Summary
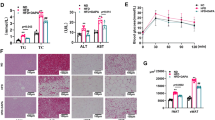
An adipose tissue engineering chamber model of spontaneous adipose tissue generation from an existing fat flap has been described. However, the chamber does not completely fill with adipose tissue in this model. Here, the effect of adipose tissue extract (ATE) on adipose tissue regeneration was investigated. In vitro, the adipogenic and angiogenic capacities of ATE were evaluated using Oil Red O and tube formation assays on adipose-derived stem cells (ASCs) and rat aortic endothelial cells (RAECs), respectively. In vivo, saline or ATE was injected into the adipose tissue engineering chamber 1 week after its implantation. At different time points post-injection, the contents were morphometrically, histologically, and immunohistochemically evaluated, and the expression of growth factors and adipogenic genes was analyzed by enzyme-linked immunosorbent assay (ELISA) and quantitative real-time PCR. With the exception of the baseline control group, in which fat flaps were not inserted into a chamber, the total volume of fat flap tissue increased significantly in all groups, especially in the ATE group. Better morphology and structure, a thinner capsule, and more vessels were observed in the ATE group than in the control group. Expression of angiogenic growth factors and adipogenic markers were significantly higher in the ATE group. ATE therefore significantly promoted adipose tissue regeneration and reduced capsule formation in an adipose tissue engineering chamber model. These data suggest that ATE provides a more angiogenic and adipogenic microenvironment for adipose tissue formation by releasing various cytokines and growth factors that also inhibit capsule formation.









Similar content being viewed by others
Abbreviations
- ATE:
-
Adipose tissue extract
- ASCs:
-
Adipose-derived stem cells
- RAECs:
-
Rabbit aortic endothelial cells
- bFGF:
-
Basic fibroblast growth factor
- H&E:
-
Hematoxylin and eosin
- PBS:
-
Phosphate-buffered saline
- PPARγ:
-
Proliferator-activated receptor gamma γ
- VEGF:
-
Vascular endothelial growth factor
References
Carmeliet P, Jain RK (2011) Molecular mechanisms and clinical applications of angiogenesis. Nature 473(7347):298–307
Chiou M, Xu Y, Longaker MT (2006) Mitogenic and chondrogenic effects of fibroblast growth factor-2 in adipose-derived mesenchymal cells. Biochem Biophys Res Commun 343(2):644–652
Deonarine K, Panelli MC, Stashower ME, Jin P, Smith K, Slade HB, Norwood C, Wang E, Marincola FM, Stroncek DF (2007) Gene expression profiling of cutaneous wound healing. J Transl Med 5:11
Desai VD, Hsia HC, Schwarzbauer JE (2014) Reversible modulation of myofibroblast differentiation in adipose-derived mesenchymal stem cells. PLoS ONE 9(1), e86865
Dolderer JH, Abberton KM, Thompson EW, Slavin JL, Stevens GW, Penington AJ, Morrison WA (2007) Spontaneous large volume adipose tissue generation from a vascularized pedicled fat flap inside a chamber space. Tissue Eng 13(4):673–681
Dvorak HF, Dvorak AM, Manseau EJ, Wiberg L, Churchill WH (1979) Fibrin gel investment associated with line 1 and line 10 solid tumor growth, angiogenesis, and fibroplasia in guinea pigs. Role of cellular immunity, myofibroblasts, microvascular damage, and infarction in line 1 tumor regression. J Natl Cancer Inst 62(6):1459–1472
Findlay MW, Dolderer JH, Trost N, Craft RO, Cao Y, Cooper-White J, Stevens G, Morrison WA (2011) Tissue-engineered breast reconstruction: bridging the gap toward large-volume tissue engineering in humans. Plast Reconstr Surg 128(6):1206–1215
