Abstract
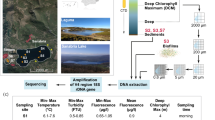
The molecular diversity of freshwater microeukaryotes, particularly phytoplankton, in the Arctic Svalbard, has been relatively unexplored. Freshwater algae are considered biological indicators of environmental change and can be useful in assessing the impact of global climate change and increased environmental pollution. In this study, freshwater microeukaryotes in an Arctic reservoir at Ny-Ǻlesund (Svalbard, Norway) were studied using the hypervariable V1–V3 small subunit rRNA and 454 pyrosequencing. On the basis of 8,956 reads, we revealed high genetic diversity in eukaryotes, representing all known eukaryotic supergroups, except Excavata. “Chromalveolata” (previously supergroup Chromalveolata) and Archaeplastida were the most and least abundant supergroups, respectively. After data mining, 57 phylotypes were detected from 7,398 pyrosequences. They were dominated by stramenopiles (84 %) and Dinoflagellata (13 %), with minor contributions from Cryptophyta, Chlorophyta, and Telonemida. The detection of algae belonging to the orders Mamiellales and Monomastigales provides a window into a fraction of the ‘rare biosphere’ that had previously been undetected in such environments. Interestingly, no haptophytes were recorded. Stramenopiles and Dinoflagellata mainly comprised taxa belonging to the families Chrysophyceae, Synurophyceae, and Dinophyceae. On the basis of the proportion of operational taxonomic units, the dominant phylotypes were found to include Ochromonas spp., Mallomonas spp., and Uroglena americana. These results demonstrate the significance of a chrysophyte-dominated microeukaryotic community, which is of great potential for future studies in terms of reconstruction of past climate trends and monitoring of environmental change in the Arctic.



Similar content being viewed by others
References
Adl SM, Simpson AGB, Farmer MA, Andersen RA, Anderson OR, Barta JR, Bowser SS, Brugerolle G, Fensome RA, Fredericq S, James TY, Karpov S, Kugrens P, Krug J, Lane CE, Lewis LA, Lodge J, Lynn DH, Mann DG, McCourt RM, Mendoza L, Moestrup O, Mozley-Standridge SE, Nerad TA, Shearer CA, Smirnov AV, Spiegel FW, Taylor MF (2005) The new higher level classification of eukaryotes with emphasis on the taxonomy of protists. J Eukaryot Microbiol 52:399–451
Adl SM, Simpson AGB, Lane CE, Lukeš J, Bass D, Bowser SS, Brown MW, Burki F, Dunthorn M, Hampl V, Heiss A, Hoppenrath M, Lara E, Le Gall L, Lynn DH, McManus H, Mitchell EA, Mozley-Stanridge SE, Parfrey LW, Pawlowski J, Rueckert S, Shadwick RS, Schoch CL, Smirnov A, Spiegel FW (2012) The revised classification of Eukaryotes. J Eukaryot Microbiol 59:429–514
Annenkova NV (2013) Phylogenetic relations of the dinoflagellate Gymnodinium baicalense from Lake Baikal. Cent Eur J Biol 8:366–373
Betts-Piper AM, Zeeb BA, Smol JP (2004) Distribution and autecology of chrysophyte cysts from High Arctic Svalbard lakes: preliminary evidence of recent environmental change. J Paleolimnol 31:467–481
Bonilla S, Villeneuve V, Vincent WF (2005) Benthic and planktonic algal communities in a High Arctic lake: pigment structure and contrasting responses to nutrient enrichment. J Phycol 41:1120–1130
Bråte J, Klaveness D, Rygh T, Jakobsen KS, Shalchian-Tabrizi K (2010) Telonemia-specific environmental 18S rDNA PCR reveals unknown diversity and multiple marine-freshwater colonizations. BMC Microbiol 10:168
Cavalier-Smith T, Lewis R, Chao EE, Oatesb B, Bass D (2009) Helkesimastix marina n. sp. (Cercozoa: Sainouroidea superfam. n.) a gliding zooflagellate of novel ultrastructure and unusual ciliary behaviour. Protist 160:452–479
Charvet S, Vincent WF, Lovejoy C (2012) Chrysophytes and other protists in High Arctic lakes; molecular gene surveys, pigment signatures and microscopy. Polar Biol 35:733–748
