Abstract
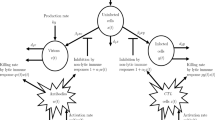
In this work, we consider an epidemic model in a two-layer network in which the dynamics of susceptible–infected–susceptible process in the physical layer coexists with that of a cyclic process of unaware–aware–unaware in the virtual layer. For such multiplex network, we shall define the basic reproduction number \(R_0^V\) in the virtual layer, which is similar to the basic reproduction number \(R_0^P\) defined in the physical layer. We show analytically that if \(R_0^P \le 1\) and \(R_0^V \le 1\), then the disease and information free equilibrium is globally stable and if \(R_0^P \le 1\) and \(R_0^V > 1\), then the disease free and information saturated equilibrium is globally stable for all initial conditions except at the origin. In the case of \(R_0^P > 1\), whether the disease dies out or not depends on the competition between how well the information is transmitted in the virtual layer and how contagious the disease is in the physical layer. In particular, it is numerically demonstrated that if the difference in \(R_0^V\) and \(R_0^P\) is greater than the product of \(R_0^P\), the deviation of \(R_0^V\) from 1 and the relative infection rate for an aware susceptible individual, then the disease dies out. Otherwise, the disease breaks out.




Similar content being viewed by others
References
Bailey NTJ (1975) The mathematical theory of infectious diseases and its applications. Griffin, London
Balcan D, Vespignani A (2011) Phase transitions in contagion processes mediated by recurrent mobility patterns. Nat Phys 7:581–586
Boccaletti S, Bianconi G, Criado R, del Genio C, Gómez-Gardeñes J, Romance M, Sendiña-Nadal I, Wang Z, Zanin M (2014) The structure and dynamics of multilayer networks. Phys Rep 544:1–122
Boguñá M, Pastor-Satorras R (2002) Epidemic spreading in correlated complex networks. Phys Rev E 66:047104
Britton T, Neal P (2010) The time to extinction for a stochastic SIS-household-epidemic model. J Math Biol 61:763
Cozzo E, Arenas A, Moreno Y (2012) Stability of boolean multilevel networks. Phys Rev E 86:036115
Dezső Z, Barabási AL (2002) Halting viruses in scale-free networks. Phys Rev E 65:055103
Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48
Funk S, Salathé M, Jansen VAA (2010) Modelling the influence of human behaviour on the spread of infectious diseases: a review. J R Soc Interface 7:1247–1256
Funka S, Gilada E, Watkinsb C, Jansena VAA (2009) The spread of awareness and its impact on epidemic outbreaks. Proc Natl Acad Sci USA 106:6872–6877
Gómez S, Diaz-Guilera A, Gómez-Gardeñes J, Pérez-Vicente CJ, Moreno Y, Arenas A (2013) Diffusion dynamics on multiplex networks. Phys Rev Lett 110:028701
Grabowski A, Kosiński RA (2004) Epidemic spreading in a hierarchical social network. Phys Rev E 70:031908
Granell C, Gómez S, Arenas A (2013) Dynamical interplay between awareness and epidemic spreading in multiplex networks. Phys Rev Lett 111:128701
Granell C, Gómez S, Arenas A (2014) Competing spreading processes on multiplex networks: awareness and epidemics. Phys Rev E 90:012808
Gross T, D’Lima CJD, Blasius B (2006) Epidemic dynamics on an adaptive network. Phys Rev Lett 96:208701
Guo Q, Jiang X, Lei Y, Li M, Ma Y, Zheng Z (2015) Two-stage effects of awareness cascade on epidemic spreading in multiplex networks. Phys Rev E 91:012822
Juang J, Liang YH (2015a) Analysis of a general SIS model with infective vectors on the complex networks. Physica A 437:382–395
Juang J, Liang YH (2015b) The impact of vaccine success and awareness on epidemic dynamics. Chaos 26:113105
Keeling MJ, Eames KTD (2005) Networks and epidemic models. J R Soc Interface 2:295–307
Korobeinikov A, Wake GC (2002) Lyapunov functions and global stability for SIR, SIRS, and SIS epidemiological models. Appl Math Lett 15:955–960