Hausman GJ (1985) Nucleoside phosphatase and nucleotide tetrazolium reductase as markers of arteriolar differentiation in fetal pig tissue. Histochemistry 83(2):121–126
Hausman GJ, Thomas GB (1984a) The development of the inner layer of backfat in fetal and young pigs. J Anim Sci 58(6):1550–1560
Hausman GJ, Thomas GB (1984b) Histochemical and cellular aspects of adipose tissue development in decapitated pig fetuses: an ontogeny study. J Anim Sci 58(6):1540–1549
Hausman GJ, Wright JT, Thomas GB (1991) Vascular and cellular development in fetal adipose tissue: lectin binding studies and immunocytochemistry for laminin and type IV collagen. Microvasc Res 41(1):111–125
Maltseva O, Folger P, Zekaria D, Petridou S, Masur SK (2001) Fibroblast growth factor reversal of the corneal myofibroblast phenotype. Invest Ophthalmol Vis Sci 42(11):2490–2495
Peng Z, Dong Z, Chang Q, Zhan W, Zeng Z, Zhang S, Lu F (2014) Tissue engineering chamber promotes adipose tissue regeneration in adipose tissue engineering models through induced aseptic inflammation. Tissue Eng Part C 20(11):875–885
Prantl L, Schreml S, Fichtner-Feigl S, Poppl N, Eisenmann-Klein M, Schwarze H, Fuchtmeier B (2007) Clinical and morphological conditions in capsular contracture formed around silicone breast implants. Plast Reconstr Surg 120(1):275–284
Roca GB, Graf R, da Silva Freitas R, Salles G Jr, Francisco JC, Noronha L, Maluf I Jr (2014) Autologous Fat Grafting for Treatment of Breast Implant Capsular Contracture: A Study in Pigs. Aesthet Surg J 34(5):769–775
Sarkanen JR, Kaila V, Mannerstrom B, Raty S, Kuokkanen H, Miettinen S, Ylikomi T (2012a) Human adipose tissue extract induces angiogenesis and adipogenesis in vitro. Tissue Eng A 18(1–2):17–25
Sarkanen JR, Ruusuvuori P, Kuokkanen H, Paavonen T, Ylikomi T (2012b) Bioactive acellular implant induces angiogenesis and adipogenesis and sustained soft tissue restoration in vivo. Tissue Eng A 18(23–24):2568–2580
Schweigerer L, Neufeld G, Friedman J, Abraham JA, Fiddes JC, Gospodarowicz D (1987) Capillary endothelial cells express basic fibroblast growth factor, a mitogen that promotes their own growth. Nature 325(6101):257–259
Shah Z, Lehman JA Jr, Tan J (1981) Does infection play a role in breast capsular contracture? Plast Reconstr Surg 68(1):34–42
Shaw TJ, Martin P (2009) Wound repair at a glance. J Cell Sci 122(Pt 18):3209–3213
Szasz T, Webb RC (2012) Perivascular adipose tissue: more than just structural support. Clin Sci (Lond) 122(1):1–12
Yamazaki Y, Morita T (2006) Molecular and functional diversity of vascular endothelial growth factors. Mol Divers 10(4):515–527
Zhu M, Dong Z, Gao J, Liao Y, Xue J, Yuan Y, Liu L, Chang Q, Lu F (2015) Adipocyte regeneration after free fat transplantation: promotion by stromal vascular fraction cells. Cell Transplant 24(1):49–62
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This work was supported by the National Nature Science Foundation of China (81471881, 81372083, 81171834, and 81201482), the Key Clinical Specialty Discipline Construction Program, and the Health Collaborative Innovation major projects of Guangzhou (7414275040815).
Additional information
Zijing Lu and Yi Yuan contributed equally to this work.
Rights and permissions
About this article
Cite this article
Lu, Z., Yuan, Y., Gao, J. et al. Adipose tissue extract promotes adipose tissue regeneration in an adipose tissue engineering chamber model. Cell Tissue Res 364, 289–298 (2016). https://doi.org/10.1007/s00441-015-2322-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-015-2322-5