Abstract
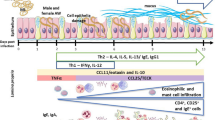
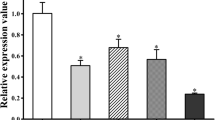
The migratory stage of Trichinella spiralis, the newborn larva (NBL), travels along the pulmonary microvascular system on its way to the skeletal muscle cells. The present work studies the capability of lung cells to kill NBL. For this purpose, in vitro cytotoxicity assays were performed using NBL, lung cell suspensions from Wistar rats, rat anti-NBL surface sera, and fresh serum as complement source. The cytotoxic activity of lung cells from rats infected on day 6 p.i. was compared with that from noninfected rats. Two and 20 h-old NBL (NBL2 and NBL20) were used as they had shown to exhibit different surface antigens altering their biological activity. Sera antibodies were analyzed by indirect immunofluorescence assay, and cell populations used in each assay were characterized by histological staining. The role of IgE in the cytotoxic attack against NBL was analyzed using heated serum. The FcεRI expression on cell suspensions was examined by flow cytometry. Results showed that lung cells were capable of killing NBL by antibody-dependent cell-mediated cytotoxicity (ADCC). Lung cells from infected animals yielded the highest mortality percentages of NBL, with NBL20 being the most susceptible to such attack. IgE yielded a critical role in the cytotoxic attack. Regarding the analysis of cell suspensions, cells from infected rats showed an increase in the percentage of eosinophils, neutrophils, and the number of cells expressing the FcεRI receptor. We conclude that lung cells are capable of killing NBL in the presence of specific antibodies, supporting the idea that the lung is one of the sites where the NBL death occurs due to ADCC.



Similar content being viewed by others
References
Bass DA, Szejda P (1979) Eosinophils versus neutrophils in host defense. Killing of newborn larvae of Trichinella spiralis by human granulocytes in vitro. J Clin Invest 64:1415–1422
Binaghi RA, Perrudet-Badoux A, Bousac-Aron Y (1981) Mechanisms of immune defense against Trichinella spiralis newborn larvae. In: Kim CW, Ruitenberg EJ, Teppema JS (eds) Trichinellosis V. Reedbooks, Chertsey pp 85-89
Bruschi F, Solfanelli S, Binaghi RA (1992) Trichinella spiralis: modifications of the cuticle of the newborn larva during passage through the lung. Exp Parasitol 75:1–9
Burke ML, McGarvey L, McSorley HJ, Bielefeldt-Ohmann H, McManus DP, Gobert GN (2011) Migrating Schistosoma japonicum schistosomula induce an innate immune response and wound healing in the murine lung. Mol Immunol 49:191–200
Capron M, Bazin H, Joseph M, Capron A (1981) Evidence for IgE-dependent cytotoxicity by rat eosinophils. J Immunol 126:1764–1768
Dennis D, Despommier DD, David N (1970) Infectivity of newborn larvae of Trichinella spiralis in the rat. J Parasitol 56:974–977
Despommier DD, Gwadz RW, Hotez PJ, Knirsch CA (2005) The nematodes. In: Despommier DD, Gwadz RW, Hotez PJ, Knirsch CA (eds) Parasitic diseases. Apple Trees Productions L.L.C, New York, pp 105–174
Dombrowicz D, Quatannens B, Papin JP, Capron A, Capron M (2000) Expression of a functional FceRI on rat eosinophils and macrophages. J Immunol 165:1266–1271
Gansmuller A, Anteunis A, Venturiello SM, Bruschi F, Binaghi RA (1987) Antibody-dependent in-vitro cytotoxicity of newborn Trichinella spiralis larvae: nature of the cells involved. Parasite Immunol 9:281–292
Gentilini MV, Nuñez GG, Roux ME, Venturiello SM (2011) Trichinella spiralis infection rapidly induces lung inflammatory response: the lung as the site of helminthocytotoxic activity. Immunobiology 216:1054–1063
