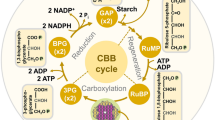
Abstract
Most plants show considerable capacity to adjust their photosynthetic characteristics to their growth temperatures (temperature acclimation). The most typical case is a shift in the optimum temperature for photosynthesis, which can maximize the photosynthetic rate at the growth temperature. These plastic adjustments can allow plants to photosynthesize more efficiently at their new growth temperatures. In this review article, we summarize the basic differences in photosynthetic reactions in C3, C4, and CAM plants. We review the current understanding of the temperature responses of C3, C4, and CAM photosynthesis, and then discuss the underlying physiological and biochemical mechanisms for temperature acclimation of photosynthesis in each photosynthetic type. Finally, we use the published data to evaluate the extent of photosynthetic temperature acclimation in higher plants, and analyze which plant groups (i.e., photosynthetic types and functional types) have a greater inherent ability for photosynthetic acclimation to temperature than others, since there have been reported interspecific variations in this ability. We found that the inherent ability for temperature acclimation of photosynthesis was different: (1) among C3, C4, and CAM species; and (2) among functional types within C3 plants. C3 plants generally had a greater ability for temperature acclimation of photosynthesis across a broad temperature range, CAM plants acclimated day and night photosynthetic process differentially to temperature, and C4 plants was adapted to warm environments. Moreover, within C3 species, evergreen woody plants and perennial herbaceous plants showed greater temperature homeostasis of photosynthesis (i.e., the photosynthetic rate at high-growth temperature divided by that at low-growth temperature was close to 1.0) than deciduous woody plants and annual herbaceous plants, indicating that photosynthetic acclimation would be particularly important in perennial, long-lived species that would experience a rise in growing season temperatures over their lifespan. Interestingly, across growth temperatures, the extent of temperature homeostasis of photosynthesis was maintained irrespective of the extent of the change in the optimum temperature for photosynthesis (T opt), indicating that some plants achieve greater photosynthesis at the growth temperature by shifting T opt, whereas others can also achieve greater photosynthesis at the growth temperature by changing the shape of the photosynthesis–temperature curve without shifting T opt. It is considered that these differences in the inherent stability of temperature acclimation of photosynthesis would be reflected by differences in the limiting steps of photosynthetic rate.






Similar content being viewed by others
References
Atkin OK, Scheurwater I, Pons TL (2006) High thermal acclimation potential of both photosynthesis and respiration in two lowland Plantago species in contrast to an alpine congeneric. Global Change Biol 12:500–515
Atkin OK, Tjoelker MG (2003) Thermal acclimation and the dynamic response of plant respiration to temperature. Trends Plant Sci 8:343–351
Atkin OK, Bruhn D, Hurry VM, Tjoelker MG (2005) The hot and the cold: unravelling the variable response of plant respiration to temperature. Funct Plant Biol 32:87–105
Barua D, Downs CA, Heckathorn SA (2003) Variation in chloroplast small heat-shock protein function is a major determinant of variation in thermotolerance of photosynthetic electron transport among ecotypes of Chenopodium album. Funct Plant Biol 30:1071–1079
