Abstract
Controversy exists on the optimal age for elective resection of asymptomatic congenital pulmonary airway malformation. Current recommendations vary widely, highlighting the overall lack of consensus. A systematic search of Embase, MEDLINE, CINAL, and CENTRAL was conducted in January 2016. Identified citations were screening independently in duplicate and consensus was required for inclusion. Results were pooled using inverse variance fixed effects meta-analysis. Meta-analysis results indicate no statistically significant differences for complications within the 3-month and 6-month age comparison groups [odds ratio (OR) 4.20, 95% confidence interval (CI) 0.78–22.77, I 2 = 0%; OR 2.39, 95% CI 0.63–9.11, I 2 = 0%, respectively]. Older patients were significantly favoured for 3-month and 6-month age comparison groups for length of hospital stay [mean difference (MD) 4.13, 95% CI 2.31–5.96, I 2 = 0%; MD 3.38, 95% CI 0.44–6.31, I 2 = 0%, respectively]. Borderline statistical significance was observed for chest tube duration in patients ≥6 months of age (MD 1.06, 95% CI 0.02–2.09, I 2 = 0%). No mortalities were recorded. Surgical treatment appears to be safe at all ages, with no mortalities and similar rates of complications between age groups. The included evidence was not sufficient to make a conclusive recommendation on optimal age for elective resection.
Similar content being viewed by others
Introduction
Congenital pulmonary airway malformation (CPAM) is a relatively rare congenital abnormality that is represented by a hamartomatous lesion of the bronchial tree [1]. Suppressed growth of alveolar tissue within these lesions causes normal lung structure to be replaced with a multicystic mass [2], potentially leading to the development of various respiratory complications including pneumonia, pneumothorax, hemoptysis, hemothorax, and the development of malignancies (carcinomas and pleuropulmonary blastomas) [3]. Presentation of these symptoms indicates the need for immediate surgical resection, however, the treatment plan for patients who are asymptomatic at diagnosis is less clear. While some paediatric surgeons choose to conservatively manage their patients through close observation, others recommend prophylactic resection of asymptomatic lesions [1, 4–10]. When families and surgeons agree to elective resection, questions still remain concerning the optimal age for surgery, with current recommendations varying from 4 weeks [11] to 3 years [12].
Evidence exists supporting both early and delayed surgical resections. Early surgery is often conducted to minimize the risk of the lesion becoming infected or malignant, as surgery is commonly believed to be easier in patients without previous sepsis or adhesions [13]. Additionally, patients who undergo early resection are believed to experience compensatory lung growth [14–17], an ability that is thought to decrease with age [18, 19]. Early resection also averts prolonged periods of observation with repeated imaging studies, decreasing overall radiation exposure and related risks [20]. Alternatively, delaying surgery to later in infancy is beneficial as older patients have a decreased anaesthetic and surgical risk [21].
We conducted a systematic review to evaluate if age at elective resection of asymptomatic paediatric CPAM patients is related to clinical outcomes in order to determine the optimal age for surgical intervention.
Methods
We registered the protocol for this systematic review in PROSPERO [22] on 25 November 2015 (CRD42015029679). Our methods are in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [23], and are briefly described here.
Generation of research question
A total of 17 paediatric surgeons were surveyed using the Delphi method to determine a topic in the management of CPAM that required further research and/or consensus [24]. Through two rounds of questionnaires, the optimal age for elective resection of asymptomatic CPAM lesions was ranked as the second most controversial question. The top ranked question has been addressed in a separate systematic review [10]. The survey was conducted on behalf of the Canadian Association of Paediatric Surgeons Evidence-Based Resource (CAPS EBR) [25]; an online resource that facilitates the rapid uptake of good evidence into practice by providing paediatric surgeons with easy-to-access up-to-date research evidence on key topics of concern.
Inclusion/exclusion criteria
We included comparative and non-comparative studies that evaluated the association between patient age at the time of elective resection and clinical outcomes. Patients must have been ≤18 years of age and received surgery for CPAM/hybrid lesions with features of CPAM while asymptomatic. Non-English language studies were excluded, as were literature reviews, case studies, editorials, letters to the editor, commentaries, and conference proceedings.