Gounni AS, Lamkhioued B, Ochiai K, Tanaka Y, Delaporte E, Capron A, Kinet JP, Capron M (1994) High-affinity IgE receptor on eosinophils is involved in defence against parasites. Nature 367:183–186
Harley JP, Gallicchio V (1971) Trichinella spiralis: migration of larvae in the rat. Exp Parasitol 30:11–21
Ishizaka K, Ishizaka T, Menzel AEO (1967) Physicochemical properties of reaginic antibody. VI. Effect of heat on γE-, γG and γA- antibodies in the sera of ragweed sensitive patients. J Immunol 99(3):610–618
Jungery M, Clark NW, Parkhouse RM (1983) A major change in surface antigens during the maturation of newborn larvae of Trichinella spiralis. Mol Biochem Parasitol 7:101–109
Kazura JW, Grove DI (1978) Stage-specific antibody-dependent eosinophil mediated destruction of Trichinella spiralis. Nature 274:588–589
Mehta K, Sindhu RK, Subrahmanyam D, Nelson DS (1980) IgE-dependent adherence and cytotoxicity of rat spleen and peritoneal cells to Litomosoides carinii microfilariae. Clin Exp Immunol 41:107–114
Moskwa B (1999) Trichinella spiralis: in vitro cytotoxicity of peritoneal cells against synchronous newborn larvae of different age. Parasitol Res 85:59–63
Nöckler K, Kapel CMO (2007) Detection and surveillance for Trichinella: meat inspection and hygiene, and legislation. In: Dupouy-Camet J, Murrell KD (eds) FAO/WHO/OIE Guidelines for the surveillance, management, prevention and control of trichinellosis. pp 69-98
Nuñez GG, Gentile T, Calcagno ML, Venturiello SM (2002) Increased parasiticide activity against Trichinella spiralis newborn larvae during pregnancy. Parasitol Res 88:661–667
Pabst R, Durak D, Roos A, Luhrmann A, Tschernig T (2008) TLR2/6 stimulation of the rat lung: effects on lymphocyte subsets, natural killer cells and dendritic cells in different parts of the air-conducting compartments and at different ages. Immunology 126:132–139
Reece JJ, Siracusa MC, Scott AL (2006) Innate immune responses to lung-stage helminth infection induce alternatively activated alveolar macrophages. Infect Immun 74:4970–4981
Silveira MR, Nunes KP, Cara DC, Souza DG, Corrêa A Jr, Teixeira MM, Negrão-Corrêa D (2002) Infection with Strongyloides venezuelensis induces transient airway eosinophilic inflammation, an increase in immunoglobulin E, and hyper-responsiveness in rats. Infect Immun 70:6263–6272
Siracusa MC, Reece JJ, Urban JF Jr, Scott AL (2008) Dynamics of lung macrophage activation in response to helminth infection. J Leukoc Biol 84:1422–1433
Venturiello SM, Giambartolomei GH, Costantino SN (1993) Immune killing of newborn Trichinella larvae by human leucocytes. Parasite Immunol 15:559–564
Venturiello SM, Giambartolomei GH, Costantino SN (1995) Immune cytotoxic activity of human eosinophils against Trichinella spiralis newborn larvae. Parasite Immunol 17:555–559
Venturiello SM, Verzoletti ML, Costantino SN, Forastiero MA, Roux ME (2007) Early pulmonary response in rats infected with Trichinella spiralis. Parasitology 134:281–288
Wang CH, Bell RG (1986) Trichinella spiralis: vascular recirculation and organ retention of newborn larvae in rats. Exp Parasitol 62:430–441
Wang CH, Bell RG (1988) Antibody-mediated in-vivo cytotoxicity to Trichinella spiralis newborn larvae in immune rats. Parasite Immunol 10:293–308
Acknowledgments
This work was supported by grants from the National Council of Scientific and Technical Research (CONICET, PIP 0973) and the University of Buenos Aires (UBACyT 20020100100923).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Falduto, G.H., Vila, C.C., Saracino, M.P. et al. Trichinella spiralis: killing of newborn larvae by lung cells. Parasitol Res 114, 679–685 (2015). https://doi.org/10.1007/s00436-014-4233-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-014-4233-x