Bernacchi CJ, Portis AR, Nakano H, Von Caemmerer S, Long SP (2002) Temperature response of mesophyll conductance. Implications for the determination of Rubisco enzyme kinetics and for limitations to photosynthesis in vivo. Plant Physiol 130:1992–1998
Bernacchi CJ, Rosenthal DM, Pimentel C, Long SP, Farquhar GD (2009) Modeling the temperature dependence of C3 photosynthesis. In: Laisk A, Nedbal L, Govindjee (eds) Photosynthesis in silico: understanding complexity from molecules to ecosystems. Springer Science + Business Media B.V., Dordrecht, pp 231–246
Berry JA, Björkman O (1980) Photosynthetic response and adaptation to temperature in higher plants. Ann Rev Plant Physiol 31:491–543
Björkman O, Mooney HA, Ehleringer J (1975) Photosynthetic responses of plants from habitats with contrasting thermal environments: comparison of photosynthetic characteristics of intact plants. Carnegie Inst Wash 74:743–748
Borland AM, Griffiths H, Hartwell J, Smith JAC (2009) Exploiting the potential of plants with crassulacean acid metabolism for bioenergy production on marginal lands. J Exp Bot 60:2879–2896
Brandon PC (1967) Temperature features of enzymes affecting crassulacean acid metabolism. Plant Physiol 42:977–984
Bukhov NG, Wiese C, Neimanis S, Heber U (1999) Heat sensitivity of chloroplasts and leaves: leakage of protons from thylakoids and reversible activation of cyclic electron transport. Photosynth Res 59:81–93
Bukhov NG, Samson G, Carpentier R (2000) Nonphotosynthetic reduction of the intersystem electron transport chain of chloroplasts following heat stress: steady-state rate. Photochem Photobiol 72:351–357
Campbell C, Atkinson L, Zaragoza-Castells J, Lundmark M, Atkin OK, Hurry V (2007) Acclimation of photosynthesis and respiration is asynchronous in response to changes in temperature regardless of plant functional group. New Phytol 176:375–389
Carter KK (1996) Provenance tests as indicators of growth response to climate change in 10 north temperate tree species. Can J For Res 26:1089–1095
Cen Y-P, Sage RF (2005) The regulation of ribulose-1,5-bisphosphate carboxylase activity in response to variation in temperature and atmospheric CO2 partial pressure in sweet potato. Plant Physiol 139:1–12
Chinthapalli B, Murmu J, Raghavendra AS (2003) Dramatic difference in the responses of phosphoenolpyruvate carboxylase to temperature in leaves of C3 and C4 plants. J Exp Bot 54:707–714
Christensen JH, Hewitson B, Busuioc A, Chen A, Gao X et al. (2007) Regional climate projections. In: Solomon S, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Tignor M, Miller HL (eds) Climate change 2007: the physical science basis. Contribution of Working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge and New York, pp 847–940
Crafts-Brandner SJ, Salvucci ME (2000) Rubisco activase constrains the photosynthetic potential of leaves at high temperature and CO2. Proc Natl Acad Sci USA 97:13430–13435
Crafts-Brandner SJ, van de Loo FJ, Salvucci ME (1997) The two forms of ribulose-1,5-bisphosphate carboxylase/oxygenase activase differ in sensitivity to elevated temperature. Plant Physiol 114:439–444
Cunningham SC, Read J (2002) Comparison of temperate and tropical rainforest tree species: photosynthetic responses to growth temperature. Oecologia 133:112–119
Deiting U, Zrenner R, Stitt M (1998) Similar temperature requirement for sugar accumulation and for the induction of new forms of sucrose phosphate synthase and amylase in cold-stored potato tubers. Plant Cell Environ 21:127–138
Dittrich P (1976) Nicotinamide adenine dinucleotide-specific “Malic” enzyme in Kalanchoe daigremontiana and other plants exhibiting crassulacean acid metabolism. Plant Physiol 57:310–314
Dittrich P, Campbell WH, Black JRCC (1973) Phosphoenolpyruvate carboxykinase in plants exhibiting crassulacean acid metabolism. Plant Physiol 52:357–361