Lajmanovich A, Yorke JA (1976) A deterministic model for gonorrhea in a nonhomogeneous population. Math Biosci 28:221–236
Liang YH, Juang J (2015) The impact of vaccine failure rate on epidemic dynamics in responsive networks. Chaos 25:043116
May RM, Lloyd AL (2001) Infection dynamics on scale-free networks. Phys Rev E 64:066112
Meloni S, Perra N, Arenas A, Gómez S, Moreno Y, Vespignani A (2011) Modeling human mobility responses to the large-scale spreading of infectious diseases. Sci Rep 1:62
Moreno Y, Pastor-Satorras R, Vespignani A (2002) Epidemic outbreaks in complex heterogeneous networks. Eur Phys J B 26:521–529
Mucha PJ, Richardson T, Macon K, Porter MA, Onnela JP (2010) Community structure in time-dependent, multiscale, and multiplex networks. Science 328:876–878
Newman MEJ (2002) Spread of epidemic disease on networks. Phys Rev E 66:016128
Pastor-Satorras R, Vespignani A (2001) Epidemic dynamics and endemic states in complex networks. Phys Rev E 63:066117
Szell M, Lambiotte R, Thurner S (2010) Multirelational organization of large-scale social networks in an online world. Proc Natl Acad Sci USA 107(31):13636–13641
Wang Z, Wang L, Perc M (2014a) Degree mixing in multilayer networks impedes the evolution of cooperation. Phys Rev E 89:052813
Wang Z, Zhang H, Wang Z (2014b) Multiple effects of self-protection on the spreading of epidemics. Chaos Solitons Fractals 61:1–7
Wu Q, Fu X, Small M, Xu XJ (2012) The impact of awareness on epidemic spreading in networks. Chaos 22:013101
Wu Q, Zhang H, Zeng G (2014) Responsive immunization and intervention for infectious diseases in social networks. Chaos 24:023108
Acknowledgements
Funding was provided by Ministry of Science and Technology, Taiwan (Grant No. 105-2115-M-009-002-MY2).
Author information
Authors and Affiliations
Corresponding author
Appendix: A The derivation of model (1)
Appendix: A The derivation of model (1)
In this section, we derive the formulation of model (1). The discrete-time disease transmission model in Granell et al. (2013), based on the MMCA reads as follows.
where
Assuming the absence of dynamical correlation, \(\gamma _i(t)\) is the transition probability at time t for individual i who is not being informed by any neighbors. Moreover, \(q_i^A(t)\) (resp., \(q_i^U(t)\)) is probability at time t of an aware (resp., unaware) individual i not being infected by any neighbors. The second term \(r_i^{US}\{[1-\gamma _i(t)][1-q_i^A(t)] + \gamma _i(t)[1-q_i^U(t)]\}\) on the first equation of (17) represents the probability of an unaware and susceptible individual becoming infected. We denote this term by \(\text {Prob}(US\rightarrow AI)\). Similar notations are introduced in other terms in (17).
To derive the continuous version of disease transmission model (17), we first shorten the time interval for the iterations from \([t, t+1)\) to \([t, t+h)\) and then let h tend to 0. In the time interval \([t, t+h)\), we assume that all the parameters \(\lambda , \delta , \beta ^U, \beta ^A\) and \(\mu \) are replaced by \(\lambda h, \delta h, \beta ^U h, \beta ^A h\) and \(\mu h\), which means that the probabilities of those quantities depend linearly on the length of exposure. We shall write the resulting model as follows.
Moreover, for h sufficiently small, we have that
Similarly,
Using (19)–(23) and letting h tend to 0, we get
Using the assumption that \(p_i^{AI}+q_i^{AS}+r_i^{US}=1\), we arrive at Eqs. (1a) and (1b).
Rights and permissions
About this article
Cite this article
Huang, YJ., Juang, J., Liang, YH. et al. Global stability for epidemic models on multiplex networks. J. Math. Biol. 76, 1339–1356 (2018). https://doi.org/10.1007/s00285-017-1179-5
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-017-1179-5