Abstract
Main conclusion
Auxin and abscisic acid regulate strawberry fruit ripening and senescence through cross-talk of their signal transduction pathways that further modulate the structural genes related to physico-chemical properties of fruit.
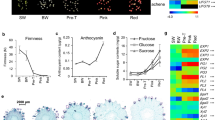
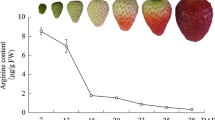
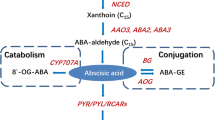
The physiological and transcriptomic changes in harvested strawberry fruits in responses to IAA, ABA and their combination were analyzed. Exogenous IAA delayed the ripening process of strawberries after harvest while ABA promoted the postharvest ripening. However, treatment with a combination of IAA and ABA did not slow down nor accelerate the postharvest ripening in the strawberry fruits. At the molecular level, exogenous IAA up regulated the expressions of genes related to IAA signaling, including AUX/IAA, ARF, TOPLESS and genes encoding E3 ubiquitin protein ligase and annexin, and down regulated genes related to pectin depolymerization, cell wall degradation, sucrose and anthocyanin biosyntheses. In contrast, exogenous ABA induced genes related to fruit softening, and genes involved in signaling pathways including SKP1, HSPs, CK2, and SRG1. Comparison of transcriptomes in responses to individual treatments with IAA or ABA or the combination revealed that there were cooperative and antagonistic actions between IAA and ABA in fruit. However, 17 % of the differentially expressed unigenes in response to the combination of IAA and ABA were unique and were not found in those unigenes responding to either IAA or ABA alone. The analyses also found that receptor-like kinases and ubiquitin ligases responded to both IAA and ABA, which seemed to play a pivotal role in both hormones’ signaling pathways and thus might be the cross-talk points of both hormones.




Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- ABI3:
-
Abscisic acid insensitive 3
- AIP2:
-
ABI-interacting protein 2
- ARF:
-
Auxin response factor
- ARP:
-
Auxin-repressed protein
- BAG:
-
Bcl-2 associated athanogene family
- CAD:
-
Cinnamyl alcohol dehydrogenase
- CK:
-
Casein kinase
- DGE:
-
Digital gene expression
- GH3:
-
Indole-3-acetic acid-amido synthetase
- HSPs:
-
Heat-shock proteins
- IAA:
-
Auxin
- KO:
-
KEGG Ortholog database
- KOG/COG:
-
Clusters of orthologous groups of proteins
- NCED:
-
9-cis-epoxycarotenoid dioxygenase
- RLKs:
-
Receptor-like kinases
- SKP1:
-
S-phase kinase-associated proteins 1
- VLCFA:
-
Very-long-chain fatty acid synthesis
- XT1:
-
Xylosyltransferase 1
- XTH:
-
Xyloglucan endotransglucosylase/hydrolase
References
Böttcher C, Keyzers RA, Boss PK, Davies C (2010) Sequestration of IAA by the indole-3-acetic acid-amido synthetase GH3-1 in grape berry (Vitis vinifera L.) and the proposed role of IAA conjugation during ripening. J Exp Bot 61:3615–3625
Callard D, Axelos M, Mazzolini L (1996) Novel molecular markers for late phases of the growth cycle of Arabidopsis thaliana cell-suspension cultures are expressed during organ senescence. Plant Physiol 112:705–715
Catalá C, Rose JKC, Bennett AB (2000) IAA-regulated genes encoding cell wall-modifying proteins are expressed during early tomato fruit growth. Plant Physiol 122:527–534
Chang S, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Rep 11:113–116
Chen JX, Mao LC, Mi HB, Zhao YY, Ying TJ, Luo ZS (2014) Detachment-accelerated ripening and senescence of strawberry (Fragaria × ananassa Duch. cv. Akihime) fruit and the regulation role of multiple phytohormones. Acta Physiol Plant 36:2441–2451
