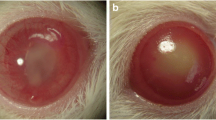
Abstract
This study evaluated the performance of direct amplification of Acanthamoeba-DNA bypassing DNA extraction in the diagnosis of Acanthamoeba keratitis in clinically suspected cases in comparison to direct microscopic examination and in vitro culture. Corneal scrapings were collected from 110 patients who were clinically suspected to have Acanthamoeba keratitis, 63 contact lens wearers (CLW), and 47 non-contact lens wearers (NCLW). Taken samples were subjected to direct microscopic examination, cultivation onto the non-nutrient agar plate surface seeded with Escherichia coli, and PCR amplification. The diagnostic performance of these methods was statistically compared. The results showed that Acanthamoeba infection was detected in 21 (19.1 %) of clinically suspected cases (110); 17 (81 %) of them were CLW and the remaining 4 (19 %) positive cases were NCLW. Regarding the used diagnostic methods, it was found that direct amplification of Acanthamoeba DNA bypassing nucleic acid extraction was superior to microscopy and culture in which 21 cases (19.1 %) were positive for Acanthamoeba by PCR compared to 19 positive cases by culture (17.3 %) and one case (0.9 %) by direct smear. The difference in detection rates between culture and direct smear was highly statistically significant (P = 0.001). On the other hand, there was no significant difference in detection rates between culture and PCR (P = 0.86). On using culture as the gold standard, PCR showed three false-positive samples that were negative by culture and one false-negative sample that was positive by culture. At the same time, direct smear showed 18 false-negative samples. The sensitivity, specificity, positive predictive value, negative predictive value, and diagnostic accuracy of PCR were 94.7, 96.7, 85.7, 98.9, and 96.4, respectively, while those of direct smear were 5.3, 100, 100, 83.5, and 83.6, respectively. In conclusion, direct amplification of Acanthamoeba-DNA bypassing DNA extraction is a reliable, specific, sensitive method in the diagnosis of Acanthamoeba keratitis in clinically suspected cases. It should set up in ophthalmological centers as an easy diagnostic tool.

Similar content being viewed by others
References
Boggild AK, Martin DS, Lee TYL, Yu B, Low DE (2009) Laboratory diagnosis of amoebic keratitis: comparison of four diagnostic methods for different types of clinical specimens. J Clin Microbiol 47(5):1314–1318
Clarke DW, Niederkorn JY (2006) The pathophysiology of Acanthamoeba keratitis. Trends Parasitol 22(4):175–180
El-Sayed NM, Ismail KA, Ahmed SAG, Hetta MH (2012) In vitro amoebicidal activity of ethanol extracts of Arachis hypogaea L., Curcuma longa L. and Pancratium maritimum L. on Acanthamoeba castellanii cysts. Parasitol Res 110(5):1985–1992
Gatti S, Rama P, Matuska S, Berrilli F, Cavallero A, Carletti S, Bruno A, Maserati R, Di Cave D (2010) Isolation and genotyping of Acanthamoeba strains from corneal infections in Italy. J Med Microbiol 59:1324–1330
Goldschmidt P, Rostane H, Saint-Jean C, Batellier L, Alouch C, Zito E, Bourcier T, Laroche L, Chaumeil C (2006) Effects of topical anesthetics and fluorescein on the real-time PCR used for the diagnosis of Herpes viruses and Acanthamoeba keratitis. Br J Ophthalmol 90(11):1354–1356
Goldschmidt P, Degorge S, Saint-Jean C, Year H, Zekhnini F, Batellier L, Laroche L, Chaumeil C (2008) Resistance of Acanthamoeba to classic DNA extraction methods used for the diagnosis of corneal infections. Br J Ophtalmol 92(1):112–115
Goldschmidt P, Degorge S, Benallaoua D, Saint-Jean C, Batellier L, Alouch C, Laroche L, Chaumeil C (2009) New tool for the simultaneous detection of 10 different genotypes of Acanthamoeba available from the American type culture collection. Br J Ophthalmol 93:1096–1100
Gupta S, Aher A (2009) Acanthamoeba keratitis—a case report. People’s J Sci Res 2(2):9–11
Hammersmith KM (2006) iagnosis and management of Acanthamoeba keratitis. Curr Opin Ophthalmol 17(4):327–331
Ibrahim YW, Boase DL, Cree IA (2009) How could contact lens wearers be at risk of Acanthamoeba infection? A review. J Optom 2(2):60–66
Init I, Lau YL, Arin-Fadzlun A, Foead AI, Neilson RS, Nissapatorn V (2010) Detection of free living Amoebae, Acanthamoeba and Naegleria, in swimming pools, Malaysia. Trop Biomed 27(3):566–577
Johnston SP, Sriram R, Qvarnstrom Y, Roy S, Verani J, Yoder J, Lorick S, Roberts J, Beach MJ, Visvesvara G (2009) Resistance of Acanthamoeba cysts to disinfection in multiple contact lens solutions. J Clin Microbiol 47(7):2040–2045
Khan NA (2006) Acanthamoeba: biology and increasing importance in human health. FEMS Microbiol Rev 30:564–595
Laummaunwai P, Ruangjirachuporn W, Boonmars T (2012) A simple PCR condition for detection of a single cyst of Acanthamoeba species. Parasitol Res 110(4):1569–1572