Du Y-C, Nose A, Wasano K (1999) Effects of chilling temperature on photosynthetic rates, photosynthetic enzyme activities and metabolite levels in leaves of three sugarcane species. Plant Cell Environ 22:317–324
Dwyer SA, Ghannoum O, Nicotra A, von Caemmerer S (2007) High temperature acclimation of C4 photosynthesis is linked to changes in photosynthetic biochemistry. Plant Cell Environ 30:53–66
Ehleringer JR (1978) Implications of quantum yield differences on the distributions of C3 and C4 grasses. Oecologia 31:255–267
Ehleringer JR, Björkman O (1977) Quantum yields for CO2 uptake of C3 and C4 plants. Dependence on temperature, CO2 and O2 concentrations. Plant Physiol 59:86–90
Evans JR (1989) Photosynthesis and nitrogen relationships in leaves of C3 plants. Oecologia 78:9–19
Falcone DL, Ogas JP, Somerville CR (2004) Regulation of membrane fatty acid composition by temperature in mutants of Arabidopsis with alterations in membrane lipid composition. BMC Plant Biol 4:17
Farquhar GD, von Caemmerer S, Berry JA (1980) A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 149:78–90
Fischer RA, Edmeades GO (2010) Breeding and cereal yield progress. Crop Sci 50:S85–S98
Furbank RT, Hatch MD (1987) Mechanism of Cq photosynthesis. The size and composition of the inorganic carbon pool in the bundle sheath cells. Plant Physiol 85:958–964
Furbank RT, Chitty JA, von Caemmerer S, Jenkins CLD (1996) Antisense RNA inhibition of rbcS gene expression reduces Rubisco level and photosynthesis in the C4 plant Flaveria bidentis. Plant Physiol 111:725–734
Furbank RT, Chitty JA, Jenkins CLD, Taylor WC, Trevanion SJ, von Caemmerer S, Ashton AR (1997) Genetic manipulation of key photosynthetic enzymes in the C4 plant Flaveria bidentis. Aust J Plant Physiol 24:477–485
Genty B, Briantais J-M, Baker NR (1989) The relationship between quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Gombos Z, Wada H, Hideg E, Murata N (1994) The unsaturation of membrane lipids stabilizes photosynthesis against heat stress. Plant Physiol 104:563–567
Guy CL, Huber JLA, Huber SC (1992) Sucrose phosphate synthase and sucrose accumulation at low temperature. Plant Physiol 99:1443–1448
Havaux M (1996) Short-term responses of photosystem I to heat stress. Photosynth Res 47:85–97
Heckathorn SA, Downs CA, Sharkey TD, Coleman JS (1998) The small, methionine-rich chloroplast heat-shock protein protects photosystem II electron transport during heat stress. Plant Physiol 116:439–444
Heckathorn SA, Ryan SL, Baylis JA, Wang DF, Hamilton EW III, Cundiff L, Luthe DS (2002) In vivo evidence from an Agrostis stolonifera selection genotype that chloroplast small heat-shock proteins can protect photosystem II during heat stress. Funct Plant Biol 29:933–944
Hendrickson L, Sharwood R, Ludwig M, Whitney SM, Badger MR, von Caemmerer S (2008) The effects of Rubisco activase on C4 photosynthesis and metabolism at high temperature. J Exp Bot 59:1789–1798
Hikosaka K (2004) Interspecific difference in the photosynthesis–nitrogen relationship: patterns, physiological causes, and ecological importance. J Plant Res 117:481–494
Hikosaka K (2005) Nitrogen partitioning in the photosynthetic apparatus of Plantago asiatica leaves grown at different temperature and light conditions: similarities and differences between temperature and light acclimation. Plant Cell Physiol 46:1283–1290
Hikosaka K, Murakami A, Hirose T (1999) Balancing carboxylation and regeneration of ribulose bisphosphate in leaf photosynthesis: temperature acclimation in an evergreen tree, Quercus myrsinaefolia. Plant Cell Environ 22:841–849
