass production, mainly due to the reduction of the root system. In the well-watered treatments, jaborandi seedlings showed higher growth when cultivated in warmer environments, regardless of the VPD. Under low-temperature conditions, there was a reduction in maximum quantum yield efficiency of PSII, indicating damage to photosystem II; also, the minimum fluorescence and the enzymatic antioxidants activity increased. The highest dry mass accumulation was obtained when the seedlings were submitted to high temperatures, indicating that the regions that experience such conditions are the most suitable for jaborandi cultivation.







Similar content being viewed by others
References
INPE (Instituto Nacional de Pesquisas Espaciais). INPE consolida 7.536 km2 de desmatamento na Amazônia em 2018. Disponível em: https://www.inpe.br/noticias/noticia.php?Cod_Noticia=5138 (acessado em 12 de agosto de 2019).
ABIQUIFI—Associação Brasileira da Indústria de Insumos Farmacêuticos, (2017) <abiquifi.org.br/abiquiflashes/abiquiflashes-091/> (acessado em 07 de março de 2019).
Adams WW, Muller O, Cohu CM, Demming-Adams B (2013) May photoinhibition be a consequence, rather than a cause of limited plant productivity? Photosynth Res 117:31–44. https://doi.org/10.1007/s11120-013-9849-7
Agban Y, Lian J, Prabakar S, Seyfoddin A, Rupenthal ID (2016) Nanoparticle cross-linked collagen shields for sustained delivery of pilocarpine hydrochloride. Int J Pharm 501:96–101. https://doi.org/10.1016/j.ijpharm.2016.01.069
Aghaie P, Hosseini Tafreshi SA, Ebrahimi MA, Haerinasab M (2018) Tolerance evaluation and clustering of fourteen tomato cultivars grown under mild and severe drought conditions. Sci Hortic 232:1–12. https://doi.org/10.1016/j.scienta.2017.12.041
Alvares CA, Stape JL, Sentelhas PC, Gonçalves JLM, Sparovek G (2013) Köppen’s climate classification map for Brazil. Meteorol Z 22(6):711–728. https://doi.org/10.1127/0941-2948/2013/0507
Alves FSM, Fisch G, Vendrame IF (1999) Microclimate and hydrology modifications due to the deforestation in the Amazônia: a case study in Rondônia State (RO), Brazil. Acta Amazônica 29:395–409. https://doi.org/10.1590/1809-43921999293409
Anderson MD, Prasad TK, Stewart CR (1995) Changes in isozyme profiles of catalase, peroxidase, and glutathione reductase during acclimation to chilling in mesocotyls of maize seedlings. Plant Physiol 109:1247–1257. https://doi.org/10.1104/pp.109.4.1247
Azevedo GFC, Marenco RA (2012) Growth and physiological changes in saplings of Minquartia guianensis and Swietenia macrophylla during acclimation to full sunlight. Photosynthetica 50(1):86–94
Baker NR (2008) Chlorophyll fluorescence: a probe of photosynthesis in vivo. Annu Rev Plant Biol 59:89–113
Barros WS, Dias LAS (2009) Biometria experimental. Editora UFV, Viçosa, pp 135–159
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assay applicable to acrylamide gels. Anal Biochem 44:2762–2787. https://doi.org/10.1016/0003-2697(71)90370-8
Bellvert J, Zarco-Tejada PJ, Girona J, Fereres E (2014) Mapping crop water stress index in a ‘Pinot-noir’ vineyard: comparing ground measurements with thermal remote sensing imagery from an unmanned aerial vehicle. Precis Agric 15:361–376. https://doi.org/10.1007/s11119-013-9334-5
Caldeira CF, Giannini TC, Ramos SJ, Vasconcelos S, Mitre SK, Pires JPA, Ferreira GC, Ohashi S, Mota JÁ, Castilho A, Siqueira JO, Furtini Neto AE (2017) Sustainability of Jaborandi in the eastern Brazilian Amazon. Perspecti Ecol Conserv 15:161–171. https://doi.org/10.1016/j.pecon2017.08.002
Calzadilla PI, Signorelli S, Escaraya FJ, Menéndez AB, Monza J, Ruiz OA, Maialea SJ (2016) Photosynthetic responses mediate the adaptation of two Lotus japonicus ecotypes to low temperature. Plant Sci 250:59–68. https://doi.org/10.1016/j.plantsci.2016.06.003
Cano FJ, López R, Warren CR (2014) Implications of the mesophyll conductance to CO2 for photosynthesis and water-use efficiency during long-term water stress and recovery in two contrasting Eucalyptus species. Plant Cell Environ 37:2470–2490. https://doi.org/10.1111/pce.12325
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103:551–560. https://doi.org/10.1093/aob/mcn125
CNCFlora. Pilocarpus microphyllus in Lista Vermelha da flora brasileira versão 2012.2 Centro Nacional de Conservação da Flora. Disponível em https://cncflora.jbrj.gov.br/portal/pt-br/profile/Pilocarpus microphyllus (acesso em 31 março 2019).
