Abstract
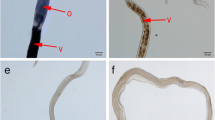
The Vasa gene is a vital germline marker to study the origin and development of germ cells and gonads in many organisms. Until now, little information was available about the characteristics of the Vasa gene in Schistosoma japonicum (S. japonicum). In this study, we cloned the open reading frame (ORF) of the S. japonicum Vasa-like gene (Sj-Vasa). The expression pattern and tissue localization of Sj-Vasa were also analyzed. Our results showed that Sj-Vasa shared the general feature of DEAD-box family member proteins. Sj-Vasa was transcribed and expressed throughout the S. japonicum life cycle with transcription exhibiting high levels at day 24 in both male and female worms, and the expression level in the female was always higher than that in the male. Sj-Vasa protein was localized in a variety of tissues of adult schistosomes, including the gonads (ovary, vitellarium, and testes), the subtegument, and some cells of the parenchyma. To our knowledge, this is the first report of preliminary characterization and expression of the Vasa-like gene that may play an important role in the development of the worm, especially in reproductive organs of S. japonicum.






Similar content being viewed by others
References
Andrade LF, Mourao Mde M, Geraldo JA, Coelho FS, Silva LL, Neves RH, Volpini A, Machado-Silva JR, Araujo N, Nacif-Pimenta R, Caffrey CR, Oliveira G (2014) Regulation of Schistosoma mansoni development and reproduction by the mitogen-activated protein kinase signaling pathway. PLoS Negl Trop Dis 8, e2949
Cogswell AA, Kommer VP, Williams DL (2012) Transcriptional analysis of a unique set of genes involved in Schistosoma mansoni female reproductive biology. PLoS Negl Trop Dis 6(11):e1907
Collins JJ 3rd, Wang B, Lambrus BG, Tharp ME, Iyer H, Newmark PA (2013) Adult somatic stem cells in the human parasite Schistosoma mansoni. Nature 494:476–479
Costache AD, Pullela PK, Kasha P, Tomasiewicz H, Sem DS (2005) Homology-modeled ligand-binding domains of zebrafish estrogen receptors alpha, beta1, and beta2: from in silico to in vivo studies of estrogen interactions in Danio rerio as a model system. Mol Endocrinol 19:2979–2990
Dearden PK (2006) Germ cell development in the Honeybee (Apis mellifera); vasa and nanos expression. BMC Dev Biol 6:6
Doenhoff MJ, Pica-Mattoccia L (2006) Praziquantel for the treatment of schistosomiasis: its use for control in areas with endemic disease and prospects for drug resistance. Exp Rev Anti Infect Ther 4:199–210
Doenhoff MJ, Cioli D, Utzinger J (2008) Praziquantel: mechanisms of action, resistance and new derivatives for schistosomiasis. Curr Opin Infect Dis 21:659–667
Fujiwara Y, Komiya T, Kawabata H, Sato M, Fujimoto H, Furusawa M, Noce T (1994) Isolation of a DEAD-family protein gene that encodes a murine homolog of Drosophila vasa and its specific expression in germ cell lineage. Proc Natl Acad Sci U S A 91:12258–12262
Galanti SE, Huang SC, Pearce EJ (2012) Cell death and reproductive regression in female Schistosoma mansoni. PLoS Negl Trop Dis 6, e1509
Gruidl ME, Smith PA, Kuznicki KA, McCrone JS, Kirchner J, Roussell DL, Strome S, Bennett KL (1996) Multiple potential germ-line helicases are components of the germ-line-specific P granules of Caenorhabditis elegans. Proc Natl Acad Sci U S A 93:13837–13842
Gustafson EA, Wessel GM (2010) Vasa genes: emerging roles in the germ line and in multipotent cells. Bioessays 32:626–637
