Abstract
Key message
Light plays a minor role in the within-crown acclimation of physiological and morphological parameters in small loblolly pine trees.
Abstract
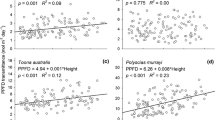
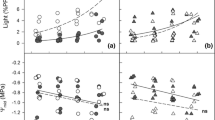
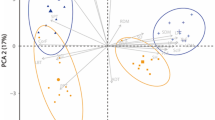
Whether the plasticity of foliage within the crown is driven by light or height is still not well known, especially for small trees. The present study was conducted in two loblolly pine (Pinus taeda L.) stands growing on two sites, during the third year after planting. The following traits were investigated in the lower and upper crowns of the same individuals twice per season (spring, summer, and autumn): photosynthesis (A sat), stomatal conductance (g s), intrinsic water use efficiency (WUEint), nitrogen concentration ***(N), specific leaf area (SLA), photosynthetic nitrogen use efficiency (PNUE), fascicle diameter, and needles per fascicle. The trees were open-grown at the beginning of the study; thus, any vertical gradient of the traits was not assumed to be driven by light. Overall, A sat, g s, PNUE, and SLA tended to be higher in the lower than in the upper crown, whereas N, fascicle diameter, and needles per fascicle were higher in the upper than in the lower crown. However, although all traits were affected by crown position, the magnitude of this effect differed by season. The fact that crown position affected most of the traits in spring and summer, but not in autumn, suggest that height might be the main driver of foliage acclimation. However, considering that crown position affected SLA in autumn but not the photosynthetic rate or N concentration, it is possible that foliar acclimation of these traits was mainly driven by light in this season.



Similar content being viewed by others
References
Ambrose AR, Sillett SC, Dawson TE (2009) Effects of tree height on branch hydraulics, leaf structure and gas exchange in California redwoods. Plant Cell Environ 32:743–757
Aspinwall MJ, King JS, McKeand SE, Domec JC (2011) Leaf-level gas-exchange uniformity and photosynthetic capacity among loblolly pine (Pinus taeda L.) genotypes of contrasting inherent genetic variation. Tree Physiol 31:78–91
Broeckx LS, Fichot R, Verlinden MS, Ceulemans R (2014) Seasonal variations in photosynthesis, intrinsic water-use efficiency and stable isotope composition of poplar leaves in a short-rotation plantation. Tree Physiol 34:701–715
Buckley TN, Cescatti A, Farquhar GD (2013) What does optimization theory actually predict about crown profiles of photosynthetic capacity when models incorporate greater realism? Plant Cell Environ 36:1547–1563
Cavaleri MA, Oberbauer SF, Clark DB, Clark DA, Ryan MG (2010) Height is more important than light in determining leaf morphology in a tropical forest. Ecology 91:1730–1739
Chmura DJ, Tjoelker MG (2008) Leaf traits in relation to crown development, light interception and growth of elite families of loblolly and slash pine. Tree Physiol 28:729–742
Cole KL, Fisher J, Arundel ST, Cannella J, Swift S (2008) Geographical and climatic limits of needle types of one- and two-needled pinyon pines. J Biogeogr 35:257–269
De Pury DGG, Farquhar GD (1997) Simple scaling of photosynthesis from leaves to canopies without the errors of big-leaf models. Plant Cell Environ 20:537–557
Ellsworth DS, Reich PB (1993) Canopy structure and vertical patterns of photosynthesis and related leaf traits in a deciduous forest. Oecologia 96:169–178
Evans JR, Poorter H (2001) Photosynthetic acclimation of plants to growth irradiance: the relative importance of specific leaf area and nitrogen partitioning in maximizing carbon gain. Plant Cell Environ 24:755–767
Fox TR, Jokela EJ, Allen HL (2007) The development of pine plantation silviculture in the southern United States. J Forest 105:337–347
