Abstract
Background
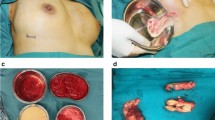
Polyacrylamide hydrogel (PAAG), once used as an injection for breast augmentation, has been banned in the medical field for cosmetic purposes for more than 10 years in China. But a large number of breasts have characteristic deformities due to the gel’s feature of erosion. Our aim is to explore a retrospective study on PAAG’s long-term effects on women, ensuing breast deformity and the strategy for breast plasticity after removing the gel in our center from 2007 to 2014.
Methods
From 2007 to 2014, 200 patients, whose breasts were injected with PAAG, underwent the operation to remove the injectable material. Complications were summarized. Ultrasound or MRI was performed before the operation to disclose the general distribution of the gel and the muscle and gland infiltration. According to the gel distribution, muscle and gland infiltration, infection, gel residue, and other factors, the patients were treated, respectively, with or without prosthesis implantation surgery after the removal of the gel. According to the decision about whether or when to undergo prosthesis implantation, the patients were classified into three types: group I—prosthesis implantation at the first stage, group II—prosthesis implantation at the second stage, and group III—only removing the material without prosthesis implantation. The scores of the BREAST-Q program were used to evaluate the preoperative and postoperative differences.
Results
Seventy-seven patients underwent prosthesis implantation at the first stage and 61 patients were operated on by placing the prosthesis at the second stage. A total of 62 patients only underwent the PAAG removal operation. By BREAST-Q evaluation, changes are summarized in Tables 1, 2, and 3 between mean preoperative scores and mean postoperative scores through categories of satisfaction with appearance of breasts, psychosocial wellbeing, sexual wellbeing, and physical wellbeing, in which all categories were presented with statistical significance (p < 0.001).
Conclusions
Timely removal is critical for women who have received the PAAG removal operation. However, the surgery may destroy the shape of the breast. It is recommended that preoperative communication and local tissue condition are guidelines for surgeons to choose conservative or aggressive surgery. A balance must be maintained between removing the gel as much as possible and retaining soft tissue to reshape breasts.
Level of Evidence IV
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.








Similar content being viewed by others
References
Okubo M, Hyakusoku H, Kanno K, Fumiiri M (1992) Complications after injection mammaplasty. Aesthetic Plast Surg 16:181–187
Christensen LH, Breiting VB, Aasted A et al (2003) Long-term effects of polyacrylamide hydrogel on human breast tissue. Plast Reconstr Surg 111:1883–1890
Cheng NX, Wang YL, Wang JH et al (2002) Complications of breast augmentation with injected hydrophilic polyacrylamide gel. Aesthetic Plast Surg 26:375–382
Amin SP, Marmur ES, Goldberg DJ (2004) Complications from injectable polyacrylamide gel, a new nonbiodegradable soft tissue filler. Dermatol Surg 30:1507–1509
Yue Y, Luan J, Qiao Q et al (2007) Retrospective analysis of complications of breast augmentation with injected polyacrylamide hydrophilic gel in 90 cases (in Chinese). Zhonghua Zheng Xing Wai Ke Za Zhi 23:221–223
Ono S, Ogawa R, Hyakusoku H (2010) Complications after polyacrylamide hydrogel injection for soft-tissue augmentation. Plast Reconstr Surg 126:1349–1357
Lin J, Qian YL, Yang Q et al (2007) Clinical analysis of complications of polyacrylamide hydrogel injection for augmentation mammoplasty in 118 cases (in Chinese). Zhonghua Zheng Xing Wai Ke Za Zhi 23:101–102
Cheng NX, Liu LG, Hui L et al (2009) Breast cancer following augmentation mammaplasty with polyacrylamide hydrogel (PAAG) injection. Aesthetic plastic surgery 33:563–569
Lee CJ, Kim SG, Kim L, Choi MS, Lee SI (2004) Unfavorable findings from hydrogel. Plast Reconstr Surg 114:1967–1968
Christensen LC, Breiting V, Janssen M et al (2005) Adverse reaction to injectable soft tissue permanent fillers. Aesthetic Plast Surg 29:34
Wolters M, Lampe H (2009) Prospective multicenter study for evaluation of safety, efficacy, and esthetic results of cross-linked polyacrylamide hydrogel in 81 patients. Dermatol Surg 35:338–343
Shen Hui, Lv Ying, Jing-hong Xu et al (2012) Complications after polyacrylamide hydrogel injection for facial soft-tissue augmentation in China: twenty-four cases and their surgical management. PRS 130(2):340–348
Breiting V, Aasted A, Jørgensen A, Opitz P, Rosetzsky A (2004) A study on patients treated with polyacrylamide hydrogel injection for facial corrections. Aesthetic Plast Surg 28:45–53
Patlazhan G, Unukovych D, Pshenisnov K (2013) Breast reconstruction and treatment algorithm for patients with complications after polyacrylamide gel injections: a 10-year experience. Aesthetic Plast Surg 37(2):312–320
Unukovych D, Khrapach V, Wickman M et al (2012) Polyacrylamide gel injections for breast augmentation: management of complications in 106 patients, a multicenter study. World J Surg 36(4):695–701
Sheng-Kang L, Gunang-Ping C, Zhong-Shen S et al (2011) Our strategy in complication management of augmentation mammaplasty with polyacrylamide hydrogel injection in 235 patients. J Plast Reconstr Aesthet Surg 64(6):731–737
Qiao Q, Wang X, Sun J et al (2005) Management for postoperative complications of breast augmentation by injected polyacrylamide hydrogel. Aesthetic Plast Surg 29(3):156–161
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None declared.
Rights and permissions
About this article
Cite this article
Chen, B., Song, H. Management of Breast Deformity After Removal of Injectable Polyacrylamide Hydrogel: Retrospective Study of 200 Cases for 7 Years. Aesth Plast Surg 40, 482–491 (2016). https://doi.org/10.1007/s00266-016-0646-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-016-0646-5