Abstract
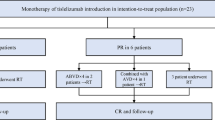
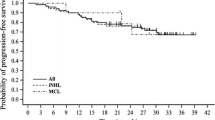
The optimal first-line treatment for advanced low-grade non-Hodgkin lymphomas (LG-NHL) is still highly debated. Recently, the StiL and the BRIGHT trials showed that the combination of rituximab and bendamustine (R-B) is non-inferior to rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) with a better toxicity profile. Utilizing a retrospective analysis, we compared the efficacy and safety of both regimens in clinical practice. From November 1995 to January 2014, 263 LG-NHL patients treated with either R-B or R-CHOP were retrospectively assessed in seven European cancer centers. Ninety patients were treated with R-B and 173 with R-CHOP. Overall response rate was 94 and 92 % for the R-B and the R-CHOP group, respectively. The percentage of complete response was similar for both groups (63 vs. 66 % with R-B and R-CHOP, respectively; p = 0.8). R-B was better tolerated and less toxic than R-CHOP. The median follow-up was 6.8 and 5.9 years for the R-CHOP and the R-B group, respectively. Overall, no difference in progression-free survival (PFS) (108 vs. 110 months; p = 0.1) was observed in the R-B group compared to the R-CHOP cohort. Nevertheless, R-B significantly prolonged PFS in FL patients (152 and 132 months in the R-B and R-CHOP group, respectively; p = 0.05). However, this result was not verified in multivariate analysis probably due to the limits of the present study. We confirm that the R-B regimen administered in patients with LG-NHL is an effective and less toxic therapeutic option than R-CHOP in clinical practice.


Similar content being viewed by others
References
Harris NL, Jaffe ES, Diebold J et al (1999) The World Health Organization classification of neoplastic diseases of the hematopoietic and lymphoid tissues. Report of the Clinical Advisory Committee meeting, Airlie House, Virginia, November, 1997. Ann Oncol 10:1419–1432
Press OW (2013) Selection of first-line therapy for advanced follicular lymphoma. J Clin Oncol 31:1496–1498
Czuczman MS, Grillo-López AJ, White CA et al (1999) Treatment of patients with low-grade B-cell lymphoma with the combination of chimeric anti-CD20 monoclonal antibody and CHOP chemotherapy. J Clin Oncol 17:268–276
Marcus R, Imrie K, Belch A et al (2005) CVP chemotherapy plus rituximab compared with CVP as first-line treatment for advanced follicular lymphoma. Blood 105:1417–1423
Federico M, Luminari S, Dondi A et al (2013) R-CVP versus R-CHOP versus R-FM for the initial treatment of patients with advanced-stage follicular lymphoma: results of the FOLL05 trial conducted by the Fondazione Italiana Linfomi. J Clin Oncol 31:1506–1513
Kath R, Blumenstengel K, Fricke HJ et al (2001) Bendamustine monotherapy in advanced and refractory chronic lymphocytic leukemia. J Cancer Res Clin Oncol 127:48–54
Ruffert K, Jahn H, Syrbe G et al (1989) Bendamustine as an alternative approach to treat malignant non-Hodgkin’s lymphoma. Z Klin Med 8:671–674
Herold M, Keinert K, Anger G (1992) Risk-adapted combined radiotherapy and chemotherapy for Hodgkin’s disease. Onkologie 15:501–505
Leoni LM, Bailey B, Reifert J et al (2008) Bendamustine (Treanda) displays a distinct pattern of cytotoxicity and unique mechanistic features compared with other alkylating agents. Clin Cancer Res 14:309–317
Rummel MJ, Niederle N, Maschmeyer G et al (2013) Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: an open-label, multicentre, randomised, phase 3 non-inferiority trial. Lancet 381:1203–1210
Flinn IW, van der Jagt R, Kahl BS et al (2014) Randomized trial of bendamustine-rituximab or R-CHOP/R-CVP in first-line treatment of indolent NHL or MCL: the BRIGHT study. Blood 123:2944–2952
Burke JM, Van der Jagt RH, Kahl BS, et al. (2012) Differences in quality of life between bendamustine plus rituximab compared with standard first- line treatments in patients with previously untreated advanced indolent non-Hodgkin’s lymphoma or mantle cell lymphoma. Blood a155
Morschhauser F, Seymour JF, Feugier P et al (2011) Impact of induction chemotherapy regimen on response, safety and outcome in the PRIMA study. Ann Oncol 22(suppl 4):89
Harris NL, Jaffe ES, Stein H et al (1994) A revised European-American classification of lymphoid neoplasms: a proposal from the International Lymphoma Study Group. Blood 84:1361–1392
Swerdlow SH, Campo E, Harris NL, Swerdlow SH, Campo E, Harris NL (2008) WHO classification of tumours of haematopoietic and lymphoid tissues, 4th edn. IARC Press, Lyon
Cheson BD, Horning SJ, Coiffier B et al (1999) Report of an international workshop to standardize response criteria for non-Hodgkin’s lymphomas. NCI Sponsored International Working Group. J Clin Oncol 17:1244
Cheson BD, Pfistner B, Juweid M et al (2007) Revised response criteria for malignant lymphoma. J Clin Oncol 25:579–586
Solal-Celigny P, Roy P, Colombat P et al (2004) Follicular Lymphoma International Prognostic Index. Blood 104:1258–1265
Singal AG, Higgins PD, Waljee AK (2014) A primer on effectiveness and efficacy trials. Clin Transl Gastroenterol 5:e45
Alamdari HS, Pinter-Brown L, Cassarino DS, Chiu MW (2010) Severe cutaneous interface drug eruption associated with bendamustine. Dermatol Online J 16:1
Fallon MJ, Heck JN (2015) Fatal Stevens-Johnson syndrome/toxic epidermal necrolysis induced by allopurinol-rituximab-bendamustinetherapy. J Oncol Pharm Pract 21:388–392
Hiddemann W, Kneba M, Dreyling M et al (2005) Frontline therapy with rituximab added to the combination of cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) significantly improves the outcome for patients with advanced-stage follicular lymphoma compared with therapy with CHOP alone: results of a prospective randomized study of the German Low-Grade Lymphoma Study Group. Blood 106:3725–3732
Acknowledgments
This article did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This analysis was approved by the local Ethical Committee (Prot. 0042654-BZ). Due to the retrospective and anonymous data collection, informed consent was not necessary.
Conflict of interest
The authors have no conflicts of interest. PM, NS, IW, FZ, RZ, AV, EM, PG, VP, and SC have no financial disclosure. SF and MM have received honoraria from Mundipharma; WW has received honoraria and research funding from Mundipharma and Roche.
Rights and permissions
About this article
Cite this article
Mondello, P., Steiner, N., Willenbacher, W. et al. Bendamustine plus rituximab versus R-CHOP as first-line treatment for patients with indolent non-Hodgkin’s lymphoma: evidence from a multicenter, retrospective study. Ann Hematol 95, 1107–1114 (2016). https://doi.org/10.1007/s00277-016-2668-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-016-2668-0