Hikosaka K, Ishikawa K, Borjigidai A, Muller O, Onoda Y (2006) Temperature acclimation of photosynthesis: mechanisms involved in the changes in temperature dependence of photosynthetic rate. J Exp Bot 57:291–302
Hill RS, Read J, Busby JR (1988) The temperature-dependence of photosynthesis of some Australian temperate rainforest trees and its biogeographical significance. J Biogeogr 15:431–449
Holaday AS, Martindale W, Alred R, Brooks AL, Leegood RC (1992) Changes in activities of enzymes of carbon metabolism in leaves during exposure of plants to low temperature. Plant Physiol 98:1105–1114
Huner NPA, Macdowall FDH (1979) Changes in the net charge and subunit properties of ribulose bisphosphate carboxylase-oxygenase during cold hardening of Puma rye. Can J Biochem 57:1036–1041
Hurry VM, Malmberg G, Gardeström P, Öquist G (1994) Effects of a short-term shift to low temperature and of long-term cold hardening on photosynthesis and ribulose 1,5-bisphosphate carboxylase/oxygenase and sucrose phosphate synthase activity in leaves of winter rye (Secale cereale L.). Plant Physiol 106:983–990
Hurry VM, Strand Å, Tobiæson M, Gardeström P, Öquist G (1995) Cold hardening of spring and winter wheat and rape results in differential effects on growth, carbon metabolism, and carbohydrate content. Plant Physiol 109:697–706
Ishikawa K, Onoda Y, Hikosaka K (2007) Intraspecific variation in temperature dependence of gas exchange characteristics of Plantago asiatica ecotypes from different temperature regimes. New Phytol 176:356–364
Jenkins CLD, Furbank RT, Hatch MD (1989) Inorganic carbon diffusion between C4 mesophyll and bundle sheath cells. Plant Physiol 91:1356–1363
Jordan DB, Ogren WL (1984) The CO2/O2 specificity of ribulose 1,5-bisphosphate carboxylase/oxygenase. Dependence on ribulose bisphosphate concentration, pH and temperature. Planta 161:308–313
Kattge J, Knorr W (2007) The temperature dependence of photosynthetic capacity in a photosynthesis model acclimates to plant growth temperature: a re-analysis of data from 36 species. Plant Cell Environ 30:1176–1190
Kingston-Smith AH, Harbinson J, WIlliams J, Foyer CH (1997) Effect of chilling on carbon assimilation, enzyme activation, and photosynthetic electron transport in the absence of photoinhibition in maize leaves. Plant Physiol 114:1039–1046
Kubien DS, Sage RF (2004) Low-temperature photosynthetic performance of a C4 grass and a co-occurring C3 grass native to high latitudes. Plant Cell Environ 27:907–916
Kubien DS, von Cammerer S, Furbank RT, Sage RF (2003) C4 photosynthesis at low temperature. A study using transgenic plants with reduced amounts of Rubisco. Plant Physiol 132:1577–1585
Kumar A, Li C, Portis Jr. AR (2009) Arabidopsis thaliana expressing a thermostable chimeric Rubisco activase exhibits enhanced growth and higher rates of photosynthesis at moderately high temperatures. Photosynth Res 100:143–153
Kurek I, Chang TK, Bertain SM, Madrigal A, Liu L, Lassner MW, Zhu G (2007) Enhanced thermostability of Arabidopsis Rubisco activase improves photosynthesis and growth rates under moderate heat stress. Plant Cell 19:3230–3241
Labate CA, Leegood RC (1988) Limitation of photosynthesis by changes in temperature. Planta 173:519–527
Law RD, Crafts-Brandner SJ (2001) High temperature stress increases the expression of wheat leaf ribulose-1,5-bisphosphate carboxylase/oxygenase activase protein. Arch Biochem Biophys 386:261–267
Law RD, Crafts-Brandner SJ, Salvucci ME (2001) Heat stress induces the synthesis of a new form of ribulose-1,5-bisphosphate carboxylase/oxygenase activase in cotton leaves. Planta 214:117–125
Leegood RC, Edwards GE (1996) Carbon metabolism and photorespiration: temperature dependence in relation to other environmental factors. In: Baker NR (ed) Photosynthesis and the environment. Kluwer Academic, Dordrecht, pp 191–121