Gustafson EA, Yajima M, Juliano CE, Wessel GM (2011) Post-translational regulation by gustavus contributes to selective Vasa protein accumulation in multipotent cells during embryogenesis. Dev Biol 349:440–450
Hay B, Jan LY, Jan YN (1990) Localization of vasa, a component of Drosophila polar granules, in maternal-effect mutants that alter embryonic anteroposterior polarity. Development 109:425–433
He YX, Gong ZX, Ma JX (1980) Scanning and transmission electron microscopy of Schistosoma japonicum egg shell. Chin Med J (Engl) 93:861–864
Ikenishi K, Tanaka TS (2000) Spatio-temporal expression of Xenopus vasa homolog, XVLG1, in oocytes and embryos: the presence of XVLG1 RNA in somatic cells as well as germline cells. Dev Growth Differ 42:95–103
Kunz W (2001) Schistosome male–female interaction: induction of germ-cell differentiation. Trends Parasitol 17:227–231
Leutner S, Oliveira KC, Rotter B, Beckmann S, Buro C, Hahnel S, Kitajima JP, Verjovski-Almeida S, Winter P, Grevelding CG (2013) Combinatory microarray and SuperSAGE analyses identify pairing-dependently transcribed genes in Schistosoma mansoni males, including follistatin. PLoS Negl Trop Dis 7, e2532
Li SZ, Zheng H, Gao J, Zhang LJ, Zhu R, Xu J, Guo JG, Xiao N, Zhou XN (2013) Endemic status of schistosomiasis in People’s Republic of China in 2012. Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi 25:557–563
Liu M, Wang X, Lei L, Zhao Z, Shen J (2010) The identification, expression profile, and preliminary characterization of Tsunagi protein from Schistosoma japonicum. Parasitol Res 107:615–621
Liu S, Cai P, Hou N, Piao X, Wang H, Hung T, Chen Q (2012) Genome-wide identification and characterization of a panel of house-keeping genes in Schistosoma japonicum. Mol Biochem Parasitol 182:75–82
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods 25:402–408
LoVerde PT, Hirai H, Merrick JM, Lee NH, El-Sayed N (2004) Schistosoma mansoni genome project: an update. Parasitol Int 53:183–192
LoVerde PT, Andrade LF, Oliveira G (2009) Signal transduction regulates schistosome reproductive biology. Curr Opin Microbiol 12:422–428
Magalhaes LG, de Castro-Borges W, de Souza GM, Guerra-Sa R, Rodrigues V (2009) Molecular cloning, sequencing, and expression analysis of presenilin cDNA from Schistosoma mansoni. Parasitol Res 106:7–13
Mann VH, Suttiprapa S, Skinner DE, Brindley PJ, Rinaldi G (2014) Pseudotyped murine leukemia virus for schistosome transgenesis: approaches, methods and perspectives. Transgenic Res 23:539–556
Mochizuki K, Nishimiya-Fujisawa C, Fujisawa T (2001) Universal occurrence of the vasa-related genes among metazoans and their germline expression in Hydra. Dev Genes Evol 211:299–308
Olsen LC, Aasland R, Fjose A (1997) A vasa-like gene in zebrafish identifies putative primordial germ cells. Mech Dev 66:95–105
Oyama A, Shimizu T (2007) Transient occurrence of vasa-expressing cells in nongenital segments during embryonic development in the oligochaete annelid Tubifex tubifex. Dev Genes Evol 217:675–690
Pfister D, de Mulder K, Hartenstein V, Kuales G, Borgonie G, Marx F, Morris J, Ladurner P (2008) Flatworm stem cells and the germ line: developmental and evolutionary implications of macvasa expression in Macrostomum lignano. Dev Biol 319:146–159
Rebscher N, Volk C, Teo R, Plickert G (2008) The germ plasm component Vasa allows tracing of the interstitial stem cells in the cnidarian Hydractinia echinata. Dev Dyn : Off Publ Am Assoc Anatomists 237:1736–1745
Ren CP, Zhang P, Zhang WN, Huang DK, Jia XM, Gui L, Liu M, Shen JJ (2013) Schistosoma japonicum: Tsunagi/Y14 protein plays a critical role in the development of the reproductive organs and eggs. Exp Parasitol 135:430–436