Coelho MFB, Teixeira VA, Azevedo RAB, Albuquerque MCF (2013) Propagation of ipecac (Psychotria ipecacuanha) on different substrates and planting position of cuttings. Hortic Bras 31:467–471. https://doi.org/10.1590/S0102-05362013000300020
Del Longo OT, Goinz´zlez CA, Pastori GM, Trippi VS (1993) Antioxidant defenses under hyperoxygenic and hyperosmotic conditions in leaves of two lines of maize with differential to drought. Plant Cell Physiol 37:1023–1028https://doi.org/10.1093/oxfordjournals.pcp.a078515
Freire JC, Ribeiro MAV, Bahia VG, Lopes AS, Aquino LH (1980) Respostas do milho cultivado em casa de vegetação a níveis de água em solos da região de Lavras (MG). Revista Brasileira de Ciência do Solo 4:5–8
García-Tejero IF, Costa JM, Egipto R, Duran-Zuazo VH, Lima RSN, Lopes CM, Chaves MM (2016) Thermal data to monitor crop-water status in irrigated Mediterranean viticulture. Agric Water Manag 176:80–90. https://doi.org/10.1016/j.agwat.2016.05.008
Gelcer E, Fraisse C, Dzotsi K, Hu Z, Mendes R, Zotarelli L (2013) Effects of El Niño Southern Oscillation on the space–time variability of Agricultural Reference Index for Drought in midlatitudes. Agric For Meteorol 174–175:110–128. https://doi.org/10.1016/j.agrformet.2013.02.006
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I., occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.1104/pp.59.2.309
Gil-Montoya JA, Silvestre FJ, Barrios R, Silvestre-Rangil J (2016) Treatment of xerostomia and hyposalivation in the elderly: a systematic review. Medicina Oral Patologia Oral y Cirugiabucal 21:355–366. https://doi.org/10.4317/medoral.20969
Guo XY, Zhang XS, Huang ZY (2010) Drought tolerance in three hybrid poplar clones submitted to different watering regimes. J Plant Ecol 3:79–87. https://doi.org/10.1093/jpe/rtq007
Guo WL, Chen RG, Du XH, Zhang Z, Yin YX, Gong ZH, Wang GY (2014) Reduced tolerance to abiotic stress in transgenic Arabidopsis overexpressing a Capsicum annuum multiprotein bridging factor 1. BMC Plant Biol 14:125–138. https://doi.org/10.1186/1471-2229-14-138
Hartley HO (1950) The use of range in analysis of variance. Biometrika Lond 37:271–280
Havir EA, McHale NA (1987) Biochemical and developmental characterization of multiple forms of catalase in tobacco leaves. Plant Physiol 84:450–455. https://doi.org/10.1104/pp.84.2.450
He Y, Yang J, Zhu B, Zhu ZJ (2014) Low root zone temperature exacerbates the ion imbalance and photosynthesis inhibition and induces antioxidant responses in tomato plants under salinity. J Integr Agric 13:89–99. https://doi.org/10.1016/s2095-3119(13)60586-9
Hikosaka K, Ishikawa K, Borjigidai A, Muller O, Onoda Y (2006) Temperature acclimation of photosynthesis: mechanisms involved in the changes in temperature dependence of photosynthetic rate. J Exp Bot 57:291–302
IBAMA (Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis). Instrução Normativa do Ministério do Meio Ambiente (MMA) no. 06, de 26 de setembro de 2008. Estabelece a Lista Oficial de Espécies Brasileiras Ameaçadas de Extinção. https://www.mma.gov.br/estruturas/179/_arquivos/179_05122008033615.pdf (acessado em 12 de julho de 2019).
