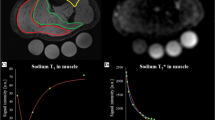
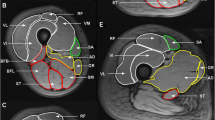
Abstract
Purpose
23Na MRI demonstrated increased tissue sodium concentrations in a number of pathologies. Acute atrophy results in muscle fibre volume shrinking that may result in a relative increase of extracellular volume and might affect sodium concentration. Thus, we hypothesized that local unloading of the calf muscles would lead to a decrease in muscle volume and an increase in muscle tissue sodium concentration.
Method
One lower leg of 12 healthy male subjects was submitted to a 60 day long period of unloading using the Hephaistos orthosis, while the other leg served as control. 23Na MRI and 2D PD-weighted Dixon turbo spin echo were obtained from the control and orthosis leg using a 3T scanner. For quantification, a sodium reference phantom was used with 10, 20, 30, and 40 mmol/L NaCl solution.
Result
Tissue sodium concentration (TSC) increased as an effect of unloading in the orthosis leg. Relative increases were 17.4 ± 16.8% (P = 0.005) in gastrocnemius medialis muscle, 11.1 ± 12.5 (P = 0.037) in gastrocnemius lateralis muscle, 16.2 ± 4.7% (P < 0.001) in soleus muscle, 10.0 ± 10.5% (P = 0.009) in the ventral muscle group, and 10.7 ± 10.0% (P = 0.003) in the central muscle group, respectively. TSC in the control leg did not significantly change. In the orthosis leg, muscle volume decreased as follows: medial gastrocnemius muscle: −5.4 ± 8.3% (P = 0.043) and soleus muscle: −7.8 ± 15.0% (P = 0.043).
Conclusion
Unloading atrophy is associated with an increase in muscle sodium concentration. 23Na MRI is capable of detecting these rather small changes.




Similar content being viewed by others
Abbreviations
- FoV:
-
Field of view (FoV)
- LME:
-
Linear mixed effects
- PD:
-
Proton density
- ROI:
-
Regions of interest
- SNR:
-
Signal-to-noise ratio
- TSC:
-
Tissue sodium concentration
- TSE:
-
Turbo spin echo
- TA:
-
Acquisition time
- TE:
-
Echo time
- TR:
-
Repetition time
- T2 :
-
Transversal relaxation time
References
Aagaard P, Suetta C, Caserotti P, Magnusson SP, Kjaer M (2010) Role of the nervous system in sarcopenia and muscle atrophy with aging: strength training as a countermeasure. Scand J Med Sci Sports 20:49–64. doi:10.1111/j.1600-0838.2009.01084.x
Adams GR, Caiozzo VJ, Baldwin KM, (2003) Skeletal muscle unweighting: spaceflight and ground-based models. J Appl Physiol 95(6):2185–2201
Allis JL, Seymour A-ML, Radda GK (1991) Absolute quantification of intracellular Na+ using triple-quantum-filtered sodium-23 NMR. J Magn Reson (1969) 93:71–76. doi:10.1016/0022-2364(91)90032-o
Bangerter NK, Tarbox GJ, Taylor MD, Kaggie JD (2016) Quantitative sodium magnetic resonance imaging of cartilage, muscle, and tendon. Quant Imaging Med Surg 6:699–714. doi:10.21037/qims.2016.12.10
Bansal N, Szczepaniak L, Ternullo D, Fleckenstein JL, Malloy CR (2000) Effect of exercise on 23Na MRI and relaxation characteristics of the human calf muscle. J Magn Reson Imaging 11:532–538. doi:10.1002/(sici)1522-2586(200005)11:5<532:aid-jmri9>3.0.co;2-#
Berendsen HJC, Edzes HT (1973) The observation and general interpretation of sodium magnetic resonance in biological material. Ann N Y Acad Sci 204:459–485. doi:10.1111/j.1749-6632.1973.tb30799.x
Boada FE, Christensen JD, Huang-Hellinger FR, Reese TG, Thulborn KR (1994) Quantitativein vivo tissue sodium concentration maps: the effects of biexponential relaxation. Magn Reson Med 32:219–223. doi:10.1002/mrm.1910320210
Borthakur A, Shapiro EM, Beers J, Kudchodkar S, Kneeland JB, Reddy R (2000) Sensitivity of MRI to proteoglycan depletion in cartilage: comparison of sodium and proton MRI osteoarthritis and cartilage/OARS. Osteoarthr Res Soc 8:288–293. doi:10.1053/joca.1999.0303