Lek-Uthai U, Passara R, Roongruangchai K (2009) Morphological features of Acanthamoeba causing keratitis contaminated from contact lens cases. J Med Assoc Thai 92(7):156–163
Liesegang TJ (2002) Physiologic changes of the cornea with contact lens wear. CLAO J 28:12–27
Lohmann CP, Linde HJ, Reischl U (2000) Improved detection of microorganisms by polymerase chain reaction in delayed endophthalmitis after cataract surgery. J Ophthalmol 107(6):1047–1051
Marciano-Cabral F, Cabral G (2003) Acanthamoeba spp as agents of disease in humans. Clin Microbiol Rev 16(2):273–307
Mathers WD, Nelson SE, Lane JL, Wilson ME, Allen RC, Folberg R (2000) Confirmation of confocal microscopy diagnosis of Acanthamoeba keratitis using polymerase chain reaction analysis. Arch Ophthalmol 118(2):178–183
Myrowitz EH, Melia M, O’Brien TP (2002) The relationship between long term contact lens wear and corneal thickness. CLAO J 28:217–220
Niyyati M, Lorenzo-Morales J, Mohebali M, Rezaie S, Rahimi F, Babaei Z, Martín-Navarro CM, Farnia S, Valladares B, Rezaeian M (2009) Comparison of a PCR-based method with culture and direct examination for diagnosis of Acanthamoeba keratitis. Iranian J Parasitol 4(2):38–43
Pasricha G, Sharma S, Garg P, Aggarwal RK (2003) Use of 18S rRNA gene-based PCR assay for diagnosis of Acanthamoeba keratitis in non-contact lens wearers in India. J Clin Microbiol 41:3206–3211
Petry F, Torzewski M, Bohl J, Wilhelm-Schwenkmezger T, Scheid P, Walochnik J, Michel R, Zöller L, Werhahn KJ, Bhakdi S, Lackner KJ (2006) Early diagnosis of Acanthamoeba infection during routine cytological examination of cerebrospinal fluid. J Clin Microbiol 44(5):1903–1904
Qvarnstrom Y, Visvesvara GS, Sriram R, da Silva AJ (2006) Multiplex real-time PCR assay for simultaneous detection of Acanthamoeba spp., Balamuthia mandrillaris, and Naegleria fowleri. J Clin Microbiol 44:3589–3595
Riviére D, Szczebara F, Berjeaud J, H’echard Y (2006) Development of real-time PCR assay for quantification of Acanthamoeba trophozoites and cysts. J Microbiol Meth 64:78–83
Scheid P, Schwarzenberger R (2012) Acanthamoeba spp. as vehicle and reservoir of adenoviruses. Parasitol Res 111(1):479–485
Seitzman GD, Cevallos V, Margolis TP (2006) Rose bengal and lissamine green inhibit detection of herpes simplex virus by PCR. Am J Ophthalmol 141:756–758
Sharma S, Garg P, Rao GN (2000) Patient characteristics, diagnosis, and treatment of non-contact lens related Acanthamoeba keratitis. Br J Ophthalmol 84(10):1103–1108
Sharma S, Pasricha G, Das D, Aggarwal RK (2004) Acanthamoeba keratitis in non-contact lens wearers in India: DNA typing-based validation and a simple detection assay. Arch Ophthalmol 122(10):1430–1434
Thompson PP, Kowalski RP, Shanks RM, Gordon YJ (2008) Validation of real-time PCR for laboratory diagnosis of Acanthamoeba keratitis. J Clin Microbiol 46:3232–3236
Visvesvara GS, Moura H, Schuster FL (2007) Pathogenic and opportunistic free-living amoebae: Acanthamoeba spp., Balamuthia mandrillaris, Naegleria fowleri, and Sappinia diploidea. FEMS Immunol Med Microbiol 50:1–26
Wanachiwanawin D, Booranapong W, Kosrirukvongs P (2012) Clinical features of Acanthamoeba keratitis in contact lens wearers and non wearers. Southeast Asian J Trop Med Pub Health 43(3):549–456
Xuejun Z, Xiaoyan L, Xiaoji S, Guoxing X, Jianzhang H (2009) Application of 18S rDNA PCR technique in the laboratory diagnosis of Acanthamoeba keratitis in the clinical. Guoji Yanke Zazhi 9:715–718
Yera H, Zamfir O, Bourcier T, Ancelle T, Batellier L, Dupouy-Camet J, Chaumeil C (2007) Comparison of PCR, microscopic examination and culture for the early diagnosis and characterization of Acanthamoeba isolates from ocular infections. Eur J Clin Microbiol Infect Dis 26(3):221–224
Yera H, Zamfir O, Bourcier T, Viscogliosi E, Noel C, Jean D, Chaumeil C (2008) The genotypic characterization of Acanthamoeba isolates from human ocular samples. Br J Ophthalmol 92(8):1139–1141
Zhang Y, Sun X, Wang Z, Li R, Luo S, Jin X, Deng S, Chen W (2004) Identification of 18S ribosomal DNA genotype of Acanthamoeba from patients with keratitis in North China. Invest Ophthalmol Vis Sci 45(6):1904–1907
Zhao G, Zhai H, Yuan Q, Sun S, Liu T, Xie L (2014) Rapid and sensitive diagnosis of fungal keratitis using direct PCR without template DNA extraction. Clin Microbiol Infect. doi:10.1111/1469-0691.12571
Acknowledgments
The authors are grateful to Assistant Prof. Naglaa Fathy Alhusseini, Department of Biochemistry, Faculty of Medicine, Benha University, Egypt, for her technical advice.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Sayed, N.M., Younis, M.S., Elhamshary, A.M. et al. Acanthamoeba DNA can be directly amplified from corneal scrapings. Parasitol Res 113, 3267–3272 (2014). https://doi.org/10.1007/s00436-014-3989-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-014-3989-3