Lobell DB, Asner GP (2003) Climate and management contributions to recent trends in U.S. agricultural yields. Science 299:1032
Long SP (1983) C4 photosynthesis at low temperatures. Plant Cell Environ 6:345–363
Lüttge U (2004) Ecophysiology of crassulacean acid metabolism (CAM). Ann Bot 93:629–652
Makino A, Sakuma H, Sudo E, Mae T (2003) Differences between maize and rice in N-use efficiency for photosynthesis and protein allocation. Plant Cell Physiol 44:952–956
Mittler R (2006) Abiotic stress, the field environment and stress combination. Trends Plant Sci 11:15–19
Mooney HA (1980) Photosynthetic plasticity of populations of Heliotropium curassavicum L. Originating from differing thermal regimes. Oecologia (Berl.) 45:372–376
Murakami Y, Tsuyama M, Kobayashi Y, Kodama H, Iba K (2000) Trienoic fatty acids and plant tolerance of high temperature. Science 287:476–479
Murata N, Los DA (1997) Membrane fluidity and temperature perception. Plant Physiol 115:875–879
Naidu SL, Moose SP, Al-Shoaibi AK, Raines CA, Long SP (2003) Cold tolerance of C4 photosynthesis in Miscanthus × giganteus: adaptation in amounts and sequence of C4 photosynthetic enzymes. Plant Physiol 132:1688–1697
Neta-Sharir I, Isaacson T, Lurie S, Weiss D (2005) Dual role for tomato heat shock protein 21: protecting photosystem II from oxidative stress and promoting color changes during fruit maturation. Plant Cell 17:1829–1838
Nobel P (1996) High productivities of certain agronomic CAM species. In: Winter K, Smith JAC (eds) Crassulacean acid metabolism. Biochemistry, ecophysiology and evolution. Springer-Verlag, Berlin, pp 255–265
Oberhuber WT, Edwards GE (1993) Temperature dependence of the linkage of quantum yield of photosystem II to CO2 fixation in C4 and C3 plants. Plant Physiol 101:507–512
Ogren E, Evans JR (1993) Photosynthetic light-response curves: I. The influence of CO2 partial pressure and leaf inversion. Planta 189:180–190
Ohta S, Ishida Y, Usami S (2006) High-level expression of cold-tolerant pyruvate, orthophosphate dikinase from a genomic clone with site-directed mutations in transgenic maize. Mol Breed 18:29–38
Osborne CP, Wythe EJ, Ibrahim DG, Gilbert ME, Ripley BS (2008) Low temperature effects on leaf physiology and survivorship in the C3 and C4 subspecies of Alloteropsis semialata. J Exp Bot 59:1743–1754
Ow LF, Griffin KL, Whitehead D, Walcroft AS, Turnbull MH (2008) Thermal acclimation of leaf respiration but not photosynthesis in Populus deltoides × nigra. New Phytol 178:123–134
Ow LF, Whitehead D, Walcroft AS, Turnbull M (2010) Seasonal variation in foliar carbon exchange in Pinus radiata and Populus deltoides: respiration acclimates fully to changes in temperature but photosynthesis does not. Global Change Biol 16:288–302
Pastenes C, Horton P (1996) Effect of high temperature on photosynthesis in bean: II CO2 assimilation and metabolite contents. Plant Physiol 112:1253–1260
Pearcy RW (1977) Acclimation of photosynthetic and respiratory carbon dioxide exchange to growth temperature in Atriplex lentiformis (Torr.). Plant Physiol 59:795–799
Pengelly JJL, Tan J, Furbank RT, von Caemmerer S (2012) Antisense reduction of NADP-malic enzyme in Flaveria bidentis reduces flow of CO2 through the C4 cycle. Plant Physiol 160:1070–1080
Pittermann J, Sage RF (2000) Photosynthetic performance at low temperature of Bouteloua gracilis Lag., a high-altitude C4 grass from the Rocky Mountains, USA. Plant Cell Environ 23:811–823
Pittermann J, Sage R (2001) The response of the high altitude C4 grass Muhlenbergia montana (Nutt.) A.S. Hitchc. to long- and short-term chilling. J Exp Bot 52:829–838
