Abstract
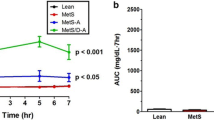
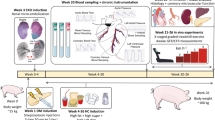
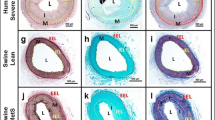
Ossabaw miniswine have been naturally selected to efficiently store large amounts of lipids offering them a survival advantage. Our goal was to evaluate the myocardial response to chronic ischemia of the Ossabaw consuming a hypercaloric, high-fat/cholesterol diet with and without metformin supplementation. At 6 weeks of age animals were fed either a regular diet (OC, n = 9), a hypercaloric high-fat/cholesterol diet (OHC, n = 9), or a hypercaloric high-fat/cholesterol diet supplemented with metformin (OHCM, n = 8). At 9 weeks, all animals underwent ameroid constrictor placement to the left circumflex coronary artery to simulate chronic ischemia. Seven weeks after ameroid placement, all animals underwent hemodynamic and functional measurements followed by cardiac harvest. Both OHC and OHCM animals developed significantly greater weight gain, total cholesterol, and LDL:HDL ratio compared to OC controls. Metformin administration reversed diet-induced hypertension and glucose intolerance. There were no differences in global and regional contractility, myocardial perfusion, capillary and arteriolar density, or total protein oxidation between groups. Myocardial protein expression of VEGF, PPAR-α, γ, and δ was significantly increased in the OHC and OHCM groups. Microvessel reactivity was improved in the OHC and OHCM groups compared to controls, and correlated with increased p-eNOS expression. Overfed Ossabaw miniswine develop several components of metabolic syndrome. However, impairments of myocardial function, neovascularization and perfusion were not present, and microvessel reactivity was paradoxically improved in hypercholesterolemic animals. The observed cardioprotection despite metabolic derangements may be due to lipid-dependant upregulation of the PPAR pathway which is anti-inflammatory and governs myocardial fatty acid metabolism.







Similar content being viewed by others
References
Belin de Chantemele EJ, Irfan Ali M, Mintz J, Stepp DW (2009) Obesity-induced insulin resistance causes endothelial dysfunction without reducing the vascular response to hindlimb ischemia. Basic Res Cardiol 104:707–717. doi:10.1007/s00395-009-0062-y
Bender SB, Tune JD, Borbouse L, Long X, Sturek M, Laughlin MH (2009) Altered mechanism of adenosine-induced coronary arteriolar dilation in early-stage metabolic syndrome. Exp Biol Med (Maywood) 234:683–692. doi:10.3181/0812-RM-350
Bhamra GS, Hausenloy DJ, Davidson SM, Carr RD, Paiva M, Wynne AM, Mocanu MM, Yellon DM (2008) Metformin protects the ischemic heart by the Akt-mediated inhibition of mitochondrial permeability transition pore opening. Basic Res Cardiol 103:274–284. doi:10.1007/s00395-007-0691-y
Boodhwani M, Nakai Y, Mieno S, Voisine P, Bianchi C, Araujo EG, Feng J, Michael K, Li J, Sellke FW (2006) Hypercholesterolemia impairs the myocardial angiogenic response in a swine model of chronic ischemia: role of endostatin and oxidative stress. Ann Thorac Surg 81:634–641. doi:10.1016/j.athoracsur.2005.07.090
Boodhwani M, Sodha NR, Mieno S, Ramlawi B, Xu SH, Feng J, Clements RT, Ruel M, Sellke FW (2007) Insulin treatment enhances the myocardial angiogenic response in diabetes. J Thorac Cardiovasc Surg 134:1453–1460. doi:10.1016/j.jtcvs.2007.08.025 (discussion 1460)
Boodhwani M, Sodha NR, Mieno S, Xu SH, Feng J, Ramlawi B, Clements RT, Sellke FW (2007) Functional, cellular, and molecular characterization of the angiogenic response to chronic myocardial ischemia in diabetes. Circulation 116:I31–I37. doi:10.1161/CIRCULATIONAHA.106.680157
