Abstract
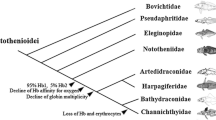
Haemoglobins are sensitive to temperature and their properties mirror the thermal conditions encountered by species during their evolutionary histories. This paper provides data on molecular phylogeny of the haemoglobin chains of Cottoperca gobio, a notothenioid fish of sub-Antarctic latitudes, belonging to the basal family Bovichtidae. Unlike most Antarctic notothenioids, C. gobio has two major haemoglobins sharing the β chain. In the molecular phylogenetic analysis, the β chain is included in the clade of the “embryonic” or minor Antarctic globins. Although, in the majority of notothenioids, “embryonic” (minor) α and β globins are expressed in traces or small amounts in the adult stage, in C. gobio the present analysis supports the occurrence of a complete “switch” to exclusive expression of the embryonic β-globin gene in adult fish. The α and β chains sequences have been used to expand our knowledge of the evolution of notothenioid haemoglobins.





Similar content being viewed by others
References
Bargelloni L, Zane L, Derome N, Lecointre G, Patarnello T (2000) Molecular zoogeography of Antarctic euphausiids and notothenioids: from species phylogenies to intraspecific patterns of genetic variation. Antarct Sci 12:259–268
Brittain T (1987) The Root effect. Comp Biochem Physiol 86B:473–481
Chan FY, Robinson J, Brownlie A, Shivdasani RA, Donovan A, Brugnara C, Kim J, Lau BC, Witkowska HE, Zon LI (1997) Characterization of adult alpha- and beta-globin genes in the zebrafish. Blood 89:688–700
Cheng C-HC, Chen L, Near TJ, Jin Y (2003) Functional antifreeze glycoprotein genes in temperate-water New Zealand nototheniid fish infer an Antarctic evolutionary origin. Mol Biol Evol 20:1897–1908
Cocca E, Ratnayake-Lecamwasam M, Parker SK, Camardella L, Ciaramella M, di Prisco G, Detrich HW (1995) Genomic remnants of alpha-globin genes in the hemoglobinless Antarctic icefishes. Proc Natl Acad Sci USA 92:1817–1821
D’Avino R, di Prisco G (1988) Antarctic fish hemoglobin: an outline of the molecular structure and oxygen binding properties – I. Molecular structure. Comp Biochem Physiol 90B:579–584
D’Avino R, Caruso C, Tamburrini M, Romano M, Rutigliano B, Polverino de Laureto P, Camardella L, Carratore V, di Prisco G (1994) Molecular characterization of the functionally distinct hemoglobins of the Antarctic fish Trematomus newnesi. J Biol Chem 269:9675–9681
Eastman JT (1993) Antarctic fish biology: evolution in a unique environment. Academic Press Inc, San Diego
Eastman JT (2005) The nature of the diversity of Antarctic fishes. Polar Biol 28:93–107
Gillemans N, McMorrow T, Tewari R, Wai AW, Burgtorf C, Drabek D, Ventress N, Langeveld A, Higgs D, Tan-Un K, Grosveld F, Philipsen S (2003) Functional and comparative analysis of globin loci in pufferfish and humans. Blood 101:2842–2849
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Kumar S, Tamura K, Jakobsen I, Nei M (2000) MEGA: molecular evolutionary genetic analysis, Version 2. Pennsylvania State University, University Park and Arizona State University, USA
Last PR, Balushkin AV, Hutchins JB (2002) Halaphritis platycephala (Notothenioidei: Bovichtidae): a new genus and species of temperate icefish from Southeastern Australia. Copeia 2:433–440
Marinakis P, Tamburrini M, Carratore V, di Prisco G (2003) Unique features of the hemoglobin system of the Antarctic notothenioid fish Gobionotothen gibberifrons. Eur J Biochem 270:3981–3987
Maruyama K, Yasumasu S, Iuchi I (2004) Evolution of globin genes of the medaka Oryzias latipes (Euteleostei; Beloniformes; Oryziinae). Mech Dev 121:753–769