Price GD, von Caemmerer S, Evans JE, Yu JE, Lloyd L, Oja V, Kell P, Harrison K, Gallagher A, Badger MR (1994) Specific reduction of chloroplast carbonic anhydrase activity by antisense RNA in transgenic tobacco plants has a minor effect on photosynthetic CO2 assimilation. Planta 193:331–340
Raines CA (2006) Transgenic approaches to manipulate the environmental responses of the C3 carbon fixation cycle. Plant Cell Environ 29:331–339
Read J (1990) Some effects of acclimation temperature on net photosynthesis in some tropical and extra-tropical Australasian Nothofagus species. J Ecol 78:100–112
Read J, Busby JR (1990) Comparative responses to temperature of the major canopy species of Tasmanian cool temperate rainforest and their ecological significance II. Net photosynthesis and climate analysis. Aust J Bot 38:185–205
Reimholz R, Geiger M, Deiting U, Krause KP, Sonnewald U, Stitt M (1997) Potato plants contain multiple forms of sucrose phosphate synthase, that show differences in their tissue distribution, their response during development, and their response to low temperature. Plant Cell Environ 20:291–305
Sage RF (2002) Variation in the kcat of Rubisco in C3 and C4 plants and some implications for photosynthetic performance at high and low temperature. J Exp Bot 53:609–620
Sage RF, Kubien DS (2007) The temperature response of C3 and C4 photosynthesis. Plant Cell Environ 30:1086–1106
Sage RF, McKown AD (2006) Is C4 photosynthesis less phenotypically plastic than C3 photosynthesis? J Exp Bot 57:303–317
Sage RF, Sharkey TD (1987) The effect of temperature on the occurrence of O2 and CO2 insensitive photosynthesis in field grown plants. Plant Physiol 84:658–664
Sage RF, Way DA, Kubien DS (2008) Rubisco, Rubisco activase, and global climate change. J Exp Bot 59:1581–1595
Salvucci ME, Crafts-Brandner SJ (2002) Sensitivity of photosynthesis in a C4 plant, maize, to heat stress. Plant Physiol 129:1773–1780
Salvucci ME, Crafts-Brandner SJ (2004a) Inhibition of photosynthesis by heat stress: the activation state of Rubisco as a limiting factor in photosynthesis. Physiol Plant 120:179–186
Salvucci ME, Crafts-Brandner SJ (2004b) Relationship between the heat tolerance of photosynthesis and the thermal stability of Rubisco activase in plants from contrasting thermal environments. Plant Physiol 134:1460–1470
Schrader SM, Wise RR, Wacholtz WF, Ort DR, Sharkey TD (2004) Thylakoid membrane responses to moderately high leaf temperature in pima cotton. Plant Cell Environ 27:725–735
Sharkey TD (1985) Photosynthesis in intact leaves of C3 plants: physics, physiology, and rate limitations. Bot Rev 51:53–105
Sharkey TD (2005) Effects of moderate heat stress on photosynthesis: importance of thylakoid reactions, Rubisco deactivation, reactive oxygen species, and thermotolerance provided by isoprene. Plant Cell Environ 28:269–277
Singsaas EL, Sharkey TD (1998) The regulation of isoprene emission responses to rapid leaf temperature fluctuations. Plant Cell Environ 21:1181–1188
Slatyer RO (1977) Altitudinal variation in the photosynthetic characteristics of snow gum, Eucalyptus pauciflora Sieb. ex Spreng. IV. Temperature response of four populations grown at different temperatures. Aust J Plant Physiol 4:583–594
Strand Å, Hurry V, Henkes S, Huner N, Gustafsson P, Gardeström P (1997) Development of Arabidopsis thaliana leaves at low temperature releases the suppression of photosynthesis and photosynthetic gene expression despite the accumulation of soluble carbohydrates. Plant J 12:605–614
Strand Å, Hurry V, Henkes S, Huner N, Gustafsson P, Gardeström P, Stitt M (1999) Acclimation of Arabidopsis leaves developing at low temperature: increasing cyto-plasmic volume accompanies increased activities of enzymes in the Calvin cycle and in the sucrose-biosynthesis pathway. Plant Physiol 119:1387–1397