Literature search
We searched CINAHL (1982 onwards), CENTRAL, EMBASE (1980 onwards), and MEDLINE (1996 onwards) on 4 January 2016 (Please see Supplementary File 1 for sample search strategy). We identified additional publications by hand-searching the reference sections of included studies.
Screening
Two reviewers independently assessed all citations identified by the literature search for relevance. At a title and abstract level, this was done using the liberal accelerated method [26]; at the full-text level reviewers reached consensus on articles to be included in the final analysis. Disagreements were resolved by discussion.
Data extraction
One reviewer extracted data on characteristics pertaining to the study in general (e.g. study design, country/region), patients (e.g. gender, prenatal diagnosis), and the intervention (e.g. age and weight at surgery, type of resection). The primary clinical outcome extracted was complications, and secondary outcomes were mortality, length of hospital stay (LOS), rate and incidence of infection, need for and length of ventilation, length of pleural drainage, pulmonary function, blood loss, and histology. All data were verified by a second reviewer.
Quality assessment
Two researchers independently assessed the methodological quality of non-randomized studies using MINORS [27], followed by a consensus process.
Statistical analysis
We conducted meta-analyses whenever possible using inverse variance fixed effects models in Review Manager Version 5.3 [28] to compare outcomes in patients who underwent surgery before and after commonly recommended ages for elective CPAM resection: ≤1 month versus >1 month, <3 months versus ≥3 months, <6 months versus ≥6 months, <1 year versus ≥1 year, <2 years versus ≥2 years. Count and dichotomous data were expressed as odds ratios (OR) and 95% confidence intervals (95% CI), while continuous data were expressed as mean difference (MD) and 95% CI. Forest plots were used to visualize the data, and statistical heterogeneity was assessed using the I 2 test with 95% CI. When the number of included studies was insufficient to allow for meta-analysis, we described results narratively. Publication bias assessment was not possible due to an insufficient number of included studies.
Results
Study characteristics
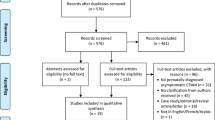
A total of 1458 citations were identified in our literature search (Fig. 1). Following de-duplication and title and abstract screening, 275 full-text studies were assessed for eligibility. While 20 studies met our a priori inclusion criteria, the authors of 17 of these studies had to be contacted to obtain data specific to only asymptomatic CPAM patients who received elective surgery. Six studies had to be excluded as the author did not respond to emails [6, 29], no longer had access to the data [30, 31], or did not have time to extract requested data [32, 33]. The 14 included studies originated in North America, Europe, or Asia, with the majority utilizing a retrospective (n = 13), single-centre (n = 9), comparative (n = 10) study design (Table 1). A total of 337 asymptomatic CPAM patients who underwent elective resection at various ages were evaluated within these studies. In studies that reported on additional patient characteristics, almost all patients were diagnosed prenatally and there were more males than females (137:95). Stocker classification of lesions was essentially equal between type I (n = 45) and type II (n = 46), with the slight majority occurring in the right lower lobe (Table 1).
Methodological quality
The methodological quality of included studies was poor to moderate, with median scores of 9.5/16 (range 6–11) and 15/24 (range 10–17) for non-comparative and comparative studies, respectfully (Table 1). Six items received perfect or near-perfect scores across all 14 included studies (clearly stated aim, consecutive patients, appropriate endpoints, minimal loss to follow-up, adequate control groups, contemporary groups), while the remaining six items were either not reported or were inadequately reported in ≥50% of included studies.
Primary outcome: complications
Seven comparative studies examined the effect of operative age on patient complications [7, 34–39]. Results generally indicated that although older patients were favoured, the odds of experiencing complications were not significantly different between younger and older patients undergoing elective resection of CPAM lesions. This was observed in meta-analysis of patients <3 months versus ≥3 months (OR 4.20, 95% CI 0.78–22.77, I 2 = 0%, P = 0.10; Fig. 2a) and <6 months versus ≥6 months (OR 2.39, 95% CI 0.63–9.11, I 2 = 0%, P = 0.20; Fig. 2b). Complications were also shown to not be significantly different between patients <6 months versus ≥6 months whether the surgery was open [37] or thoracoscopic [36].