Idso SB (1982) Non-water-stressed baselines: a key to measuring and interpreting plant water stress. Agric Meteorol 27:59–70
Jackson RD, Idso SB, Reginato RJ, Pinter PJ (1981) Canopy temperature as a crop water stress indicator. Water Resour Res 17:1133–1138. https://doi.org/10.1029/wr017i004p01133
Khanal N, Bray GE, Grisnich A, Moffatt BA, Gray GR (2017) Differential mechanisms of photosynthetic acclimation to light and low temperature in arabidopsis and the extremophile eutrema salsugineum. Plants 6(4):32. https://doi.org/10.3390/plantas6030032
Koshiba T (1993) Cytosolic ascorbate peroxidase in seedlings and leaves of maize (Zea mays). Plant Cell Physiol 34:713–721. https://doi.org/10.1093/oxfordjournals.pcp.a078474
Lima DF, Lima LI, Rocha JA, Andrade IM, Grazina LG, Villa C, Meira L, Véras LMC, Azevedo IFS, Biase AG, Costa J, Oliveira MBPP, Mafra I, Leite JRSA (2017) Seasonal change in main alkaloids of Jaborandi (Pilocarpus microphyllus Stapf ex Wardleworth), an economically important species from the Brazilian flora. PLoS One 12:1–19. https://doi.org/10.1371/journal.pone.0170281
Li J, Cang Z, Jiao F, Bai X, Zhang D, Zhai R (2017) Influence of drought stress on photosynthetic characteristics and protective enzymes of potato at seedling stage. J Saudi Soc Agric Sci 16:82–88. https://doi.org/10.1016/j.jssas.2015.03.001
Marenco RA, Antezana-Vera AS, Gouvêa PRS, Camargo MAB, Oliveira MF, Santos JKS (2014) Physiology of Amazon tree species: photosynthesis, respiration and water relations. Revista Ceres 61:786–799. https://doi.org/10.1590/0034-737x201461000004
Matese A, Baraldi R, Berton A, Cesaraccio C, Di Gennaro SF, Duce P, Facini O, Mameli MG, Piga A, Zaldei A (2018) Estimation of water stress in grapevines using proximal and remote sensing methods. Rem Sens 10:1–16. https://doi.org/10.3390/rs10010114
Mishra KB, Mishra A, Kubásek J, Urban O, Heyer AG, Govindjee, (2018) Low temperature induced modulation of photosynthetic induction in non-acclimated and cold-acclimated Arabidopsis thaliana: chlorophyll a fluorescence and gas-exchange measurements. Photosynth Res. https://doi.org/10.1007/s11120-018-0588-7
Murchie EH, Lawson T (2013) Chlorophyll fluorescence analysis: a guide to good practice and understanding some new applications. J Exp Bot 64:3983–3998. https://doi.org/10.1093/jxb/ert208
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplast. Plant Cell Physiology 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Nóia Júnior RS, Sentelhas PC (2019) Soybean-maize off-season double crop system in Brazil as affected by El Niño Southern Oscillation phases. Agric Syst 173:254–267. https://doi.org/10.1016/j.agsy.2019.03.012
Nóia Júnior RS, Pezzopane JEM, Vinco JS, Xavier TMT, Cecílio RA, Pezzopane JRM (2018) Characterization of photosynthesis and transpiration in two rubber tree clones exposed to thermal stress. Braz J Bot 41:785–794. https://doi.org/10.1007/s40415-018-0495-3
Nóia Júnior RS, Amaral GC, Pezzopane JEM, Toledo JV, Xavier TMT (2018) Ecophysiology of C3 and C4 plants in terms of responses to extreme soil temperatures. Theor Exp Plant Physiol 30:261–274. https://doi.org/10.1007/s40626-018-0120-7
Nóia Júnior RS, Amaral GC, Pezzopane JEM, Fonseca MDS, Silva APC, Xavier TMT (2019) Ecophysiological acclimatization to cyclic water stress in Eucalyptus. J For Res 1:1–10. https://doi.org/10.1007/s11676-019-00926-9