Doukhanina EV, Chen S, van der Zalm E, Godzik A, Reed J, Dickman MB (2006) Identification and functional characterization of the BAG protein family in Arabidopsis thaliana. J Biol Chem 281:18793–18801
Esaka M, Fujisawa K, Goto M, Kisu Y (1992) Regulation of ascorbate oxidase expression in pumpkin by auxin and copper. Plant Physiol 100:231–237
Fofana B, Duguid S, Cloutier S (2004) Cloning of fatty acid biosynthetic genes β-ketoacyl CoA synthase, fatty acid elongase, stearoyl-ACP desaturase, and fatty acid desaturase and analysis of expression in the early developmental stages of flax (Linum usitatissimum L.) seeds. Plant Sci 166:1487–1496
García-Hernández M, Murphy A, Taiz L (1998) Metallothioneins 1 and 2 have distinct but overlapping expression patterns in Arabidopsis. Plant Physiol 118:387–397
Given NK, Venis MA, Grierson D (1988a) Phenylalanine ammonia-lyase activity and anthocyanin synthesis in ripening strawberry fruit. J Plant Physiol 133:25–30
Given NK, Venis MA, Gierson D (1988b) Hormonal regulation of ripening in the strawberry, a non-climacteric fruit. Planta 174:402–406
Gou XP, He K, Yang H, Yuan T, Lin HH, Clouse SD, Li J (2010) Genome-wide cloning and sequence analysis of leucine-rich repeat receptor-like protein kinase genes in Arabidopsis thaliana. BMC Genom 11:19
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I, Adiconis X, Fan L, Raychowdhury R, Zeng QD (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652
Gray WM, Hellmann H, Dharmasiri S, Estelle M (2002) Role of the Arabidopsis RING-H2 protein RBX1 in RUB modification and SCF function. Plant Cell Online 14:2137–2144
Hu XL, Liu RX, Li YH, Wang W, Tai FJ, Xue RL, Li CH (2010) Heat shock protein 70 regulates the abscisic acid-induced antioxidant response of maize to combined drought and heat stress. Plant Growth Regul 60:225–235
Hubbard NL, Pharr DM, Huber SC (1991) Sucrose phosphate synthase and other sucrose metabolizing enzymes in fruits of various species. Physiol Plant 82:191–196
Jami SK, Clark GB, Ayele BT, Ashe P, Kirti PB (2012) Genome-wide comparative analysis of annexin superfamily in plants. Plos One 7:11
Ji K, Chen P, Sun L, Wang YP, Dai SJ, Li Q, Li P, Sun YF, Wu Y, Duan CR, Leng P (2012) Non-climacteric ripening in strawberry fruit is linked to ABA, FaNCED2 and FaCYP707A1. Funct Plant Biol 39:351–357
Jia HF, Chai YM, Li CL, Lu D, Luo JJ, Qin L, Shen YY (2011) Abscisic acid plays an important role in the regulation of strawberry fruit ripening. Plant Physiol 157:188–199
Jiang YM, Joyce DC (2003) ABA effects on ethylene production, PAL activity, anthocyanin and phenolic contents of strawberry fruit. Plant Growth Regul 39:171–174
Jones B, Frasse P, Olmos E, Zegzouti H, Li ZG, Latche A, Pech JC, Bouzayen M (2002) Down-regulation of DR12, an IAA-response-factor homolog, in the tomato results in a pleiotropic phenotype including dark green and blotchy ripening fruit. Plant J 32:603–613
Kabbage M, Dickman MB (2008) The BAG proteins: a ubiquitous family of chaperone regulators. Cell Mol Life Sci 65:1390–1402
Knee M, Sargent JA, Osborne DJ (1977) Cell-wall metabolism in metabolism in developing strawberry fruits. J Exp Bot 28:377–396
Lee S, Lee EJ, Yang EJ, Lee JE, Park AR, Song WH, Park OK (2004) Proteomic identification of annexins, calcium-dependent membrane binding proteins that mediate osmotic stress and abscisic acid signal transduction in Arabidopsis. Plant Cell 16:1378–1391