Ginn SE, Seiler JR, Cazell BH, Kreh RE (1991) Physiological and growth responses of eight-year-old loblolly pine stands to thinning. For Sci 37:1030–1040
Gough CM, Seiler JR, Johnsen KH, Sampson DA (2004) Seasonal photosynthesis in fertilized and nonfertilized loblolly pine. For Sci 50:1–9
Gratani L (2014) Plant phenotypic plasticity in response to environmental factors. Adv Bot 2014:1–17
Hubbard RM, Bond BJ, Senock RS, Ryan MG (2002) Effects of branch height on leaf gas exchange, branch hydraulic conductance and branch sap flux in open-grown ponderosa pine. Tree Physiol 22:575–581
Ingwers MW, Urban J, McGuire MA, Bhuiyan RA, Teskey RO (2016) Physiological attributes of three- and four-needle fascicles of loblolly pine (Pinus taeda L.). Trees 30:1923–1933
Ishii HT, Jennings GM, Sillett SC, Koch GW (2008) Hydrostatic constraints on morphological exploitation of light in tall Sequoia sempervirens trees. Oecologia 156:751–763
Kazda M, Salzer J, Reiter I (2000) Photosynthetic capacity in relation to nitrogen in the canopy of a Quercus robur, Fraxinus angustifolia and Tilia cordata flood plain forest. Tree Physiol 20:1029–1037
Kenzo T, Inoue Y, Yoshimura M, Yamashita M, Tanaka-Oda A, Ichie T (2015) Height-related changes in leaf photosynthetic traits in diverse Bornean tropical rain forest trees. Oecologia 177:191–202
Larbi A, Vazquez S, El-Jendoubi H, Msallem M, Abadia J, Abadia A, Morales F (2015) Canopy light heterogeneity drives leaf anatomical, eco-physiological, and photosynthetic changes in olive trees grown in a high-density plantation. Photosynth Res 123:141–155
Le Roux X, Bariac T, Sinoquet H, Genty B, Piel C, Mariotti A, Girardin C, Richard P (2001) Spatial distribution of leaf water-use efficiency and carbon isotope discrimination within an isolated tree crown. Plant Cell Environ 24:1021–1032
Lewis JD, McKane RB, Tingey DT, Beedlow PA (2000) Vertical gradients in photosynthetic light response within an old-growth Douglas-fir and western hemlock canopy. Tree Physiol 20:447–456
Li T, Yang Q (2015) Advantages of diffuse light for horticultural production and perspectives for further research. Front Plant Sci 6:704
Li T, Heuvelink E, Dueck TA, Janse J, Gort G, Marcelis LFM (2014) Enhancement of crop photosynthesis by diffuse light: quantifying the contributing factors. Ann Bot 114:146–156
Limousin JM, Misson L, Lavoir AV, Martin NK, Rambal S (2010) Do photosynthetic limitations of evergreen Quercus ilex leaves change with long-term increased drought severity? Plant Cell Environ 33:863–875
Maier CA, Johnsen KH, Butnor J, Kress LW, Anderson PH (2002) Branch growth and gas exchange in 13-year-old loblolly pine (Pinus taeda) trees in response to elevated carbon dioxide concentration and fertilization. Tree Physiol 22:1093–1106
Marshall JD, Monserud RA (2003) Foliage height influences specific leaf area of three conifer species. Can J For Res 33:164–170
McGarvey RC, Martin TA, White TL (2004) Integrating within-crown variation in net photosynthesis in loblolly and slash pine families. Tree Physiol 24:1209–1220
Meinzer FC, Bond BJ, Karanian JA (2008) Biophysical constraints on leaf expansion in a tall conifer. Tree Physiol 28:197–206
Misson L, Limousin JM, Rodriguez R, Letts MG (2010) Leaf physiological responses to extreme droughts in Mediterranean Quercus ilex forest. Plant Cell Environ 33:1898–1910
Munger GT, Will RE, Borders BE (2003) Effects of competition control and annual nitrogen fertilization on gas exchange of different-aged Pinus taeda. Can J For Res 33:1076–1083
Niinemets U (2007) Photosynthesis and resource distribution through plant canopies. Plant Cell Environ 30:1052–1071
Niinemets U, Keenan TF, Hallik L (2015) A worldwide analysis of within-canopy variations in leaf structural, chemical and physiological traits across plant functional types. New Phytol 205:973–993
