Abstract
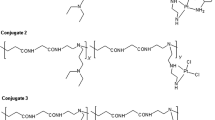
Polyamidoamine conjugates of neridronic acid were synthesized by a one-pot aqueous phase Michael addition reaction of N, N′-methylenebisacrylamide with selected primary and secondary amines and neridronic acid. This reaction involved the use of water and it was performed at room temperature thereby making the reaction environmentally friendly and economically viable. These conjugates are potential prodrugs and they were characterized by nuclear magnetic resonance spectroscopy (NMR), Fourier transform spectroscopy (FTIR), X-ray diffraction (XRD), scanning electron microscope (SEM) and energy-dispersive X-ray analysis (EDX). NMR, TGA, XRD and FTIR confirmed the successful incorporation of the bisphosphonate on to the carriers. The mass percentage incorporation of neridronic acid was found to be between 0.9 and 3.74 percent. 31P NMR exhibited the signal peak for the bisphosphonate at 19.0 ppm.


















Similar content being viewed by others
References
Larson N, Ghandehari H (2012) Polymeric conjugates for drug delivery. Chem Mater 24:840–853
Knop K, Hoogenboom R, Fischer D, Schubert US (2010) Poly(ethylene glycol) in drug delivery: pros and cons as well as potential alternatives. Angew Chem Int Ed 49:6288–6308
Bentley MD, Harris JM, Kozlowski A (1999), “Heterobifunctional Poly(ethylene glycol) Derivatives and Methods for their preparation,” P.C.T. US99/23536
Banerjee SS, Aher N, Patil R, Khandare J (2012), Review Article: Poly(ethylene glycol)-Prodrug Conjugates: Concept, Design, and Applications. J Drug Deliv, Volume 2012, Article ID 103973, 17 pages
Agrawal RN, Joseph A, Mukerji A (2013) Polymeric prodrugs: recent achievements and general strategies. J Antivir Antiretrovir S15:12
Zhou Y, Cheng C, Yang S-T. Production of Malic Acid and Poly(l-malic acid) from Biomass Hydrolysates by Aureobasidium pullulans. http://www3.aiche.org/proceedings/content/Annual-2013/extended-abstracts/P343556.pdf. Accessed 09/04/2014
Richardson S, Ferruti P, Duncan R (1999) Poly(amidoamine)s as Potential Endosomolytic Polymers: evaluation In Vitro and In Vivo Body Distribution in Normal and Tumour-Bearing Animals. J Drug Targeting 6:391–404
Danusso F, Ferruti P (1970) Synthesis of Tertiary Amine Polymers. Polymer 11:88–113
Ferruti P, Marchisio MA, Duncan R (2002) Poly(amidoamine)s: biomedical Applications. Macromol Rapid Commun 23:332–355
Ferruti P, Ranucci E, Sartore L, Bignotti F, Marchisio MA, Bianciardi P, Veronese FM (1994) Recent Results on Functional Polymers and Macromonomers of Interest as Biomaterials or for Biomaterial Modification. Biomaterial 15:1235–1241
N’Da DD, Neuse EW (2006) Polyamidoamines as Drug Carriers: synthesis of Polymers Featuring Extrachain-type Primary Amino Groups as Drug-anchoring Sites. S Afr J Chem 59:65–70
Caldwell G, Neuse EW, Stephanou A (1993) Synthesis of water-soluble polyimidoamines for biomedical application. II. Polymers possessing intrachain-type secondary amino groups suitable for side-chain attachment. J Appl Polym Sci 50:393–401
Cavalli R, Bisazza A, Bussano R, Trotta M, Civra A, Lembo D, Ranucci E, Ferruti P (2011) Poly(amidoamine)–cholesterol conjugate nanoparticles obtained by electrospraying as novel tamoxifen delivery system. J Drug Deliv volume 2011, p 9
Ferruti P, Ranucci E, Trotta F, Gianasi E, Evagorou EG, Wasil M, Wilson G, Duncan R (1999) Synthesis, characterisation and antitumour activity of platinum(II) complexes of novel functionalised poly(amido amine)s. Macromol Chem Phys 200:1644–1654
Ranucci E, Suardi MA, Annunziata R, Ferruti P, Chiellini F, Bartoli C (2008) Poly(amidoamine) conjugates with disulfide-linked cholesterol pendants self-assembling into redox-sensitive nanoparticles. Biomacromol 9:2693–2704
Davide G, Maurizio R, Ombretta V, Luca I, Silvano A (2013) Clinical development of neridronate: potential for new applications. Ther Clin Risk Manag 9:139–147
Quarta L, Corrado A, Melillo N, Trotta A, Scotto G, d’Onofrio F, Santoro N, Cantatore FP (2008) Combined effect of neridronate and specific antibiotic therapy in a case of tuberculous spondylodiscitis. Rheumatol Int 28:495–498
Morabito N, Gaudio A, Lasco A, Catalano A, Atteritano M, Trifiletti A, Anastasi G, Melloni D, Frisina N (2004) Neridronate prevents bone loss in patients receiving androgen deprivation therapy for prostate cancer. J Bone Miner Res 19:1766–1770