While this outcome was examined in two comparative studies for patients ≤1 month versus >1 months (with complication rates of 0/1 (0%) versus 13/34 (38%) [7] and 1/6 (17%) versus 0/4 (0%) [38]) and one comparative study for patients <24 months versus ≥24 months (with complication rates of 0/8 (0%) vs. 1/1 (100%) [39]), meta-analysis was not deemed to be appropriate due to low patient numbers.
Data from an additional three studies allowed for non-comparative assessment of complications following elective surgery [1, 40, 41]. Within the neonatal period (<1 month), 31% (4/13) of patients reviewed by Waszak et al. [1] experienced complications including pleural effusion, repeated pneumothorax, and repeated bronchiolitis, with the majority occurring shortly after surgery [3/31 (10%)]. For patients younger than 4 months of age, Laje et al. [41] showed that complications only occurred in 7% (10/147), primarily consisting of persistent air leaks, need for reintubation, and chest tube reinsertion. Kongstad et al. [40] also showed that complications were relatively rare for patients undergoing surgery at approximately 12 months of age [1/13 (8%)].
Secondary outcome: length of hospital stay (LOS)
A meta-analysis of results from three comparative studies evaluating LOS for two different age groups indicated that older age groups are consistently favoured over the comparative younger group [34, 36, 37]. More specifically, total LOS was significantly reduced in patients who were ≥3 months of age at the time of their operation compared to patients <3 months of age (MD 4.13, 95% CI 2.31–5.96, I 2 = 0%, P < 0.001; Fig. 3a) and in patients ≥6 months of age compared to <6 months of age (MD 3.38, 95% CI 0.44–6.31, I 2 = 0%, P = 0.02; Fig. 3b). LOS in the intensive care unit was evaluated in one study, indicating older patients were again favoured (<6 months: 2.0 ± 2.2 days vs. ≥6 months: 0.4 ± 0.7 days) [36]. When type of surgery (open or thoracoscopic) was examined in the 6-month age group, LOS was significantly reduced in patients ≥6 months following open surgery (P = 0.03) [37], but no difference was observed in patients undergoing thoracoscopic surgery (P = 0.23) [36].
In the one comparative study that evaluated LOS for the 1-month age group, only one patient ≤1 month of age was included (≤1 month: 7 days vs. >1 month: 11.5 ± 5.8 days) [7], making analysis inappropriate.
Data from two additional studies allowed for non-comparative assessment of LOS following elective surgery, one within the neonatal period (<1 month of age) [42] and one before 4 months of age [41]. Only two patients <1 month old were evaluated for LOS by Chen et al., showing an average stay of 5.5 ± 0.7 days [42]. Patients <4 months of age had an overall average LOS of 3.3 ± 2.0 days, which was similar whether surgery was open or thoracoscopic (3.4 ± 2.0 days and 3.1 ± 2.0 days, respectively) [41].
Secondary outcome: pleural drainage/chest tube duration
Meta-analysis of the two studies that evaluated pleural drainage in the 6 months of age comparison group showed borderline statistical significance favouring patients undergoing elective resection at ≥6 month of age compared to patients <6 months of age (MD 1.06, 95% CI 0.02–2.09, I 2 = 0%, P = 0.05; Fig. 4) [36, 37]. When these two studies were further examined based on type of surgery, open surgery showed borderline statistical significance favouring ≥6 month of age (P = 0.05) [37], while thoracoscopic surgery showed no difference (P = 0.58) [36].
Length of pleural drainage was similar for patients undergoing surgery before and after 3 months of age (<3 months: 5.7 ± 2.2 days vs. ≥3 months: 5.1 ± 1.5 days) [37]. In the single comparative study that evaluated pleural drainage for the 1-month age group only one patient was ≤1 month of age, compared to 34 patients >1 month of age (5 days vs. 8.3 ± 6.2 days, respectively) [7]. As such, analysis of the data was thought to be inappropriate.
A non-comparative assessment of patients less than 4 months of age indicated that the average chest tube duration was 1.5 ± 1.5 days, a number that was similar whether patients received open (1.6 ± 1.3 days) or thoracoscopic (1.6 ± 2.0 days) surgery [41].