Chou HH, Holmes MH (2001) DNA sequences quality and trimming and vector removal. Bioinformatics 17:1093–1104
Douglas MSV, Smol JP (1999) Freshwater diatoms as indicators of environmental change in the High Arctic. In: Stoermer EF, Smol JP (eds) The diatoms: applications for the environmental and earth sciences, Cambridge University Press, Cambridge, pp 227–243
Faria DG, Lee MD, Lee JB, Lee J, Chang M, Youn SH, Suh YU, Ki JS (2014) Molecular diversity of phytoplankton in the East China Sea around Jeju Island (Korea) unraveled by pyrosequencing. J Oceanogr 70:11–23
Gerland S, Winther JG, Ørbæk JB, Ivanov BV (1999) Physical properties, spectral reflectance and thickness development of first year fast ice in Kongsfjorden, Svalbard. Polar Res 18:275–282
Git A, Dvinge H, Salmon-Divon M, Osborne M, Kutter C, Hadfield J, Bertone P, Caldas C (2010) Systematic comparison of microarray profiling, real-time PCR, and next-generation sequencing technologies for measuring differential microRNA expression. RNA 16:991–1006
Harder T, Lau CKS, Dobretsov S (2003) A distinctive epibiotic bacterial community on the soft coral Dendrophthya sp., and antibacterial activity of coral tissue extracts suggest a chemical mechanism against bacterial epibiosis. FEMS Microbiol Ecol 43:337–347
Hert DG, Fredlake CP, Barron AE (2008) Advantages and limitations of next generation sequencing technologies: a comparison of electrophoresis and non-electrophoresis methods. Electrophoresis 29:4618–4626
Holmgren SK (1984) Experimental lake fertilization in the Kuokkel area, Northern Sweden: phytoplankton biomass and algal composition in natural and fertilized subarctic lakes. Int Revue ges Hydrobiol Hydrogr 69:781–817
Hop H, Pearson T, Hegseth EN, Kovacs KM, Wiencke C, Kwasniewski S, Eiane K, Mehlum F, Gulliksen B, Wlodarska-Kowalczuk M, Lydersen C, Weslawski JM, Cochrane S, Gabrielsen GW, Leakey RJG, Lønne OJ, Zajaczkowski M, Falk-Petersen S, Kendall M, Wängberg S-Å, Bischof K, Voronkov AY, Kovaltchouk NA, Wiktor J, Poltermann M, di Prisco G, Papucci C, Gerland S (2002) The marine ecosystem of Kongsfjorden, Svalbard. Polar Res 21:167–208
Huber JA, Mark Welch DB, Morrison HG, Huse SM, Neal PR, Butterfield DA, Sogin ML (2007) Microbial population structures in the deep marine biosphere. Science 318:97–100
Jones VJ, Birks HJB (2004) Lake-sediment records of recent environmental change on Svalbard: results of diatom analysis. J Paleolimnol 31:445–466
Kaštovská K, Elster J, Stibal M, Šantrůčková H (2005) Microbial assemblages in soil microbial succession after glacial retreat in Svalbard (High Arctic). Microb Ecol 50:396–407
Ki JS (2012) Hypervariable regions (V1–V9) of the dinoflagellate 18S rRNA using a large dataset for marker considerations. J Appl Phycol 24:1035–1043
Ki JS, Kang SH, Jung SW, Park BS, Han MS (2006) A study on the freshwater algal flora occurring in temporary ponds around the Dasan Arctic Station, Ny-Ǻlesund, (Norway), and the molecular characteristics of Chlamydomonas 18S rDNA. Ocean Polar Res 28:107–117
Ki JS, Cho SY, Katano T, Jung SW, Lee J, Park BS, Kang SH, Han MS (2009) Comprehensive comaprisons of three pennate diatoms Diatoma tenuae, Fragilaria vaucheriae, and Navicula pelliculosa, isolated from summer Arctic reservoirs (Svalbard 79oN), by fine-scale morphology and nuclear 18S ribosomal DNA. Polar Biol 32:147–159
Kim GH, Klochkova TA, Kang SH (2008) Notes on freshwater and terrestrial algae from Ny-Ǻlesund, Svalbard (High Arctic sea area). J Environ Biol 29:485–491
Kim GH, Klochkova TA, Han JW, Kang SH, Choi HG, Chung KW, Kim SJ (2011) Freshwater and terrestrial algae from at Ny-Ǻlesund and Blomstrandhalvøya Island (Svalbard). Arctic 64:25–31
Lefranc M, Thénot A, Lepère C, Debroas D (2005) Genetic diversity of small eukaryotes in lakes differing by their trophic status. Appl Environ Microbiol 71:5935–5942