Borbouse L, Dick GM, Payne GA, Payne BD, Svendsen MC, Neeb ZP, Alloosh M, Bratz IN, Sturek M, Tune JD (2010) Contribution of BK(Ca) channels to local metabolic coronary vasodilation: effects of metabolic syndrome. Am J Physiol Heart Circ Physiol 298:H966–H973. doi:10.1152/ajpheart.00876.2009
Chu LM, Robich MP, Lassaletta AD, Feng J, Laham RJ, Burgess T, Clements RT, Sellke FW (2011) Resveratrol supplementation abrogates pro-arteriogenic effects of intramyocardial vascular endothelial growth factor in a hypercholesterolemic swine model of chronic ischemia. Surgery 150:390–399. doi:10.1016/j.surg.2011.06.009
Clements RT, Sodha NR, Feng J, Boodhwani M, Liu Y, Mieno S, Khabbaz KR, Bianchi C, Sellke FW (2009) Impaired coronary microvascular dilation correlates with enhanced vascular smooth muscle MLC phosphorylation in diabetes. Microcirculation 16:193–206. doi:10.1080/10739680802461950
de Groot D, Pasterkamp G, Hoefer IE (2009) Cardiovascular risk factors and collateral artery formation. Eur J Clin Invest 39:1036–1047. doi:10.1111/j.1365-2362.2009.02205.x
Dohner JW (2001) The encyclopedia of historic and endangered livestock and poultry breeds. Yale University Press
Hattan N, Chilian WM, Park F, Rocic P (2007) Restoration of coronary collateral growth in the Zucker obese rat: impact of VEGF and ecSOD. Basic Res Cardiol 102:217–223. doi:10.1007/s00395-007-0646-3
Iliadis F, Kadoglou N, Didangelos T (2011) Insulin and the heart. Diabetes Res Clin Pract 93(Suppl 1):S86–S91. doi:10.1016/S0168-8227(11)70019-5
Katz PS, Trask AJ, Souza-Smith FM, Hutchinson KR, Galantowicz ML, Lord KC, Stewart JA Jr, Cismowski MJ, Varner KJ, Lucchesi PA (2011) Coronary arterioles in type 2 diabetic (db/db) mice undergo a distinct pattern of remodeling associated with decreased vessel stiffness. Basic Res Cardiol 106:1123–1134. doi:10.1007/s00395-011-0201-0
Lassaletta AD, Chu LM, Sellke FW (2011) Therapeutic neovascularization for coronary disease: current state and future prospects. Basic Res Cardiol 106:897–909. doi:10.1007/s00395-011-0200-1
Li Z, Woollard JR, Wang S, Korsmo MJ, Ebrahimi B, Grande JP, Textor SC, Lerman A, Lerman LO (2011) Increased glomerular filtration rate in early metabolic syndrome is associated with renal adiposity and microvascular proliferation. Am J Physiol Renal Physiol 301:F1078–F1087. doi:10.1152/ajprenal.00333.2011
Liepinsh E, Skapare E, Svalbe B, Makrecka M, Cirule H, Dambrova M (2011) Anti-diabetic effects of mildronate alone or in combination with metformin in obese Zucker rats. Eur J Pharmacol 658:277–283. doi:10.1016/j.ejphar.2011.02.019
Liu Y, Gutterman DD (2009) Vascular control in humans: focus on the coronary microcirculation. Basic Res Cardiol 104:211–227. doi:10.1007/s00395-009-0775-y
Lloyd PG, Fang M, Brisbin ILJ, Anderson L, Sturek M (2006) AMP kinase gene mutation is consistent with a thrifty phenotype (metabolic syndrome) in a population of feral swine. FASEB J 20:A299
Maejima Y, Okada H, Haraguchi G, Onai Y, Kosuge H, Suzuki J, Isobe M (2011) Telmisartan, a unique ARB, improves left ventricular remodeling of infarcted heart by activating PPAR gamma. Lab Invest 91:932–944. doi:10.1038/labinvest.2011.45
Moreno-Navarrete JM, Ortega FJ, Rodriguez-Hermosa JI, Sabater M, Pardo G, Ricart W, Fernandez-Real JM (2011) OCT1 expression in adipocytes could contribute to increased metformin action in obese subjects. Diabetes 60:168–176. doi:10.2337/db10-0805
Neeb ZP, Edwards JM, Alloosh M, Long X, Mokelke EA, Sturek M (2010) Metabolic syndrome and coronary artery disease in Ossabaw compared with Yucatan swine. Comp Med 60:300–315
Oltman CL, Richou LL, Davidson EP, Coppey LJ, Lund DD, Yorek MA (2006) Progression of coronary and mesenteric vascular dysfunction in Zucker obese and Zucker diabetic fatty rats. Am J Physiol Heart Circ Physiol 291:H1780–H1787. doi:10.1152/ajpheart.01297.2005