Secondary outcome: ventilation outcomes
Conforti et al. showed that length of ventilation was similar between patients <3 months and ≥3 months of age at surgery (0.6 ± 0.8 days vs. 0.7 ± 0.7 days, respectively), and patients < 6 months and ≥6 months of age at surgery (0.6 ± 0.7 days vs. 0.8 ± 0.9 days, respectively) [37]. While comparative data were not available for the 1-month age group, two non-comparative studies presented data for this outcome within the neonatal period (<1 month). With only two patients, Chen et al. found an average length of ventilation of 4.0 ± 1.4 days [42], while Waszak et al. reported an average of 1.6 ± 0.5 [1] days for 13 patients <1 month of age.
The number of patients who required ventilation following elective resection was also similar between age groups. Conforti et al. observed a comparable number of patients with post-operative ventilation within the 3 months and 6 months of age comparison groups (<3 months: 9/19 (47%) vs. ≥3 months: 9/16 (56%); <6 months: 14/27 (52%) vs. ≥6 months: 4/8 (50%) [37]). However, Aspirot et al. did find a greater number of patients with same day extubation in patients <3 month of age at surgery compared to those ≥3 months of age [3/5 (60%) vs. 11/11 (100%), respectively] [34]. Finally, non-comparative study results indicate that all 15 patients who underwent elective surgery at <1 month of age required post-operative ventilation [1, 42], and that only 5% (7/147) of patients <4 months of age required ventilation following their elective surgery [41].
Secondary outcome: blood loss
Average blood loss was minimal overall, but on average was higher in older patients (≤1 month: 10.7 ± 9.0 g vs. >1 month: 12.5 ± 13.3 g [38]; <3 months: 5.5 ± 6.4 cc vs. ≥3 months: 8.2 ± 2.5 cc [34]). In contrast, Boubnova et al. [36] found that patients <6 months of age were more likely to require a blood transfusion for their resection compared to those ≥6 months of age at surgery [2/8 (25%) vs. 0/10 (0%), respectively].
Patients <4 months of age rarely required blood transfusion, with only one open and three thoracoscopic elective surgery patients receiving a transfusion intraoperatively in the study conducted by Laje et al. (3%, 4/147) [41].
Secondary outcome: pulmonary function
Pulmonary function was found to be normal in the vast majority of patients who underwent elective resection of their asymptomatic CPAM lesions. Prospective comparative evaluation of patients revealed that FVC and total lung capacity (TLC) were normal for all patients who underwent surgery before and after 24 months of age. And while FEV1 was normal in the one patient who underwent surgery at ≥24 months of age, FEV1 was abnormal in 50% (4/8) of patients who had surgery at <24 months of age [39]. Cardiopulmonary exercise testing indicated that power was fairly similar between operative groups (<24 months: 91.7 ± 8.9% predicted vs. ≥24 months: 88.0% predicted), but that the maximum rate of oxygen consumption during incremental exercise (maximal oxygen uptake; VO2 max) was superior in patients who were <24 months of age at the time of surgery (<24 months: 42.2 ± 6.3 L min vs. ≥24 months: 34.9 L min) [39].
A non-comparative assessment of children undergoing lung resection at <12 months of age revealed that almost all had normal pulmonary function 5–11 years after surgery [43]. Diffusion capacity, respiratory muscle strength, FVC, and TLC were normal for all children tested. FEV1 was only abnormal in one evaluated patient [1/5 (20%)] who had a pre-existing diagnosis of asthma [43].
Secondary outcome: histological assessments
Histological evaluation of resected specimens indicated that waiting until ≥6 months of age to perform surgery resulted in a greater number of infections [<6 months: n = 1/5 (20%) versus ≥6 months: n = 4/8 (50%)] [4]. Non-comparative histological assessment of resection specimens from patients submitted to elective surgery at 3 months of age showed that 50% (n = 3/6) were already inflamed [44].