Borthakur A, Mellon E, Niyogi S, Witschey W, Kneeland JB, Reddy R (2006) Sodium and T1rho MRI for molecular and diagnostic imaging of articular cartilage. NMR Biomed 19:781–821. doi:10.1002/nbm.1102
Chang G, Wang L, Schweitzer ME, Regatte RR (2010) 3D 23Na MRI of human skeletal muscle at 7 Tesla: initial experience. Eur Radiol 20:2039–2046. doi:10.1007/s00330-010-1761-3
Constantinides CD, Gillen JS, Boada FE, Pomper MG, Bottomley PA (2000) Human skeletal muscle: sodium MR imaging and quantification-potential applications in exercise and disease. Radiology 216:559–568. doi:10.1148/radiology.216.2.r00jl46559
Dahlmann A et al (2015) Magnetic resonance-determined sodium removal from tissue stores in hemodialysis patients. Kidney Int 87:434–441. doi:10.1038/ki.2014.269
Desplanches D, Hoppeler H, Mayet MH, Denis C, Claassen H, Ferretti G (1998) Effects of bedrest on deltoideus muscle morphology and enzymes. Acta Physiol Scand 162:135–140. doi:10.1046/j.1365-201X.1998.0288f.x
Drummer C, Hesse C, Baisch F, Norsk P, Elmann-Larsen B, Gerzer R, Heer M (2000) Water and sodium balances and their relation to body mass changes in microgravity. Eur J Clin Investig 30:1066–1075. doi:10.1046/j.1365-2362.2000.00766.x
Fischer MA, Pfirrmann CW, Espinosa N, Raptis DA, Buck FM (2014) Dixon-based MRI for assessment of muscle-fat content in phantoms, healthy volunteers and patients with achillodynia: comparison to visual assessment of calf muscle quality. Eur Radiol 24:1366–1375. doi:10.1007/s00330-014-3121-1
Goodpaster BH, Kelley DE, Thaete FL, He J, Ross R (2000) Skeletal muscle attenuation determined by computed tomography is associated with skeletal muscle lipid content. J Appl Physiol 89:104–110
Hudlicka O (1985) Development and adaptability of microvasculature in skeletal muscle. J Exp Biol 115:215–228
Inglese M et al (2010) Brain tissue sodium concentration in multiple sclerosis: a sodium imaging study at 3 tesla. Brain: J Neurol 133:847–857. doi:10.1093/brain/awp334
Konstandin S, Nagel AM (2014) Measurement techniques for magnetic resonance imaging of fast relaxing nuclei. Magma 27:5–19. doi:10.1007/s10334-013-0394-3
Kopp C et al (2012) (23)Na magnetic resonance imaging of tissue sodium. Hypertension 59:167–172. doi:10.1161/HYPERTENSIONAHA.111.183517
Kopp C et al (2013) 23Na magnetic resonance imaging-determined tissue sodium in healthy subjects and hypertensive patients. Hypertension 61:635–640. doi:10.1161/HYPERTENSIONAHA.111.00566
LeBlanc A et al (2000) Muscle volume, MRI relaxation times (T2), and body composition after spaceflight. J Appl Physiol 89:2158–2164
Lu A, Atkinson IC, Claiborne TC, Damen FC, Thulborn KR (2010) Quantitative sodium imaging with a flexible twisted projection pulse sequence. Magn Reson Med 63:1583–1593. doi:10.1002/mrm.22381
Maarouf A et al (2014) Topography of brain sodium accumulation in progressive multiple sclerosis. Magma 27:53–62. doi:10.1007/s10334-013-0396-1
Madelin G, Regatte RR (2013) Biomedical applications of sodium MRI in vivo. J Magn Reson Imaging: JMRI 38:511–529. doi:10.1002/jmri.24168
Nagel AM, Laun FB, Weber MA, Matthies C, Semmler W, Schad LR (2009) Sodium MRI using a density-adapted 3D radial acquisition technique. Magn Reson Med 62:1565–1573. doi:10.1002/mrm.22157
Nagel AM, Amarteifio E, Lehmann-Horn F, Jurkat-Rott K, Semmler W, Schad LR, Weber MA (2011) 3 Tesla sodium inversion recovery magnetic resonance imaging allows for improved visualization of intracellular sodium content changes in muscular channelopathies. Investig Radiol 46:759–766. doi:10.1097/RLI.0b013e31822836f6
