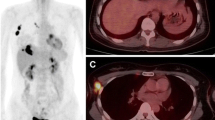
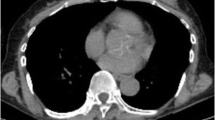
Abstract
Objectives
To evaluate the association between tumour FDG uptake on preoperative PET/CT and axillary lymph node metastasis (ALNM) according to breast cancer subtype.
Methods
The records of 671 patients with invasive breast cancer who underwent 18 F-FDG PET/CT and surgery were reviewed. Using immunohistochemistry, tumours were divided into three subtypes: oestrogen receptor (ER)-positive/human epidermal growth factor receptor 2 (HER2)-negative, HER2-positive, and triple-negative. Tumour FDG uptake, expressed as maximum standardized uptake value (SUVmax), and clinicopathological variables were analysed.
Results
ALNM was present in 187 of 461 ER-positive/HER2-negative, 54 of 97 HER2-positive, and 38 of 113 triple-negative tumours. On multivariate analysis, high tumour SUVmax (≥4.25) (P < 0.001), large tumour size (>2 cm) (P = 0.003) and presence of lymphovascular invasion (P < 0.001) were independent variables associated with ALNM. On subset analyses, tumour SUVmax maintained independent significance for predicting ALNM in ER-positive/HER2-negative (adjusted odds ratio: 3.277, P < 0.001) and HER2-positive tumours (adjusted odds ratio: 14.637, P = 0.004). No association was found for triple-negative tumours (P = 0.161).
Conclusions
Tumour SUVmax may be an independent prognostic factor for ALNM in patients with invasive breast cancer, especially in ER-positive/HER2-negative and HER2-positive subtypes, but not in those with triple-negative subtype.
Key points
• Tumour SUVmax could be an imaging biomarker for predicting ALNM
• Tumour SUVmax predicting ALNM is effective in ER-positive/HER2-negative and HER2-positive subtypes
• Tumour SUVmax predicting ALNM is inaccurate in triple-negative subtypes
• Accurate prognostic prediction based on molecular subtype may facilitate individualized management




Similar content being viewed by others
References
Weiss RB, Woolf SH, Demakos E et al (2003) Natural history of more than 20 years of node-positive primary breast carcinoma treated with cyclophosphamide, methotrexate, and fluorouracil-based adjuvant chemotherapy: a study by the cancer and leukemia group B. J Clin Oncol 21:1825–1835
Arriagada R, Le MG, Dunant A, Tubiana M, Contesso G (2006) Twenty‐five years of follow‐up in patients with operable breast carcinoma. Cancer 106:743–750
Colleoni M, Rotmensz N, Maisonneuve P et al (2007) Prognostic role of the extent of peritumoral vascular invasion in operable breast cancer. Ann Oncol 18:1632–1640
Viale G, Zurrida S, Maiorano E et al (2005) Predicting the status of axillary sentinel lymph nodes in 4351 patients with invasive breast carcinoma treated in a single institution. Cancer 103:492–500
Yoshihara E, Smeets A, Laenen A et al (2013) Predictors of axillary lymph node metastases in early breast cancer and their applicability in clinical practice. Breast 22:357–361
Carey LA, Perou CM, Livasy CA et al (2006) Race, breast cancer subtypes, and survival in the carolina breast cancer study. JAMA 295:2492–2502
Van’t Veer LJ, Dai H, De Vijver V et al (2002) Gene expression profiling predicts clinical outcome of breast cancer. Nature 415:530–536
Nguyen PL, Taghian AG, Katz MS et al (2008) Breast cancer subtype approximated by estrogen receptor, progesterone receptor, and HER-2 is associated with local and distant recurrence after breast-conserving therapy. J Clin Oncol 26:2373–2378
Desmedt C, Haibe-Kains B, Wirapati P et al (2008) Biological processes associated with breast cancer clinical outcome depend on the molecular subtypes. Clin Cancer Res 14:5158–5165
Sánchez-Muñoz A, García-Tapiador AM, Martínez-Ortega E et al (2008) Tumour molecular subtyping according to hormone receptors and HER2 status defines different pathological complete response to neoadjuvant chemotherapy in patients with locally advanced breast cancer. Clin Transl Oncol 10:646–653
de Ronde JJ, Hannemann J, Halfwerk H et al (2010) Concordance of clinical and molecular breast cancer subtyping in the context of preoperative chemotherapy response. Breast Cancer Res Treat 119:119–126
Fuster D, Duch J, Paredes P et al (2008) Preoperative staging of large primary breast cancer with [18 F] fluorodeoxyglucose positron emission tomography/computed tomography compared with conventional imaging procedures. J Clin Oncol 26:4746–4751
Eubank WB, Mankoff D, Bhattacharya M et al (2004) Impact of FDG PET on defining the extent of disease and on the treatment of patients with recurrent or metastatic breast cancer. AJR Am J Roentgenol 183:479–486
Hatt M, Groheux D, Martineau A et al (2013) Comparison between 18 F-FDG PET image-derived indices for early prediction of response to neoadjuvant chemotherapy in breast cancer. J Nucl Med 54:341–349
Song B, Hong CM, Lee HJ et al (2011) Prognostic value of primary tumor uptake on F-18 FDG PET/CT in patients with invasive ductal breast cancer. Nucl Med Mol Imaging 45:117–124
Ueda S, Tsuda H, Asakawa H et al (2008) Clinicopathological and prognostic relevance of uptake level using 18 F-fluorodeoxyglucose positron emission tomography/computed tomography fusion imaging (18 F-FDG PET/CT) in primary breast cancer. Jpn J Clin Oncol 38:250–258