Secondary outcome: mortality
Five studies representing four different age group comparisons evaluated mortality [7, 34, 37, 41, 42]. Overall, no mortalities were recorded for asymptomatic CPAM patients who underwent elective operative treatment for their lesions (≤1 month: 0/2 vs. >1 month: 0/34 [7, 42]; <3 months: 0/24 vs. ≥3 months: 0/27 [34, 37]; <6 months: 0/27 vs. ≥6 months: 0/8 [37], and <4 months: 0/147) [41].
Discussion
Uncertainty regarding the optimal age for elective resection of asymptomatic CPAM has been documented for at least the past 25 years, yet to the best of our knowledge this is the first systematic review to address this question. Meta-analysis of the primary outcome, complications, indicates that while patients who are older at the time of surgery appear to be favoured compared to their younger counterparts, the differences are not statistically significant. These results seem to be in conflict with the belief that as patients get older there exists a higher incidence of inflammation/infection, making surgery more technically difficult [4, 6, 45]. Histological results from this systematic review support that older asymptomatic patients have an increased incidence of inflammation and infection, yet this did not appear to result in increased complications, as predicted. Our findings may reflect improvements in both thoracic surgery (e.g. minimally invasive techniques) and anaesthesia in paediatric patients, resulting in minimal morbidity for these elective surgeries regardless of patient age [30, 42]. It is noteworthy that complication rates in the included studies were consistently higher than the commonly cited value of 8.5–9% [6, 30, 46]; however, this may be due to variations in the definition of complications used by the authors [34].
Overall length of pleural drainage showed only borderline statistical significance (P = 0.05) for patients ≥6 months of age. Chest drains are common following lung resection, as pleural drainage is required when complications such as air leaks and pleural effusion arise. Because air leaks are the most frequent post-operative complication experienced by patients [47], with up to 50% of patients experiencing air leaks following lung resection [48], it was anticipated that the overall trend observed in differences in chest tube duration would be similar to that of overall complications.
While no statistically significant difference was observed in LOS for the 6-month thoracoscopic surgery study, open surgery within this age group, as well as overall meta-analysis for 3-month and 6-month age groups, significantly favoured older patients. Although unexpected, examination of additional factors that influence LOS may explain these meta-analysis results. For example, one of the main reasons for prolonged LOS is chest tube duration [49], which favoured patients ≥6 months of age in a borderline statistically significant manner in the overall meta-analysis and following open surgery, but not following thoracoscopic surgery. In addition, comparative evidence favours patients ≥3 months of age for same day extubation [34], and ≥6 months of age for blood transfusion requirement [36] (where blood transfusion was not considered a complication in any study).
Ventilation outcomes, including length of ventilation and number of patients requiring ventilation, did not differ between older and younger operative age groups. This is contrary to the long-held belief that older children are less likely to require ventilator support [6, 34], with some authors citing patient age as an indication for continued post-operative ventilation, in addition to the need for transfusion and intraoperative cardiorespiratory stability [34]. With only one comparative paper evaluating ventilation outcomes [37], assessing only a small number of patients for which individual patient data was not provided, it is difficult to postulate why no observed differences were seen between younger and older patients.
The only outcome for which younger patients were favoured was blood loss, which may be attributed to the reduced incidence of inflammation/infection observed in these patients. Furukawa et al. found that the disturbance of adhesions and lung abscess formations after infectious episodes to get good operative view is often the main cause of wider skin incision, longer operative times, and larger amounts of bleeding [38].
Pulmonary function following recovery from surgery was predominately normal for patients who underwent elective lung resection, whether they were <12 months, <24 months, or ≥24 months of age at the time of the operation. Most likely this is attributed to lung growth, the nature of which is not yet clear. Thus, it appears as though impaired pulmonary function is uncommon unless more than one lobe is resected [50].
Death resulting from pulmonary resection is rare [34, 42], and in centres with expertise and experience, CPAM lesions can be safely resected with virtually no mortality [51]. This is further supported by the results of this systematic review for which no mortalities were recorded in any study.