Peixoto PHP, Cambraia J, Sant’s Anna R, Mosquin PR, Moreira MA (1999) Aluminum effects on lipid peroxidation and on the activities of enzymes of oxidative metabolism in sorghum. Revista Brasileira de Fisiologia Vegetal. 3:137–143
Pou A, Diago MP, Medrano H, Baluja J, Tardaguila J (2014) Validation of thermal indices for water status identification in grapevine. Agric Water Manag 134:60–72. https://doi.org/10.1016/j.agwat.2013.11.010
Prieto P, Peñuelas J, Llusià J, Asensio D, Estiarte M (2009) Effects of long-term experimental night-time warming and drought on photosynthesis, Fv/Fm and stomatal conductance in the dominant species of a Mediterranean shrubland. Acta Physiol Plant 31(4):729–739. https://doi.org/10.1007/s11738-009-0285-4
R Core Team, 2017. R: A Language and Environment for Statistical Computing. R Found. Stat. Comput., Vienna, Austria (2017) https://www.R-project.org/
Ribeiro FNS, Hüther CM, TdeB M, Lameira AO, Pereira CR (2017) Propagação de ipecacuanha por estacas de raiz em diferentes condições ambientais e substratos. Enciclopédia Biosfera 14:1072–1083
Rocha JÁ, Vasconcelos S, Silva FMM, Melo AJ, Silva MFS, Miranda JAL, Benko-Iseppon AM, Andrade IM (2014) ISSR Primer selection for genetic variability analyses with Jaborandi (Pilocarpus microphyllus Stapf ex Wardlew., Rutaceae). For Res 126:1–5. https://doi.org/10.4172/2168-9776.1000126
Salazar-Parra C, Aguirreolea J, Sánchez-Díaz M, Irigoyen JJ, Morales F (2012) Climate change (elevated CO2, elevated temperature and moderate drought) triggers the antioxidant enzymes’ response of grapevine cv. Tempranillo, avoiding oxidative damage. Physiol Plantarum 144:99–110. https://doi.org/10.1111/j.1399-3054.2011.01524.x
Sales CRG, Ribeiro RV, Machado DFSP, Machado DS, Dovis VL, Lagôa AMMA (2012) Trocas gasosas e balanço de carboidratos em plantas de cana-de-açúcar sob condições de estresses radiculares. Bragantina 71:319–327. https://doi.org/10.1590/s0006-87052012000300001
Sánchez-Rodríguez E, Rubio-Wilhelmi MDM, Blasco B, Leyva R, Romero L, Ruiz JM (2012) Antioxidant response resides in the shoot in reciprocal grafts of drought-tolerant and drought-sensitive cultivars in tomato under water stress. Plant Sci 188–189:89–96. https://doi.org/10.1016/j.plantsci.2011.12.019
Santos CMA, Ribeiro RV, Magalhães Filho JR, Machado DFSP, Machado EC (2011) Low substrate temperature imposes higher limitation to photosynthesis of orange plants as compared to atmospheric chilling. Photosynthetica 49:546–554. https://doi.org/10.1007/s11099-011-0071-6
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika, London 52(3/4):591–611
Shibasaki K, Uemura M, Tsurumi S, Rahman A (2009) Auxin response in Arabidopsis under cold stress: underlying molecular mechanisms. Plant Cell 21:3823–3838. https://doi.org/10.1105/tpc.109.069906
Silva VG, Silva RO, Damasceno SR, Carvalho NS, Prudêncio RS, Aragão KS, Guimarães MA, Campos SA, Véras LM, Godejohann M, Leite JR, Barbosa AL, Medeiros JV (2013) Anti-inflammatory and antinociceptiva activity of epiisopiloturina, an imidazole alkaloid isolated from Pilocarpus microphyllus. J Nat Prod 76:1071–1077. https://doi.org/10.1021/np400099m
Stott P (2016) How climate change affects extreme weather events. Science 352(6293):1517–1518. https://doi.org/10.1126/science.aaf7271
Taiz L, Zeiger E (2013) Fisiologia vegetal, 5th edn. Artmed, Porto Alegre, p 918