Narici MV, de Boer MD (2011) Disuse of the musculo-skeletal system in space and on earth. Eur J Appl Physiol 111:403–420. doi:10.1007/s00421-010-1556-x
Ooms KJ, Cannella M, Vega AJ, Marcolongo M, Polenova T (2008) 23Na TQF NMR imaging for the study of spinal disc tissue. J Magn Reson 195:112–115. doi:10.1016/j.jmr.2008.07.024
Ouwerkerk R, Bleich KB, Gillen JS, Pomper MG, Bottomley PA (2003) Tissue sodium concentration in human brain tumors as measured with 23Na MR imaging. Radiology 227:529–537. doi:10.1148/radiol.2272020483
Paling D et al (2013) Sodium accumulation is associated with disability and a progressive course in multiple sclerosis. Brain: J Neurol 136:2305–2317. doi:10.1093/brain/awt149
Ra JB, Hilal SK, Cho ZH (1986) A method forin vivo MR imaging of the shortT2 component of sodium-23. Magn Reson Med 3:296–302. doi:10.1002/mrm.1910030213
Rittweger J, Frost HM, Schiessl H, Ohshima H, Alkner B, Tesch P, Felsenberg D (2005) Muscle atrophy and bone loss after 90 days’ bed rest and the effects of flywheel resistive exercise and pamidronate: results from the LTBR study. Bone 36:1019–1029. doi:10.1016/j.bone.2004.11.014
Rittweger J, Moller K, Bareille MP, Felsenberg D, Zange J (2013) Muscle X-ray attenuation is not decreased during experimental bed rest. Muscle Nerve 47:722–730. doi:10.1002/mus.23644
Rooney WD, Springer CS (1991) A comprehensive approach to the analysis and interpretation of the resonances of spins 3/2 from living systems. NMR Biomed 4:209–226. doi:10.1002/nbm.1940040502
Roubenoff R (2000) Sarcopenia and its implications for the elderly. Eur J Clin Nutr 54:S40–S47. doi:10.1038/sj.ejcn.1601024
Sangwan S, Green RA, Taylor NF (2014) Characteristics of Stabilizer Muscles: a Systematic Review. Physiother Can 66(4):348–358
Shinar H, Navon G (1986) Sodium-23 NMR relaxation times in body fluids. Magn Reson Med 3:927–934. doi:10.1002/mrm.1910030613
Titze J et al (2003) Osmotically inactive skin Na+ storage in rats. Am J Physiol Renal Physiol 285:F1108–F1117. doi:10.1152/ajprenal.00200.2003
Weber MA, Nagel AM, Wolf MB, Jurkat-Rott K, Kauczor HU, Semmler W, Lehmann-Horn F (2012) Permanent muscular sodium overload and persistent muscle edema in Duchenne muscular dystrophy: a possible contributor of progressive muscle degeneration. J Neurol 259:2385–2392. doi:10.1007/s00415-012-6512-8
Weber T, Ducos M, Mulder E, Herrera F, Bruggemann GP, Bloch W, J W (2013) The specific role of gravitational accelerations for arterial adaptations. J Appl Physiol (1985) 114:387–393. doi:10.1152/japplphysiol.01117.2012
Zange J et al (2017) Using the Hephaistos orthotic device to study countermeasure effectiveness of neuromuscular electrical stimulation and dietary lupin protein supplementation, a randomised controlled trial. PLoS One 12:e0171562. doi:10.1371/journal.pone.0171562
Acknowledgements
K. Schopen received a Helmholtz Space Life Sciences Research School (SpaceLife) Ph.D. scholarship. SpaceLife was funded in equal parts by the Helmholtz Association (Grant No.: VH-KO-300) and the German Aerospace Center (DLR). The NutriHEP study was funded by the DLR internal cost object 2475 101.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Guido Ferretti.
Rights and permissions
About this article
Cite this article
Gerlach, D.A., Schopen, K., Linz, P. et al. Atrophy of calf muscles by unloading results in an increase of tissue sodium concentration and fat fraction decrease: a 23Na MRI physiology study. Eur J Appl Physiol 117, 1585–1595 (2017). https://doi.org/10.1007/s00421-017-3647-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-017-3647-4