Limitations and future directions
There are several limitations to our systematic review. We were not able to examine publication bias in our primary outcome as there was only ever a maximum of three studies in a single meta-analysis. A lack of patients and studies also meant that meta-analysis was only ever possible in the 3- and 6-month age groups. Additionally, further examination of outcomes based on type of surgery (open or thoracoscopic) was infrequently possible. As a result, it is difficult to assess if and how type of surgery affects outcomes. Only English language studies were included and therefore it is possible that relevant studies published in other languages are missing from our analysis. The design of included studies also limits the conclusions as all but one study was retrospective in nature, allowing for the introduction of error due to confounding and bias. Also, most studies combined multiple patient populations, such as emergency and elective CPAM resection, and different types of lung lesions (e.g. bronchopulmonary sequestration, congenital lobar emphysema, etc.). This necessitated corresponding with authors in 85% (n = 17/20) of studies that met inclusion criteria to obtain data pertaining only to asymptomatic CPAM patients, and excluding 35% (n = 6/17) of those studies as authors were not able to provide requested data. Additionally, the comparison of only one age group (e.g. before or after 6 months of age) in all but one study makes it difficult to fully assess how age impacts patient outcomes following elective resection of CPAM. Overall, our systematic review suffered from small patient numbers and studies that were underpowered, and therefore it was difficult to form conclusive recommendations for practice. In the future, it is recommended that authors focus only on elective CPAM resection, and investigate multiple age comparisons, allowing for a more comprehensive assessment of the effect of age on clinical outcomes.
Conclusion
Surgical treatment was found to be safe in all age groups, with no recorded mortalities at any age and similar odds of complications between patients undergoing surgery before and after 1, 3, and 6 months of age. While meta-analysis results significantly favoured older patients for LOS (≥3 months and ≥6 months) and chest tube duration (≥6 months), the included evidence was not sufficient to make a conclusive recommendation as to what the exact optimal age is for elective resection of asymptomatic CPAM.
Abbreviations
- CPAM:
-
Congenital pulmonary airway malformation
- PRISMA:
-
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- LOS:
-
Length of hospital stay
- CAPS EBR:
-
Canadian Association of Paediatric Surgeons Evidence-Based Resource
- FVC:
-
Forced vital capacity
- FEV1 :
-
Forced expiratory volume in 1 s
- OR:
-
Odds ratio
- 95% CI:
-
95% confidence interval
- MD:
-
Mean difference
References
Waszak P, Claris O, Lapillonne A, Picaud JC, Basson E, Chappuis JP, Salle BL (1999) Cystic adenomatoid malformation of the lung: neonatal management of 21 cases. Pediatr Surg Int 15:326–331
Laje P, Liechty KW (2008) Postnatal management and outcome of prenatally diagnosed lung lesions. Prenat Diagn 28:612–618
Laberge JM, Puligandla P, Flageole H (2005) Asymptomatic congenital lung malformations. Semin Pediatr Surg 14:16–33
Calvert JK, Lakhoo K (2007) Antenatally suspected congenital cystic adenomatoid malformations of the lung: postnatal investigation and timing of surgery. J Pediatr Surg 42:411–414
Davenport M, Warne SA, Cacciaguerra S, Patel S, Greenough A, Nicolaides K (2004) Current outcome of antenatally diagnosed cystic lung disease. J Pediatr Surg 39:549–556
Khosa JK, Leong SL, Borzi PA (2004) Congenital cystic adenomatoid malformation of the lung: indications and timing of surgery. Pediatr Surg Int 20:505–508
Kim YT, Kim JS, Park JD, Kang CH, Kim JH (2005) Treatment of congenital cystic adenomatoid malformation - does resection in the early postnatal period increase surgical risk? Eur J Cardiothorac Surg 27:658–661