Lei GJ, Zhu XF, Wang ZW, Dong F, Dong NY, Zheng SJ (2014) Abscisic acid alleviates iron deficiency by promoting root iron reutilization and transport from root to shoot in Arabidopsis. Plant Cell Environ 37:852–863
Li B, Dewey C (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform 12:323
Li CJ, Liu ZJ, Zhang QR, Wang RZ, Xiao LT, Ma H, Chong K, Xu YY (2012) SKP1 is involved in abscisic acid signalling to regulate seed germination, stomatal opening and root growth in Arabidopsis thaliana. Plant Cell Environ 35:952–965
Li H, Mao WJ, Liu W, Dai HY, Liu YX, Ma Y, Zhang ZH (2013) Deep sequencing discovery of novel and conserved microRNAs in wild type and a white-flesh mutant strawberry. Planta 238:695–713
Liu D, Chen J, Lu W (2011) Expression and regulation of the early IAA-responsive Aux/IAA genes during strawberry fruit development. Mol Biol Rep 38:1187–1193
Lunkenbein S, Bellido M, Aharoni A, Salentijn EMJ, Kaldenhoff R, Coiner HA, Muñoz-Blanco J, Schwab W (2006) Cinnamate metabolism in ripening fruit. characterization of a UDP-glucose: cinnamate glucosyltransferase from strawberry. Plant Physiol 140:1047–1058
Mcatee P, Karim S, Schaffer R, David K (2013) A dynamic interplay between phytohormones is required for fruit development, maturation, and ripening. Front Plant Sci 4:79
Medina-Escobar N, Cardenas J, Munoz-Blanco J, Caballero JL (1998) Cloning and molecular characterization of a strawberry fruit ripening-related cDNA corresponding a mRNA for a low-molecular-weight heat-shock protein. Plant Mol Biol 36:33–42
Miszczak A, Forney CF, Prange RK (1995) Development of aroma volatiles and color during postharvest ripening of `Kent’ strawberries. J Am Soc Hortic Sci 120:650–655
Mulekar JJ, Bu Q, Chen F, Huq E (2012) Casein kinase II α subunits affect multiple developmental and stress-responsive pathways in Arabidopsis. Plant J 69:343–354
Munne-Bosch S, Muller M (2013) Hormonal cross-talk in plant development and stress responses. Front Plant Sci 4:529
OfosuAnim J, Kanayama Y, Yamaki S (1996) Sugar uptake into strawberry fruit is stimulated by abscisic acid and indoleacetic acid. Physiol Plant 97:169–174
Ohlrogge JB, Jaworski JG (1997) Regulation of fatty acid synthesis. Annu Rev Plant Physiol 48:109–136
Ornelas-Paz J, Yahia EM, Ramírez-Bustamante N, Pérez-Martínez JD, Escalante-Minakata MP, Ibarra-Junquera V, Acosta-Muñiz C, Guerrero-Prieto V, Ochoa-Reyes E (2013) Physical attributes and chemical composition of organic strawberry fruit (Fragaria x ananassa Duch, Cv. Albion) at six stages of ripening. Food Chem 138:372–381
Pierre-Jerome E, Moss BL, Nemhauser JL (2013) Tuning the IAA transcriptional response. J Exp Bot 64:2557–2563
Reddy ASN, Poovaiah BW (1990) Molecular cloning and sequencing of a cDNA for an IAA-repressed mRNA: correlation between fruit growth and repression of the IAA-regulated gene. Plant Mol Biol 14:127–136
Rodriguez MCS, Petersen M, Mundy J (2010) Mitogen-activated protein kinase signaling in plants. Annu Rev Plant Biol 61:621–649
Roudier F, Gissot L, Beaudoin F, Haslam R, Michaelson L, Marion J, Molino D, Lima A, Bach L, Morin H, Tellier F, Palauqui JC, Bellec Y, Renne C, Miquel M, DaCosta M, Vignard J, Rochat C, Markham JE, Moreau P, Napier J, Faure JD (2010) Very-long-chain fatty acids are involved in polar IAA transport and developmental patterning in Arabidopsis. Plant Cell 22:364–375
Shi HY, Li ZH, Zhang YX, Chen L, Xiang DY, Zhang YF (2014) Two pear glutathione S-transferases genes are regulated during fruit development and involved in response to salicylic acid, IAA, and glucose signaling. PloS One 9(2):e89926. doi:10.1371/journal.phone.0089926