Gatti D, Viapiana O, Idolazzi L, Fracassi E, Adami S (2009) Neridronic acid for the treatment of bone metabolic diseases. Expert Opin Drug Metab Toxicol 5:1305–1311
Van Beek ER, Lowik CWGM, Papapoulos SE (2002) Bisphosphonates suppress bone resorption by a direct effect on early osteoclast precursors without affecting the osteoclastogenic capacity of osteogenic cells: the role of protein geranylgeranylation in the action of nitrogen-containing bisphosphonates on osteoclast precursors. Bone 30:64–70
Russell RG (2007) Bisphosphonates: mode of Action and Pharmacology. Paediatrics 119:S150
Dunford JE (2010) molecular targets of the nitrogen containing bisphosphonates: the molecular pharmacology of prenyl synthase inhibition. Curr Pharm Des 16:2961
General Side Effects of Bisphosphonates. http://cancerhelp.cancerresearchuk.org/about-cancer/treatment/bisphosphonate/general-side-effects-of-bisphosphonates. Accessed 11th October 2012
Bisphosphonate Drugs—Side Effects of Bisphosphonate Drugs. http://www.algaecal.com/bisphosphonate-drugs-side-effects.html. Accessed 11th October 2012
Barbucci R (2008) Amido-phosphonate or amido-bisphosphonate derivatives of carboxylated polysaccharides, preparation and biomedical uses thereof. Eur Patent Application, EP 1 994 945 A1
Holmberg A (2011) Modified Hydroxypolymer Publication Classification Conjugates with Bone Seeking and Tumor Killing Moieties. US Patent Application Publication Pub No. US 2011/0118207 A1
Miller K, Erez R, Segal E, Shabat D, Satchi-Fainaro R (2009) Targeting bone metastases with a bispecific anticancer and antiangiogenic polymer–alendronate–taxane conjugate. Angew Chem Int Ed 48:2949–2954
Fresta M, Giammona G, Cavallaro G, Licciardi M, Paolino D (2012) Bisphosphonates–polyaspartamide conjugates as polymeric carrier for drug targeting to bones (From PATENTSCOPE database). World intellectual Property Organization, International Bureau. WO2012/098222 A1. http://patentscope.wipo.int/search/en/search.jsf
Kieczykowski GR, Jobson RB, Melillo DG, Reinhold DF, Grenda VJ, Shinkai I (1995) Preparation of (4-amino-1-hydroxybutylidene) bisphosphonic acid sodium salt, MK-217 (alendronate sodium). An improved procedure for the preparation of 1-hydroxy-1, 1-bisphosphonic acids. J Org Chem 60:8310–8312
Neuse EW (2008) Synthetic Polymers as Drug-Delivery Vehicles in Medicine, Metal-Based Drugs, Hindawi Publishing Corporation, Volume 2008, Article ID 469531, 19 pages. doi:10.1155/2008/469531
Pasut G, Veronese FM (2007) Polymer–drug conjugation, recent achievements and general strategies. Prog Polym Sci 32:933–961
Maeda H, Wu J, Sawa T, Matsumura Y, Hori K (2000) Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release 65:271–284
Ringsdorf H (1975) Structure and properties of pharmacologically active polymers. J Polym Sci Symp 51:135–153
Miller PD (2005) Optimizing the management of postmenopausal osteoporosis with bisphosphonates: the emerging role of intermittent therapy. Clin Ther 27:361–376
Tamami B, Fadavi A, Tamami M (2006) Polyacrylamide supported phenolate as a heterogeneous, efficient, recyclable, and selective catalyst for Aza- and thio-michael addition in aqueous media. Iran Polym J 15:799–807
da Silva FM, Jones J Jr (2001) Organic Reaction in Water. Part 3: Diastereoselectivity in Michael Additions of Thiophenol to Nitro Olefins in Aqueous Media. J. Braz. Chem. Soc. 12, no.2 São Paulo Mar./Apr
Ferruti P, Bertoglio Riolo C, Soldi T, Pesavento M, Barbucci R, Beni MC, Casolaro M (1982) Applied macroinorganics. II. protonation and heavy metal ions complex-formation behavior of three crosslinked resins of poly(amido-amine) structure. J Appl Polym Sci 27:2239–2248
Caldwell G, Neuse EW, Stephanou A (1993) Synthesis of water-soluble polyamidoamines for biomedical applications. 11. polymers possessing intrachain-type secondary amino groups suitable for side-chain attachment. J Appl Polym Sci 50:393–401
Mulla SM, Phale PS, Saraf MR (2012) AdMet, Paper No.OM 006. www.metrologyindia.org/ebooks1/OM_006.pdf
Acknowledgments
The financial assistance of the National Research Foundation (NRF) towards this research is hereby acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aderibigbe, B.A., Sadiku, E.R., Sinha Ray, S. et al. Synthesis and characterization of polyamidoamine conjugates of neridronic acid. Polym. Bull. 72, 417–439 (2015). https://doi.org/10.1007/s00289-014-1286-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-014-1286-z