Miller JA, Corteville JE, Langer JC (1996) Congenital cystic adenomatoid malformation in the fetus: natural history and predictors of outcome. J Pediatr Surg 31:805–808
Sapin E, Lejeune V, Barbet JP, Carricaburu E, Lewin F, Baron JM, Barbotin-Larrieu F, Helardot PG (1997) Congenital adenomatoid disease of the lung: prenatal diagnosis and perinatal management. Pediatr Surg Int 12:126–129
Kapralik J, Wayne C, Chan E, Nasr A (2016) Surgical versus conservative management of congenital pulmonary airway malformation in children: a systematic review and meta-analysis 48. J Pediatr Surg 51:508–512
Khalek N, Johnson MP (2013) Management of prenatally diagnosed lung lesions. Semin Pediatr Surg 22:24–29
Liao SL, Lai SH, Hsueh C, Wong KS (2010) Comparing late-onset and neonatally-diagnosed congenital cystic adenomatoid malformations of the lung. Chang Gung Med J 33:36–43
Chuang S, Sugo E, Jaffe A (2009) A review of postnatal management of congenital pulmonary airway malformations. Fetal Matern Med Rev 20:179–204
Thurlbeck WM (1982) Postnatal human lung growth. Throax 37:564–571
Zeltner TB, Caduff JH, Gehr P, Pfenninger J, Burri PH (1987) The postnatal development and growth of the human lung. I. Morphometry. Respir Physiol 67:247–268
Zeltner TB, Burri PH (1987) The postnatal development and growth of the human lung. II. Morphology. Respir Physiol 67:269–282
Joshi S, Kotecha S (2007) Lung growth and development. Early Hum Dev 83:789–794
Laberge JM, Bratu I, Flageole H (2004) The management of asymptomatic congenital lung malformations. Paediar Respir Rev 5(Suppl A):S305–S312
Adzick NS (2003) Management of fetal lung lesions. Clin Perinatol 30:481–492
Frush DP, Donnelly LF, Rosen NS (2003) Computed tomography and radiation risk: what pediatric health care providers should know. Pediatrics 112:951–957
Eber E (2007) Antenatal diagnosis of congenital thoracic malformations: early surgery, late surgery or no surgery? Semin Respir Crit Care Med 28:355–366
PROSPERO: International prospective register of systematic reviews. University of York, centre for Reviews and Dissemination
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analysis: the PRISMA statement. Ann Intern Med 151:264–269
Linstone HA, Turoff M (1975) Delphi method: techniques and applications. Addison-Wesley Publishing, Boston, MA
Canadian Association of Paediatric Surgeons Evidence-Based Resource. Canadian Association of Paediatric Surgeons
Khangura S, Konnyu K, Cushman R, Grimshaw J, Moher D (2012) Evidence summaries: the evolution of a rapid review approach 17. Systematic Reviews 1:10
Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, Chipponi J (2003) Methodological index for non-randomized studies (MINORS): development and validation of a new instrument 18. ANZ J Surg 73:712–716
The Nordic Cochrane Centre. Review Manager (RevMan). [5.3]. 2014. The Cochrane Collaboration. Ref Type: Computer Program
Komori K, Kamagata S, Hirobe S, Toma M, Okumura K, Muto M, Kasai S, Hayashi A, Suenaga M, Miyakawa T (2009) Radionuclide imaging study of long-term pulmonary function after lobectomy in children with congenital cystic lung disease. J Pediatr Surg 44:2096–2100
Colon N, Schlegel C, Pietsch J, Chung DH, Purcell Jackson G (2012) Congenital lung anomalies: can we postpone resection? J Pediatr Surg 47:87–92
Keijzer R, Chiu PPL, Ratjen F, Langer JC (2009) Pulmonary function after early vs. late lobectomy during childhood: a preliminary study. J Pediatr Surg 44:893–895
Rothenberg S, Middlesworth W, Kadennhe-Chiweshe A, Aspelung G, Kuenzler K, Cowles R, Bodenstein L, Kay S, Shipman K, Rothenberg C, Stolar C, Stylianos S (2015) Two decades of experience with thoracoscopic lobectomy in infants and children: standardizing techniques for advanced thoracoscopic surgery. J Laparoendosc Adv Surg Tech 25:423–428
Rothenberg SS, Kuenzler KA, Middlesworth W, Kay S, Yoder S, Shipman K, Rodriguez R, Stolar CJ (2011) Thoracoscopic lobectomy in infants less than 10 kg with prenatally diagnosed cystic lung disease. J Laparoendosc Adv Surg Tech 21:181–184