Sung DY, Kaplan F, Lee KJ, Guy CL (2003) Acquired tolerance to temperature extremes. Trends Plant Sci 8:179–187
Tebaldi C, Hayhoe K, Arblaster JM, Meehl GA (2006) Going to the extremes: an intercomparison of model-simulated historical and future changes in extreme events. Clim Change 79:185–211
Terzaghi WB, Fork DC, Berry JA, Field CB (1989) Low and high temperature limits to PSII. A survey using trans-parinaric acid, delayed light emission, and Fo chlorophyll fluorescence. Plant Physiol 91:1494–1500
Vierling E (1991) The roles of heat shock proteins in plants. Annu Rev Plant Physiol Plant Mol Biol 42:579–620
von Caemmerer S (2000) Biochemical models of leaf photosynthesis. CSIRO publishing, Collingwood, pp 1–165
von Caemmerer S, Furbank RT (1999) Modeling C4 photosynthesis. In: Sage RF, Monson RK (eds) C4 plant biology. Academic Press, San Diego, pp 173–211
von Caemmerer S, Quinn V, Hancock NC, Price GD, Furbank RT, Ludwig M (2004) Carbonic anhydrase and C4 photosynthesis: a transgenic analysis. Plant Cell Environ 27:697–703
von Caemmerer S, Hendrickson L, Quinn V, Vella N, Millgate AG, Furbank RT (2005) Reductions of Rubisco activase by antisense RNA in the C4 plant Flaveria bidentis reduces Rubisco carbamylation and leaf photosynthesis. Plant Physiol 137:747–755
von Caemmerer S, Farquhar GD, Berry JA (2009) Biochemical model of C3 photosynthesis. In: Laisk A, Nedbal L, Govindjee (eds) Photosynthesis in silico: understanding complexity from molecules to ecosystems. Springer Science + Business Media B.V., Dordrecht, pp 209–230
Wagner D (1996) Scenarios of extreme temperature events. Clim Change 33:385–407
Wang WX, Vinocur B, Shoseyov O, Altman A (2004) Role of plant heat shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci 5:244–252
Wang D, Naidu SL, Portis AR Jr, Moose SP, Long SP (2008) Can the cold tolerance of C4 photosynthesis in Miscanthus × giganteus relative to Zea mays be explained by differences in activities and thermal properties of Rubisco? J Exp Bot 59:1779–1787
Way DA, Oren R (2010) Differential responses to changes in growth temperature between trees from different functional groups and biomes: a review and synthesis of data. Tree Physiol 30:669–688
Way DA, Pearcy RW (2012) Sunflecks in trees and forests: from photosynthetic physiology to global change biology. Tree Physiol 32:1066–1081
Way DA, Sage RF (2008a) Elevated growth temperatures reduce the carbon gain of black spruce (Picea mariana (Mill.) B.S.P.). Global Change Biol 14:624–636
Way DA, Sage RF (2008b) Thermal acclimation of photosynthesis in black spruce (Picea mariana (Mill.) B.S.P.). Plant Cell Environ 31:1250–1262
Way DA, Yamori W (2013) Thermal acclimation of photosynthesis: on the importance of adjusting our definitions and accounting for thermal acclimation of respiration. Photosynth Res. doi:10.1007/s11120-013-9873-7
Wise RR, Olson AJ, Schrader SM, Sharkey TD (2004) Electron transport is the functional limitation of photosynthesis in field-grown Pima cotton plants at high temperature. Plant Cell Environ 27:717–724
Wright IJ, Reich PB, Cornelissen JHC, Falster DS, Garnier E, Hikosaka K, Lamont BB, Lee W, Oleksyn J, Osada N, Poorter H, Villar R, Warton DI, Westoby M (2005) Assessing the generality of global leaf trait relationships. New Phytol 166:485–496
Yamane Y, Kashino Y, Koike H, Satoh K (1998) Effects of high temperatures on the photosynthetic systems in spinach: oxygen-evolving activities, fluorescence characteristics and the denaturation process. Photosynth Res 57:51–59
