Abstract
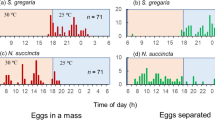
This laboratory study examined the stimuli involved in controlling egg hatching in the desert locust Schistocerca gregaria. We found that eggs that were kept in their egg pods hatched in synchrony, whereas those that were kept individually hatched over an extended period of time and took significantly longer to hatch. Synchronous hatching was also observed in pairs of eggs that were kept in contact with each other in the same plastic wells, but not in those that were kept individually in separate wells. This demonstrates that the eggs needed to be placed in contact with each other to achieve synchronous hatching and suggests that a hatching individual stimulates adjacent eggs to hatch via some unknown stimulus. Additional experiments suggested that neither auditory nor pheromonal stimuli were important in this process, but rather some sort of vibratory stimulus. This inference was supported by the results of another experiment, which demonstrated that when isolated eggs were vibrated with a vortex mixer, most of them hatched shortly after the stimulation. Our results suggest the possibility that direct mechanical stimulation caused by the first egg to hatch stimulates adjacent eggs to also hatch, thus producing synchronous hatching in the entire egg pod. However, the exact nature of this stimulus remains to be determined.
Significance statement
Synchronous hatching is often observed in insects. It helps hatchlings to form aggregations and to reduce the risk of predation and cannibalism. Yet, the mechanism controlling this phenomenon has received little attention except for a few subsocial species. This study demonstrates that eggs kept in their egg pods hatched in synchrony, whereas those kept individually in sand hatched over an extended period of time and took longer to hatch. Synchronous hatching was also observed in pairs of eggs that were kept in contact with each other but not placed some distance apart from each other. We also show that artificial vibration induced hatching.










Similar content being viewed by others
References
Alcock J (2005) Animal behavior: an evolutionary approach, 8th edn. Sinauer, Sunderland
Allee WC (1938) The social life of animals. William Heinemann, London
Ashall C, Ellis PE (1962) Studies on numbers and mortality in field populations of the desert locust (Schistocerca gregaria Forskål). Anti-Locust Bulletin 38:1–59
Bernays EA (1971) Hatching in Schistocerca gregaria (Foskål) (Orthoptera, Acrididae). Acrida 1:41–60
Cocroft RB, Rodríguez RL (2005) The behavioral ecology of insect vibrational communication. Bioscience 55:323–334
Crespi BJ (1992) Cannibalism and trophic eggs in subsocial and eusocial insects. In: Elgar MA, Crespi BJ (eds) Cannibalism; ecology and evolution among diverse taxa. Oxford University Press, Oxford, pp 176–213
Driver PM (1965) ‘Clicking’ in the egg-young of nidifugous birds. Nature 206:315
Elgar MA, Crespi BJ (1992) Ecology and evolution of cannibalism. In: Elgar MA, Crespi BJ (eds) Cannibalism; ecology and evolution among diverse taxa. Oxford University Press, Oxford, pp 1–12
Ellis PE, Ashall C (1957) Field studies on diurnal behaviour, movement and aggregation in the desert locust (Schistocerca gregaria Forskål). Anti-Locust Bull 25:1–94
Griem JN, Martin KLM (2000) Wave action: the environmental trigger for hatching in the California grunion Leuresthes tenuis (Teleostei: Atherinopsidae). Mar Biol 137:177–181
Hopper KR, Crowley PH, Kielman D (1996) Density dependence, hatching synchrony, and within-cohort cannibalism in young dragonfly larvae. Ecology 77:191–200
Hosokawa T, Hironaka M, Mukai H, Inadomi K, Suzuki N, Fukatsu T (2012) Mothers never miss the moment: a fine-tuned mechanism for vertical symbiont transmission in a subsocial insect. Anim Behav 83:293–300
Hunter-Jones P (1964) Egg development in the desert locust (Schistocerca gregaria Forsk.) in relation to the availability of water. Proc R Entomol Soc A 39:25–33
Ims RA (1990) On the adaptive value of reproductive synchrony as a predator-swamping strategy. Am Nat 136:485–498
Kiritani K (1964) The effect of colony size upon the survival of larvae of the southern green stink bug, Nezara viridula. Jpn J Appl Entomol Zool 8:45–54
Lockwood JA, Story RN (1985) Photic, thermic, and sibling influences on the hatching rhythm of the southern green stink bug, Nezara viridula (L.). Environ Entomol 14:562–567
Morgan SG, Christy JH (1995) Adaptive significance of the timing of larval release by crabs. Am Nat 145:457–479