Aspirot A, Puligandla PS, Bouchard S, Su W, Flageole H, Laberge JM (2008) A contemporary evaluation of surgical outcomes in neonates and infants undergoing lung resection. J Pediatr Surg 43:508–512
Aziz D, Langer JC, Tuuha SE, Ryan G, Ein SH, Kim PCW (2004) Perinatally diagnosed asymptomatic congenital cystic adenomatoid malformation: to resect or not? J Pediatr Surg 39:329–334
Boubnova J, Peycelon M, Garbi O, David M, Bonnard A (2011) Thoracoscopy in the management of congenital lung disease in infancy. Surg Endosc 25:593–596
Conforti A, Aloi I, Trucchi A, Morini F, Nahom A, Inserra A, Bagolan P (2009) Asymptomatic congenital cystic adenomatoid malformation of the lung: is it time to operate? J Thorac Cardiovasc Surg 138:826–830
Furukawa T, Kimura O, Sakai K, Higashi M, Fumino S, Aoi S, Tajiri T (2015) Surgical intervention strategies for pediatric congenital cystic lesions of the lungs: a 20-years single-institution experience. J Pediatr Surg 50:2025–2027
Naito Y, Beres A, Lapidus-Krol E, Ratjen F, Langer JC (2012) Does earlier lobectomy result in better long-term pulmonary function in children with congenital lung anomalies? A prospective study. J Pediatr Surg 47:852–856
Kongstad T, Buchvald F, Brenoe J, Petersen BL, Tabor A, Nielsen KG (2012) Radiology, histology and short-term outcome of asymptomatic congenital thoracic malformations. Acta Paediatr 101:155–158
Laje P, Pearson EG, Simpao AF, Rehman MA, Sinclair T, Hedrick HL, Adzick NS, Flake AW (2015) The first 100 infant thoracoscopic lobectomies: observations through the learning curve and comparison to open lobectomy. J Pediatr Surg 50:1811–1816
Chen HW, Hsu WM, Lu FL, Chen PC, Jeng SF, Peng SSF, Chen CY, Chou HC, Tsao PN, Hsieh Wu (2010) Management of congenital cystic adenomatoid malformation and bronchopulmonary sequestration in newborns. Pediatr Neonatol 51:172–177
Beres A, Aspirot A, Paris C, Berube D, Bouchard S, Laberge J-M, Lands LC, Puligandla P (2011) A contemporary evaluation of pulmonary function in children undergoing lung resection in infancy. J Pediatr Surg 46:829–832
Pelizzo G, Barbi E, Codrich D, Lembo MA, Zennaro F, Bussani R, Schleef J (2009) Chronic inflammation in congenital cystic adenomatoid malformations. An underestimated risk factor? J Pediatr Surg 44:616–619
Saeed A, Kazmierski M, Khan A, McShane D, Gomez A, Aslam A (2013) Congenital lung lesions: preoperative three-dimensional reconstructed CT scan as the definitive investigation and surgical management. Eur J Pediatr Surg 23:53–56
Ayed AK, Owayed A (2003) Pulmonary resection in infants for congenital pulmonary malformation. Chest 124:98–101
Van Schil PE, Hendriks JM, Lauwers P (2014) Focus on treatment complications and optimal management surgery. Translational Lung. Cancer Res 3:181–186
Mueller MR, Marzluf BA (2014) The anticipation and management of air leaks and residual spaces post lung resection. J Thorac Dis 6:271–284
Pompili C, Miserocchi G (2016) Air leak after lung resection: pathophysiology and patients’ implications. J Thorac Dis 8:S46–S54
Chuang S, Sugo E, Jaffe A (2009) A review of postnatal management of congenital pulmonary airway malformation. Fetal Matern Med Rev 20:179–204
Azizkhan RG, Crombleholme TM (2008) Congenital cystic lung disease: contemporary antenatal and postnatal management. Pediatr Surg Int 24:643–657
Acknowledgements
We would like to thank Dr. Margaret Sampson, MLIS, PhD, AHIP, for designing our electronic search strategy. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The authors have no conflicts of interest relevant to this article to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sullivan, K.J., Li, M., Haworth, S. et al. Optimal age for elective surgery of asymptomatic congenital pulmonary airway malformation: a meta-analysis. Pediatr Surg Int 33, 665–675 (2017). https://doi.org/10.1007/s00383-017-4079-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-017-4079-2