Shin Y, Ryu JA, Liu RH, Nock JF, Watkins CB (2008) Harvest maturity, storage temperature and relative humidity affect fruit quality, antioxidant contents and activity, and inhibition of cell proliferation of strawberry fruit. Postharvest Biol Technol 49:201–209
Storey JD, Tibshirani R (2003) Statistical significance for genomewide studies. Proc Natl Acad Sci USA 100:9440–9445
Symons GM, Chua YJ, Ross JJ, Quittenden LJ, Davies NW, Reid JB (2012) Hormonal changes during non-climacteric ripening in strawberry. J Exp Bot 63:4741–4750
Szemenyei H, Hannon M, Long JA (2008) TOPLESS mediates IAA-dependent transcriptional repression during Arabidopsis embryogenesis. Science 319:1384–1386
ten Hove C, Bochdanovits Z, Jansweijer VA, Koning F, Berke L, Sanchez-Perez G, Scheres B, Heidstra R (2011) Probing the roles of LRR RLK genes in Arabidopsis thaliana roots using a custom T-DNA insertion set. Plant Mol Biol 76:69–83
Trainotti L, Tadiello A, Casadoro G (2007) The involvement of IAA in the ripening of climacteric fruits comes of age: the hormone plays a role of its own and has an intense interplay with ethylene in ripening peaches. J Exp Bot 58:3299–3308
Wang WX, Vinocur B, Shoseyov O, Altman A (2004) Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci 9:244–252
Wang LK, Feng ZX, Wang X, Wang XW, Zhang XG (2010) DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 26:136–138
Whitelaw CA, Le Huquet JA, Thurman DA, Tomsett AB (1997) The isolation and characterisation of type II metallothionein-like genes from tomato (Lycopersicon esculentum L.). Plant Mol Biol 33:503–511
Yamamoto KT, Mori H, Imaseki H (1992) Novel mRNA sequences induced by indole-3-acetic acid in sections of elongating hypocotyls of mung bean (Vigna radiata). Plant Cell Physiol 33:13–20
Zhang X, Garreton V, Chua NH (2005) The AIP2 E3 ligase acts as a novel negative regulator of ABA signaling by promoting ABI3 degradation. Genes Dev 19:1532–1543
Acknowledgments
This research is supported by the National Basic Research Program (973 program) of China (2013CB127101) and the National Natural Science Foundation of China (31372113).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1
Fig. S1 Length distribution of unigenes (TIFF 1055 kb)
Supplementary material 2
Fig. S2 KEGG classification of unigenes (TIFF 1772 kb)
Supplementary material 3
Fig. S3 KOG classification of unigenes (TIFF 1966 kb)
Supplementary material 4
Fig. S4 GO classification of unigenes (TIFF 3190 kb)
Supplementary material 5
Fig. S5 Correlation analysis of gene expression levels revealed by RT-qPCR and RNA-seq digital data (12 randomly selected unigenes are comp48373_c0, comp49239_c0, comp29094_c0, comp45854_c0, comp44817_c0, comp9725_c0, comp38169_c0, comp44449_c0, comp50216_c0, comp34077_c1, comp54510_c0, comp46511_c0) (TIFF 1078 kb)
Supplementary material 6
Fig. S6 Soluble solid (a), soluble sugar (b) and total anthocyanin (c) contents of strawberry fruits in response to exogenous IAA and ABA. 0, Control, IAA, ABA, IAA+ABA are noted as in the legend to Fig. 1. Lower-case letters (a, b, c, d) on the bar chart indicate the significant differences between different treatments (Duncan’s test, P < 0.05) (TIFF 1840 kb)
Rights and permissions
About this article
Cite this article
Chen, J., Mao, L., Lu, W. et al. Transcriptome profiling of postharvest strawberry fruit in response to exogenous auxin and abscisic acid. Planta 243, 183–197 (2016). https://doi.org/10.1007/s00425-015-2402-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-015-2402-5