Koo HR, Park JS, Kang KW et al (2014) 18 F-FDG uptake in breast cancer correlates with immunohistochemically defined subtypes. Eur Radiol 24:610–618
Greene FL (2002) AJCC cancer staging manual. Springer
Elston C, Ellis I (1991) Pathological prognostic factors in breast cancer. I. the value of histological grade in breast cancer: experience from a large study with long‐term follow‐up. Histopathology 19:403–410
Allred DC, Harvey JM, Berardo M, Clark GM (1998) Prognostic and predictive factors in breast cancer by immunohistochemical analysis. Mod Pathol 11:155–168
Moeder CB, Giltnane JM, Harigopal M et al (2007) Quantitative justification of the change from 10 % to 30 % for human epidermal growth factor receptor 2 scoring in the american society of clinical oncology/college of american pathologists guidelines: tumor heterogeneity in breast cancer and its implications for tissue microarray based assessment of outcome. J Clin Oncol 25:5418–5425
Yamashita H, Toyama T, Nishio M et al (2006) p53 protein accumulation predicts resistance to endocrine therapy and decreased post-relapse survival in metastatic breast cancer. Breast Cancer Res 8:R48
Cheang MC, Chia SK, Voduc D et al (2009) Ki67 index, HER2 status, and prognosis of patients with luminal B breast cancer. J Natl Cancer Inst 101:736–750
Bader AA, Tio J, Petru E et al (2002) T1 breast cancer: identification of patients at low risk of axillary lymph node metastases. Breast Cancer Res Treat 76:11–17
Ridriguez-Pinilla SM, Sarrio D, Honrado E et al (2006) Prognostic significance of basal-like phenotype and fascin expression in node-negative invasive breast cancers. Clin Cancer Res 12:1533–1539
Krag DN, Anderson SJ, Julian TB et al (2007) Technical outcomes of sentinel-lymph-node resection and conventional axillary-lymph-node dissection in patients with clinically node-negative breast cancer: results from the NSABP B-32 randomised phase III trial. Lancet Oncol 8:881–888
McMasters KM, Tuttle TM, Carlson DJ et al (2000) Sentinel lymph node biopsy for breast cancer: a suitable alternative to routine axillary dissection in multi-institutional practice when optimal technique is used. J Clin Oncol 18:2560–2566
Wilke LG, McCall LM, Posther KE et al (2006) Surgical complications associated with sentinel lymph node biopsy: results from a prospective international cooperative group trial. Ann Surg Oncol 13:491–500
Valente SA, Levine GM, Silverstein MJ et al (2012) Accuracy of predicting axillary lymph node positivity by physical examination, mammography, ultrasonography, and magnetic resonance imaging. Ann Surg Oncol 19:1825–1830
Kim J, Lee J, Chang E et al (2009) Selective sentinel node plus additional non-sentinel node biopsy based on an FDG-PET/CT scan in early breast cancer patients: single institutional experience. World J Surg 33:943–949
Veronesi U, De Cicco C, Galimberti VE et al (2007) A comparative study on the value of FDG-PET and sentinel node biopsy to identify occult axillary metastasis. Ann Oncol 18:473–478
Gil-Rendo A, Zornoza G, Garcia-Velloso MJ et al (2006) Fluorodeoxyglucose positron emission tomography with sentinel lymph node biopsy for evaluation of axillary involvement in breast cancer. Br J Surg 93:707–712
Fletcher JW, Djulbegovic B, Soares HP et al (2008) Recommendations on the use of 18 F-FDG PET in oncology. J Nucl Med 49:480–508
Groves AM, Shastry M, Rodriguez-Justo M et al (2011) 18 F-FDG PET and biomarkers for tumour angiogenesis in early breast cancer. Eur J Nucl Med Mol Imaging 38:46–52
Song BI, Lee SW, Jeong SY et al (2012) 18 F-FDG uptake by metastatic axillary lymph nodes on pretreatment PET/CT as a prognostic factor for recurrence in patients with invasive ductal breast cancer. J Nucl Med 53:1337–1344
Carey LA, Dees EC, Sawyer L et al (2007) The triple negative paradox: primary tumor chemosensitivity of breast cancer subtypes. Clin Cancer Res 13:2329–2334
Dent R, Trudeau M, Pritchard KI et al (2007) Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res 13:4429–4434
Voduc KD, Cheang MC, Tyldesley S, Gelmon K, Nielsen TO, Kennecke H (2010) Breast cancer subtypes and the risk of local and regional relapse. J Clin Oncol 28:1684–1691
Vicente AMG, Castrejón ÁS, Martín AL et al (2013) Molecular subtypes of breast cancer: metabolic correlation with 18 F-FDG PET/CT. Eur J Nucl Med Mol Imaging 40:1304–1311
Hugh J, Hanson J, Cheang MC et al (2009) Breast cancer subtypes and response to docetaxel in node-positive breast cancer: use of an immunohistochemical definition in the BCIRG 001 trial. J Clin Oncol 27:1168–1176
Jatoi I, Hilsenbeck SG, Clark GM, Osborne CK (1999) Significance of axillary lymph node metastasis in primary breast cancer. J Clin Oncol 17:2334–2340
Acknowledgments
The scientific guarantor of this publication is Jin You Kim. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was waived by the Institutional Review Board. No study subjects or cohorts have been previously reported. Methodology: retrospective, observational, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, J.Y., Lee, S.H., Kim, S. et al. Tumour 18 F-FDG Uptake on preoperative PET/CT may predict axillary lymph node metastasis in ER-positive/HER2-negative and HER2-positive breast cancer subtypes. Eur Radiol 25, 1172–1181 (2015). https://doi.org/10.1007/s00330-014-3452-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3452-y