Tang YY, Yuan YH, Shu S, Guo SR (2018) Regulatory mechanism of NaCl stress on photosynthesis and antioxidant capacity mediated by transglutaminase in cucumber (Cucumis sativus L.) seedlings. Sci Hortic 235:294–306. https://doi.org/10.1016/j.scienta.2018.02.045
Tardieu F, Granier C, Muller B (2011) Water deficit and growth. Co-ordinating processes without an orchestrator? Curr Opin Plant Biol 14:283–289. https://doi.org/10.1016/j.pbi.2011.02.002
Tezara W, Mitchell V, Driscoll SP, Lawlor DW (2002) Effects of water deficit and its interaction with CO2 supply on the biochemistry and physiology of photosynthesis in sunflower. J Exp Bot 53:1781–1791. https://doi.org/10.1093/jxb/erf021
Thimmanaik S, Kumar SG, Kumari GJ, Suryanarayana N, Sudhakar C (2002) Photosynthesis and the enzymes of photosynthetic carbon reduction cycle in mulberry during water stress and recovery. Photosynthetica 40:233–236. https://doi.org/10.1016/b978-0-12-675408-7.50011-2
Vazquez P (2013) Comparison of water stress indices from information collected by the MODIS sensor in the Argentine Pampean region. Geographic Cuadernos 52:46–68
Véras L, Guimaraes M, Campelo Y, Vieira M, Nascimento C, Lima DF, Vasconcelos L, Nakano E, Kuckelhaus SS, Batista MC, Leite JR, Moraes J (2012) Activity of epiisopiloturine against Schistosoma mansoni. Curr Med Chem 19:2051–2058. https://doi.org/10.2174/092986712800167347
Warren CR, Aranda I, Cano FJ (2011) Responses to water stress of gas exchange and metabolites in Eucalyptus and Acacia spp. Plant Cell Environ 34:1609–1629. https://doi.org/10.1111/j.1365-3040.2011.02357.x
Xavier TMT, Pezzopane JEM, Pereira CL, Nóia Junior RS, Cruz GPR (2017) Crescimento de espécies florestais da mata atlântica sob diferentes condições microclimáticas. Re. C. E. E. F. 30: 1–14
Xavier TMT, Pezzopane JEM, Penchel RM, Pezzopane RM (2018) Initial growth of eucalyptus under different atmospheric demands and water replacement intervals. Irriga 23:426–441. https://doi.org/10.15809/irriga.2018v23n3p426-441
Yamori W, Hikosaka K, Way DA (2014) Temperature response of photosynthesis in C3, C4, and CAM plants: temperature acclimation and temperature adaptation. Photosynth Res 119:101–117. https://doi.org/10.1007/s11120-013-9874-6
Zha TS, Wu YJ, Jia X, Zhang MY, Bai YJ, Liu P, Ma JY, Bourque CPA, Peltola H (2017) Diurnal response of effective quantum yield of PSII photochemistry to irradiance as an indicator of photosynthetic acclimation to stressed environments revealed in a xerophytic species. Ecol Ind 74:191–197. https://doi.org/10.1016/j.ecolind.2016.11.027
Zlatev Z, Lidon FC (2012) An overview on drought induced changes in plant growth, water relations and photosynthesis. Emir J Food Agric 24:57–72. https://doi.org/10.9755/ejfa.v24i1.10599
Acknowledgements
This work was supported by FAPES (Fundação de Apoio à Pesquisa do Estado do Espírito Santo) with research funding and a doctoral scholarship to the first author (PROCAP 2016 call).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Fernandez-Marin.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Amaral, G.C., Pezzopane, J.E.M., de Souza Nóia Júnior, R. et al. Ecophysiology of Pilocarpus microphyllus in response to temperature, water availability and vapour pressure deficit. Trees 35, 543–555 (2021). https://doi.org/10.1007/s00468-020-02055-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-020-02055-x