Yamori W, von Caemmerer S (2009) Effect of Rubisco activase deficiency on the temperature response of CO2 assimilation rate and Rubisco activation state: insights from transgenic tobacco with reduced amounts of Rubisco activase. Plant Physiol 151:2073–2082
Yamori W, Noguchi K, Terashima I (2005) Temperature acclimation of photosynthesis in spinach leaves: analyses of photosynthetic components and temperature dependencies of photosynthetic partial reactions. Plant Cell Environ 28:536–547
Yamori W, Noguchi K, Hanba YT, Terashima I (2006a) Effects of internal conductance on the temperature dependence of the photosynthetic rate in spinach leaves from contrasting growth temperatures. Plant Cell Physiol 47:1069–1080
Yamori W, Suzuki K, Noguchi K, Nakai M, Terashima I (2006b) Effects of Rubisco kinetics and Rubisco activation state on the temperature dependence of the photosynthetic rate in spinach leaves from contrasting growth temperatures. Plant Cell Environ 29:1659–1670
Yamori W, Noguchi K, Kashino Y, Terashima I (2008) The role of electron transport in determining the temperature dependence of the photosynthetic rate in spinach leaves grown at contrasting temperatures. Plant Cell Physiol 49:583–591
Yamori W, Noguchi K, Hikosaka K, Terashima I (2009) Cold-tolerant crop species have greater temperature homeostasis of leaf respiration and photosynthesis than cold-sensitive species. Plant Cell Physiol 50:203–215
Yamori W, Evans JR, von Caemmerer S (2010a) Effects of growth and measurement light intensities on temperature dependence of CO2 assimilation rate in tobacco leaves. Plant Cell Environ 33:332–343
Yamori W, Noguchi K, Hikosaka K, Terashima I (2010b) Phenotypic plasticity in photosynthetic temperature acclimation among crop species with different cold tolerances. Plant Physiol 152:388–399
Yamori W, Nagai T, Makino A (2011a) The rate-limiting step for CO2 assimilation at different temperatures is influenced by the leaf nitrogen content in several C3 crop species. Plant Cell Environ 34:764–777
Yamori W, Sakata N, Suzuki Y, Shikanai T, Makino A (2011b) Cyclic electron flow around photosystem I via chloroplast NAD(P)H dehydrogenase (NDH) complex performs a significant physiological role during photosynthesis and plant growth at low temperature in rice. Plant J 68:966–976
Yamori W, Takahashi S, Makino A, Price GD, Badger MR, von Caemmerer S (2011c) The roles of ATP synthase and the cytochrome b6/f complexes in limiting chloroplast electron transport and determining photosynthetic capacity. Plant Physiol 155:956–962
Yamori W, Masumoto C, Fukayama H, Makino A (2012) Rubisco activase is a key regulator of non steady-state photosynthesis at any leaf temperature and, to a lesser extent, of steady-state photosynthesis at high temperature. Plant J 71:871–880
Acknowledgments
We thank Professor Susanne von Caemmerer and Professor Ichiro Terashima for valuable comments on the manuscript. The experiment on CAM photosynthesis was performed in Professor von Caemmerer’s laboratory. This work was supported by a grant from the Japan Society for the Promotion Science (Postdoctoral Fellowships to W.Y.) and by grants from NSERC, the US Department of Agriculture (#2011-67003-30222), the US Department of Energy, Terrestrial Ecosystem Sciences (#11-DE-SC-0006967) and the US-Israeli Bi-National Science Foundation to D.A.W. and by a grant from CREST, JST, Japan (K.H.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yamori, W., Hikosaka, K. & Way, D.A. Temperature response of photosynthesis in C3, C4, and CAM plants: temperature acclimation and temperature adaptation. Photosynth Res 119, 101–117 (2014). https://doi.org/10.1007/s11120-013-9874-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-013-9874-6