Orlandi A, Chavakis E, Seeger F, Tjwa M, Zeiher AM, Dimmeler S (2010) Long-term diabetes impairs repopulation of hematopoietic progenitor cells and dysregulates the cytokine expression in the bone marrow microenvironment in mice. Basic Res Cardiol 105:703–712. doi:10.1007/s00395-010-0109-0
Park Y, Yang J, Zhang H, Chen X, Zhang C (2011) Effect of PAR2 in regulating TNF-alpha and NAD(P)H oxidase in coronary arterioles in type 2 diabetic mice. Basic Res Cardiol 106:111–123. doi:10.1007/s00395-010-0129-9
Petznick A (2011) Insulin management of type 2 diabetes mellitus. Am Fam Physician 84:183–190
Robich MP, Osipov RM, Nezafat R, Feng J, Clements RT, Bianchi C, Boodhwani M, Coady MA, Laham RJ, Sellke FW (2010) Resveratrol improves myocardial perfusion in a swine model of hypercholesterolemia and chronic myocardial ischemia. Circulation 122:S142–S149. doi:10.1161/CIRCULATIONAHA.109.920132
Schaper W (2009) Collateral circulation: past and present. Basic Res Cardiol 104:5–21. doi:10.1007/s00395-008-0760-x
Setty S, Sun W, Tune JD (2003) Coronary blood flow regulation in the prediabetic metabolic syndrome. Basic Res Cardiol 98:416–423. doi:10.1007/s00395-003-0418-7
Sodha NR, Boodhwani M, Clements RT, Xu SH, Khabbaz KR, Sellke FW (2008) Increased antiangiogenic protein expression in the skeletal muscle of diabetic swine and patients. Arch Surg 143:463–470. doi:10.1001/archsurg.143.5.463
Sodha NR, Clements RT, Boodhwani M, Xu SH, Laham RJ, Bianchi C, Sellke FW (2009) Endostatin and angiostatin are increased in diabetic patients with coronary artery disease and associated with impaired coronary collateral formation. Am J Physiol Heart Circ Physiol 296:H428–H434. doi:10.1152/ajpheart.00283.2008
Spurlock ME, Gabler NK (2008) The development of porcine models of obesity and the metabolic syndrome. J Nutr 138:397–402
Varga O, Harangi M, Olsson IA, Hansen AK (2010) Contribution of animal models to the understanding of the metabolic syndrome: a systematic overview. Obes Rev 11:792–807. doi:10.1111/j.1467-789X.2009.00667.x
Volzke H, Henzler J, Menzel D, Robinson DM, Hoffmann W, Vogelgesang D, John U, Motz W, Rettig R (2007) Outcome after coronary artery bypass graft surgery, coronary angioplasty and stenting. Int J Cardiol 116:46–52. doi:10.1016/j.ijcard.2006.02.008
Wolkart G, Schrammel A, Dorffel K, Haemmerle G, Zechner R, Mayer B (2011) Cardiac dysfunction in adipose triglyceride lipase deficiency: treatment with a PPARalpha agonist. Br J Pharmacol. doi:10.1111/j.1476-5381.2011.01490.x (Epub ahead of print)
Ye Y, Perez-Polo JR, Aguilar D, Birnbaum Y (2011) The potential effects of anti-diabetic medications on myocardial ischemia-reperfusion injury. Basic Res Cardiol 106:925–952. doi:10.1007/s00395-011-0216-6
Zhang H, Potter BJ, Cao JM, Zhang C (2011) Interferon-gamma induced adipose tissue inflammation is linked to endothelial dysfunction in type 2 diabetic mice. Basic Res Cardiol 106:1135–1145. doi:10.1007/s00395-011-0212-x
Acknowledgments
Funding was provided by grants from the National Heart, Lung, and Blood Institute (R01HL46716, R01HL69024, and R01HL85647, Dr. Sellke), NIH Training grant 5T32-HL076134 (Dr. Lassaletta), NIH Training grant 5T32-HL094300 (Dr. Chu, Dr. Elmadhun), NIH Training grant T32HL007734 (Dr. Robich), and through the Thoracic Surgery Foundation for Research and Education Fellowship (Dr. Lassaletta). We would like to thank the animal facility staff at the Rhode Island Hospital.
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lassaletta, A.D., Chu, L.M., Robich, M.P. et al. Overfed Ossabaw swine with early stage metabolic syndrome have normal coronary collateral development in response to chronic ischemia. Basic Res Cardiol 107, 243 (2012). https://doi.org/10.1007/s00395-012-0243-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-012-0243-y