Near T, Pesavento JJ, Cheng C-HC (2004) Phylogenetic investigations of Antarctic notothenioid fishes (Perciformes: Notothenioidei) using complete gene sequences of the mitochondrial encoded 16S rRNA. Mol Phylogenet Evol 32:881–891
Pisano E, Cocca E, Mazzei F, Ghigliotti L, di Prisco G, Detrich HW, Ozouf-Costaz C (2003) Mapping of α- and β-globin genes on Antarctic fish chromosomes by fluorescence in-situ hybridization. Chromosome Res 11:633–640
di Prisco G, D’Avino R, Caruso C, Tamburrini M, Camardella L, Rutigliano B, Carratore V, Romano M (1991) The biochemistry of oxygen transport in red-blooded Antarctic fish. In: di Prisco G, Maresca B, Tota B (eds) Biology of Antarctic fish. Springer Verlag, Berlin, pp 263–281
di Prisco G, Cocca E, Parker SK, Detrich HW (2002) Tracking the evolutionary loss of hemoglobin expression by the white-blooded Antarctic icefishes. Gene 295:185–191
Riccio A, Tamburrini M, Carratore V, di Prisco G (2000) Functionally distinct hemoglobins of the cryopelagic Antarctic teleost Pagothenia borchgrevinki. J Fish Biol 57:20–32
Riggs A (1988) The Bohr effect. Ann Rev Physiol 50:181–204
Ritchie PA, Bargelloni L, Meyer A, Taylor JA, Macdonald JA, Lambert M (1996) Mitochondrial phylogeny of trematomid fishes (Nototheniidae, Perciformes) and the evolution of Antarctic fish. Mol Phylogenet Evol 5:383–390
Ruud JT (1954) Vertebrates without erythrocytes and blood pigment. Nature 173:848–850
Stam WT, Beintema JJ, D’Avino R, Tamburrini M, di Prisco G (1997) Molecular evolution of hemoglobins of Antarctic fishes (Notothenioidei). J Mol Evol 45:437–445
Tamburrini M, D’Avino R, Fago A, Carratore V, Kunzmann A, di Prisco G (1996) The unique hemoglobin system of Pleuragramma antarcticum, an Antarctic migratory teleost. Structure and function of the three components. J Biol Chem 271:23780–23785
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The Clustal X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acid Res 25:4876–4882
Verde C, Carratore V, Riccio A, Tamburrini M, Parisi E, di Prisco G (2002) The functionally distinct hemoglobins of the Arctic spotted wolffish Anarhichas minor. J Biol Chem 277:36312–36320
Verde C, Howes BD, De Rosa MC, Raiola L, Smulevich G, Williams R, Giardina B, Parisi E, di Prisco G (2004) Structure and function of the Gondwanian hemoglobin of Pseudaphritis urvillii, a primitive notothenioid fish of temperate latitudes. Prot Sci 13:2766–2781
Acknowledgments
We thank the captain, crew and personnel of Raytheon Polar Services aboard the RVIB Nathaniel B. Palmer for their excellent assistance during the cruise. This study is financially supported by the Italian National Programme for Antarctic Research (PNRA). It is in the framework of the Arctic Strategic Programme of the Italian National Research Council, EVOLANTA, EBA. The ICEFISH cruise was supported by National Science Foundation grant OPP 01-32032 to H. William Detrich (Northeastern University). This is publication number 15 from the ICEFISH Cruise of 2004 (H-W Detrich, Chief Scientist). The manuscript has undergone substantial improvements, thanks to the constructive comments of the reviewers (J. T. Eastman, E. Pisano and H. W. Detrich).
Author information
Authors and Affiliations
Corresponding author
Additional information
The protein sequence data reported in this paper will appear in the UniProt Knowledge base under the accession number: P84652 (β chain), P84653 (α 1 chain).
Rights and permissions
About this article
Cite this article
Giordano, D., Grassi, L., Parisi, E. et al. Embryonic β-globin in the non-Antarctic notothenioid fish Cottoperca gobio (Bovichtidae). Polar Biol 30, 75–82 (2006). https://doi.org/10.1007/s00300-006-0162-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-006-0162-1