Mukai H, Hironaka M, Tojo S, Nomakuchi S (2012) Maternal vibration induces synchronous hatching in a subsocial burrower bug. Anim Behav 84:1443–1448
Mukai H, Hironaka M, Tojo S, Nomakuchi S (2014) Maternal vibration: an important cue for embryo hatching in a subsocial shield bug. PLoS One 9:e87932
Nicolai CA, Sedinger JS, Wege ML (2004) Regulation of development time and hatch synchronization in black brant (Branta bernicla nigricans). Funct Ecol 18:475–482
Nishide Y, Tanaka S, Saeki S (2015a) Adaptive difference in daily timing of hatch in two locust species, Schistocerca gregaria and Locusta migratoria: the effects of thermocycles and phase polyphenism. J Insect Physiol 72:79–87
Nishide Y, Tanaka S, Saeki S (2015b) Egg hatching of two locusts, Schistocerca gregaria and Locusta migratoria, in response to light and temperature cycles. J Insect Physiol 76:24–29
Ohba S (2002) Synchronized mechanism and its meaning in the egg hatching of the giant water bug, Lethocerus deyrolli (Heteroptera: Belostomatidae). Japanese Journal of Entomology (New Series) 5: 157–164 (in Japanese, with English Summary)
Padgham DE (1981) Hatching rhythms in the desert locust, Schistocerca gregaria. Physiol Entomol 6:191–198
Papillon M (1960) Etude préliminaire de la répercussion du groupement des parents sur les larves nouveau-nées de Schistocerca gregaria Forsk. Bulletin Biologique de la France et de la Belgique 94:203–263
Saunders DS (2002) Insect clocks, 3rd edn. Elsevier, Amsterdam
Savage JM (2002) The amphibians and reptiles of Costa Rica. University of Chicago Press, Chicago
Shulov A, Pener MP (1963) Studies on the development of eggs of the desert locust (Schistocerca gregaria Forskål) and its interruption under particular conditions of humidity. Anti-Locust Bull 41:1–59
Skov MW, Harnoll RG, Ruwa RK, Shunula JP, Vannini M, Cannicci S (2005) Marching to a different drummer: crabs synchronize reproduction to a 14-month lunar-tidal cycle. Ecology 86:1164–1171
Smith AR, Nowak A, Wagner P, Yates R, Janci E, Bernales R, Dietz T, Earhart A, Fogle A, Fullerton N, Gromer K, Kliver B, Larson W, Ludwikowski J, Martini T, McGrath J, Polino A, Schumacher M, Weick S, Casto JM, Whitman DW (2013) Daily temperature cycle induces daily hatching rhythm in eastern lubber grasshoppers, Romalea microptera. J Orthoptera Res 22:51–55
Tanaka S, Maeno K (2008) Maternal effects on progeny size and color in the desert locust, Schistocerca gregaria: examination of a current view. J Insect Physiol 54:612–618
Uvarov BP (1966) Grasshoppers and locusts, vol 1. Cambridge University Press, Cambridge
Venter IG, Potgieter WJ (1967) Systematic discontinuities between eggs and hoppers from individual egg pods of the brown locust. South Afr J Agric Sci 10:293–297
Vergne AL, Mathevon N (2008) Crocodile egg sounds signal hatching time. Curr Biol 18:R513–R514
Walker BW (1952) A guide to the grunion. Calif Fish Game 38:409–420
Warkentin KM (1995) Adaptive plasticity in hatching age: a response to predation risk trade-offs. Proc Natl Acad Sci U S A 92:3507–3510
Warkentin KM (2005) How do embryos assess risk? Vibrational cues in predator-induced hatching of red-eyed treefrogs. Anim Behav 70:59–71
Warkentin KM, Caldwell MS, Siok TD, D’Amato AT, McDaniel JG (2007) Flexible information sampling in vibrational assessment of predation risk by red-eyed treefrog embryos. J Exp Biol 210:614–619
Acknowledgments
We thank Ms. Hiroko Ikeda, Ms. Noriko Totsuka, Ms. Masako Higuchi, and Mr. Shinjiro Saeki (Locust Research Laboratory, NARO) for maintaining the locust colonies. Special thanks are due to Dr. Sinzo Masaki (Professor emeritus of Hirosaki University) who reviewed the manuscript and corrected our English. The grass used in this experiment was raised by the Field Management Section of NARO, and we would like to thank Mr. Kameo Tsukada for growing the grass. Two anonymous reviewers improved the manuscript greatly.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was founded in part by a Kakenhi Grant of Japan (No. 23380038) to S.T.
Conflict of interest
The authors declare no competing interests.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Communicated by J. C. Choe
Rights and permissions
About this article
Cite this article
Nishide, Y., Tanaka, S. Desert locust, Schistocerca gregaria, eggs hatch in synchrony in a mass but not when separated. Behav Ecol Sociobiol 70, 1507–1515 (2016). https://doi.org/10.1007/s00265-016-2159-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-016-2159-2