Rosner A, Rinkevich B (2007) The DDX3 subfamily of the DEAD box helicases: divergent roles as unveiled by studying different organisms and in vitro assays. Curr Med Chem 14:2517–2525
Roussell DL, Bennett KL (1993) glh-1, a germ-line putative RNA helicase from Caenorhabditis, has four zinc fingers. Proc Natl Acad Sci U S A 90:9300–9304
Sano H, Nakamura A, Kobayashi S (2002) Identification of a transcriptional regulatory region for germline-specific expression of vasa gene in Drosophila melanogaster. Mech Dev 112:129–139
Schupbach T, Wieschaus E (1986) Germline autonomy of maternal-effect mutations altering the embryonic body pattern of Drosophila. Dev Biol 113:443–448
Skinner DE, Rinaldi G, Suttiprapa S, Mann VH, Smircich P, Cogswell AA, Williams DL, Brindley PJ (2012) Vasa-Like DEAD-box RNA helicases of Schistosoma mansoni. PLoS Negl Trop Dis 6, e1686
Updike DL, Hachey SJ, Kreher J, Strome S (2011) P granules extend the nuclear pore complex environment in the C. elegans germ line. J Cell Biol 192:939–948
Vanderstraete M, Gouignard N, Cailliau K, Morel M, Hahnel S, Leutner S, Beckmann S, Grevelding CG, Dissous C (2014) Venus kinase receptors control reproduction in the platyhelminth parasite Schistosoma mansoni. PLoS Pathog 10, e1004138
Wang X, Li H, Qi X, Shi Y, Xia Y, Yang J, Yuan C, Feng X, Lin J (2011) Characterization and expression of a novel Frizzled 9 gene in Schistosoma japonicum. Gene Expr Patterns : GEP 11:263–270
Wang KG, Zeng QR, Zhang YK, Zhou J, Cai LT, Liang Y, Liu Y (2012) Preparation of agar-paraffin double-embedded longitudinal sections of Schistosoma japonicum. Zhongguo ji sheng chong xue yu ji sheng chong bing za zhi = Chin J Parasitol Parasitic Dis 30:415–417
WHO (2013) Schistosomiasis WHO Media Centre, Fact sheet Nu115 Available: http://www.who.int/mediacentre/factsheets/fs115/en/
Wilson MS, Mentink-Kane MM, Pesce JT, Ramalingam TR, Thompson R, Wynn TA (2007) Immunopathology of schistosomiasis. Immunol Cell Biol 85:148–154
Wolke U, Weidinger G, Koprunner M, Raz E (2002) Multiple levels of posttranscriptional control lead to germ line-specific gene expression in the zebrafish. Curr Biol: CB 12:289–294
Wu HR, Chen YT, Su YH, Luo YJ, Holland LZ, Yu JK (2011) Asymmetric localization of germline markers Vasa and Nanos during early development in the amphioxus Branchiostoma floridae. Dev Biol 353:147–159
Xu J, Ren Y, Xu X, Chen J, Li Y, Gan W, Zhang Z, Zhan H, Hu X (2014) Schistosoma japonicum tegumental protein 20.8, role in reproduction through its calcium binding ability. Parasitol Res 113:491–497
Yajima M, Wessel GM (2011) The multiple hats of Vasa: its functions in the germline and in cell cycle progression. Mol Reprod Dev 78:861–867
Zhou SH, Wang SP (2007) A simplified method for rapid isolation of Schistosoma japonicum immature eggs. Chin J Schisto Control 19:128–130
Acknowledgments
This work was supported by the Natural Science Foundation of China (no. 81271815) and Scientific Research of BSKY (XJ201321) from Anhui Medical University. We thank Dr. Lili Xu (Anhui Huaqi Biotech Engineering, LLC) for modifying the manuscript. And we thank Fei Yang, Haiyan Cheng, and Dake Huang in our laboratory for their generous assistance during the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Diao, Y., Hua, M., Shao, Y. et al. Preliminary characterization and expression of Vasa-like gene in Schistosoma japonicum . Parasitol Res 114, 2679–2687 (2015). https://doi.org/10.1007/s00436-015-4473-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4473-4