Lenzenweger R, Lütz C (2006) A contribution to knowledge of the desmid flora (Desmidiaceae, Zygnemaphyceae) of Spitzbergen. Algol Stud 119:79–89
Lepère C, Boucher D, Jardillier L, Domaizon I, Debroas D (2006) Succession and regulation factors of small eukaryote community composition in a lacustrine ecosystem (Lake Pavin). Appl Environ Microbiol 72:2971–2981
Lepère C, Domaizon I, Debroas D (2008) Unexpected importance of potential parasites in the composition of the Freshwater Small-eukaryote community. Appl Environ Microbiol 74:2940–2949
Lund TR (1983) Hydrography and production of phytoplankton in a glacier affected watercourse at Svalbard (in Norwegian). Dissertation, University of Oslo
Monchy S, Grattepanche JD, Breton E, Meloni D, Sanciu G, Chabé M, Delhaes L, Viscogliosi E, Sime-Ngando T, Christaki U (2012) Microplanktonic community structure in a coastal system relative to a Phaeocystis Bloom inferred from morphological and tag pyrosequencing methods. PLoS One 7:e39924
Mueller DR, Vincent WF, Bonilla S, Laurion I (2005) Extremotrophs, extremophiles and broadband pigmentation strategies in a high arctic ice shelf ecosystem. FEMS Microbiol Ecol 53:73–87
Müller T, Bleiß W, Martin CD, Rogaschewski S, Fuhr G (1998) Snow algae from north Svalbard: their identification, distribution, pigment and nutrient content. Polar Biol 20:14–32
Rose NL, Rose CL, Boyle JF, Appleby PG (2004) Lake-sediment evidence for local and remote sources of atmospherically deposited pollutants on Svalbard. J Paleolimnol 31:499–513
Sorvari S, Korhola A, Thompson R (2002) Lake diatom response to recent Arctic warming in Finnish Lapland. Glob Change Biol 8:171–181
Stoeck T, Behnke A, Richard C, Amaral-Zettler A, Rodriguez-Mora MJ, Chistoserdov A, Orsi W, Edgcomb VP (2009) Massively parallel tag sequencing reveals the complexity of anaerobic marine protistan communities. BMC Biol 7:72
Taib N, Mangot JF, Domaizon I, Bronner G, Debroas D (2013) Phylogenetic affiliation of SSU rRNA genes generated by massively parallel sequencing: new insights into the freshwater protist diversity. PLoS One 8:e58950
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Tarbe A, Stenuite S, Balagué V, Sinyinza D, Descy JP, Massana R (2011) Molecular characterisation of the small-eukaryote community in a tropical Great Lake (Lake Tanganyika, East Africa). Aquat Microb Ecol 62:177–190
Taylor FJR, Hoppenrath M, Saldarriaga JF (2008) Dinoflagellate diversity and distribution. Biodivers Conserv 17:407–418
Thompson JD, Higgins DG, Gibbson TJ (1994) Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Varin T, Lovejoy C, Jungblut AD, Vincent WF, Corbeil J (2012) Metagenomic analysis of stress genes in microbial mat communities from Antarctica and the High Arctic. Appl Environ Microbiol 78:549–559
Yergeau E, Hogues H, Whyte LG, Greer CW (2010) The functional potential of high Arctic permafrost revealed by metagenomic sequencing, qPCR and microarray analyses. ISME J 4:1206–1214
Acknowledgments
We thank the anonymous reviewers for their valuable comments, which helped us to improve the quality of this manuscript. This work was supported by the “Polar Academic Program (PAP)” of the Korea Polar Research Institute (KOPRI) funded to Jang-Seu Ki, by the Korea Institute of Ocean Science and Technology Research Fund (#PE99201), and by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2013R1A1A2013596).
Author information
Authors and Affiliations
Corresponding author
Additional information
Thangavelu Boopathi and Daphne Georgina Faria have equal contribution to this paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Boopathi, T., Faria, D.G., Lee, MD. et al. A molecular survey of freshwater microeukaryotes in an Arctic reservoir (Svalbard, 79°N) in summer by using next-generation sequencing. Polar Biol 38, 179–187 (2015). https://doi.org/10.1007/s00300-014-1576-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-014-1576-9