Oldham AR, Sillett SC, Tomescu AM, Koch GW (2010) The hydrostatic gradient, not light availability, drives height-related variation in Sequoia sempervirens (Cupressaceae) leaf anatomy. Am J Bot 97:1087–1097
Palardy SG (2008) Physiology of woody plants. Academic Press, San Diego
Poorter H, Niinemets Ü, Poorter L, Wright IJ, Villar R (2009) Causes and consequences of variation in leaf mass per area (LMA): a meta-analysis. New Phytol 182:565–588
Sellin A, Kupper P (2004) Within-crown variation in leaf conductance of Norway spruce: effects of irradiance, vapour pressure deficit, leaf water status and plant hydraulic constraints. Ann For Sci 61:419–429
Shiraki A, Azuma W, Kuroda K, Ishii HR (2016) Physiological and morphological acclimation to height in cupressoid leaves of 100-year-old Chamaecyparis obtusa. Tree Physiol 36:1–10
Tang Z, Chambers JL, Sword MA, Barnett JP (2003) Seasonal photosynthesis and water relations of juvenile loblolly pine relative to stand density and canopy position. Trees Struct Funct 17:424–430
Tarvainen L, Räntfors M, Wallin G (2015) Seasonal and within-canopy variation in shoot-scale resource-use efficiency trade-offs in a Norway spruce stand. Plant Cell Environ 38:2487–2496
Tyree MC, Seiler JR, Maier CA, Johnsen KH (2009) Pinus taeda clones and soil nutrient availability: effects of soil organic matter incorporation and fertilization on biomass partitioning and leaf physiology. Tree Physiol 29:1117–1131
Wang X, Zhou H, Han F, Li Y, Hu D (2016) Dynamic spatial patterns of leaf traits affect total respiration on the crown scale. Sci Rep 6:1–12
Woodruff DR, Bond BJ, Meinzer FC (2004) Does turgor limit growth in tall trees? Plant Cell Environ 27:229–236
Wyka TP, Oleksyn J, Zytkowiak R, Karolewski P, Jagodzinski AM, Reich PB (2012) Responses of leaf structure and photosynthetic properties to intra-canopy light gradients: a common garden test with four broadleaf deciduous angiosperm and seven evergreen conifer tree species. Oecologia 170:11–24
Yáñez MA, Fox TR, Seiler JR (2015) Early growth responses of loblolly pine varieties and families to silvicultural intensity. For Ecol Manag 356:204–215
Yáñez MA, Seiler JR, Fox TR (2017) Crown physiological responses of loblolly pine clones and families to silvicultural intensity: assessing the effect of crown ideotype. For Ecol Manag 398:25–36
Zhang S, Allen HL (1996) Foliar nutrient dynamics of 11-year-old loblolly pine (Pinus taeda) following nitrogen fertilization. Can J For Res 26:1426–1439
Zhang Y, Equiza MA, Zheng Q, Tyree MT (2012a) Factors controlling plasticity of leaf morphology in Robinia pseudoacacia L. II: the impact of water stress on leaf morphology of seedlings grown in a controlled environment chamber. Ann For Sci 69:39–47
Zhang Y, Zheng Q, Tyree MT (2012b) Factors controlling plasticity of leaf morphology in Robinia pseudoacacia L. I: height-associated variation in leaf structure. Ann For Sci 69:29–37
Acknowledgements
This research was funded through the NSF Center for Advanced Forestry Systems (CAFS) and the Forest Productivity Cooperative (FPC). Funding for this work was also provided in part by the Virginia Agricultural Experiment Station and the Program McIntire Stennis of the National Institute of Food and Agriculture, US Department of Agriculture. We also thank John Peterson and Samuel Frye for their help in the field work, Timothy Albaugh for providing the meteorological data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by G. Wieser.
Rights and permissions
About this article
Cite this article
Yáñez, M.A., Fox, T.R., Seiler, J.R. et al. Within-crown acclimation of leaf-level physiological and morphological parameters in young loblolly pine stands. Trees 31, 1849–1857 (2017). https://doi.org/10.1007/s00468-017-1589-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-017-1589-4