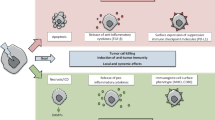
Abstract
Radiotherapy (RT) utilizes the DNA-damaging properties of ionizing radiation to control tumor growth and ultimately kill tumor cells. By modifying the tumor cell phenotype and the tumor microenvironment, it may also modulate the immune system. However, out-of-field reactions of RT mostly assume further immune activation. Here, the sequence of the applications of RT and immunotherapy is crucial, just as the dose and fractionation may be. Lower single doses may impact on tumor vascularization and immune cell infiltration in particular, while higher doses may impact on intratumoral induction and production of type I interferons. The induction of immunogenic cancer cell death seems in turn to be a common mechanism for most RT schemes. Dendritic cells (DCs) are activated by the released danger signals and by taking up tumor peptides derived from irradiated cells. DCs subsequently activate T cells, a process that has to be tightly controlled to ensure tolerance. Inhibitory pathways known as immune checkpoints exist for this purpose and are exploited by tumors to inhibit immune responses. Cytotoxic T lymphocyte antigen 4 (CTLA-4) and programmed cell death protein 1 (PD-1) on T cells are two major checkpoints. The biological concepts behind the findings that RT in combination with anti-CTLA-4 and/or anti-PD-L1 blockade stimulates CD8+ T cell-mediated anti-tumor immunity are reviewed in detail. On this basis, we suggest clinically significant combinations and sequences of RT and immune checkpoint inhibition. We conclude that RT and immune therapies complement one another.


Similar content being viewed by others
Abbreviations
- Ab:
-
Antibody
- Ag:
-
Antigen
- APC:
-
Antigen-presenting cell
- ATP:
-
Adenosine triphosphate
- CpG:
-
Cytosine–guanine-rich motifs
- CRT:
-
Calreticulin
- CT:
-
Chemotherapy
- CTLA-4:
-
Cytotoxic T lymphocyte antigen 4
- DAMP:
-
Damage-associated molecular pattern
- DC:
-
Dendritic cell
- DNA:
-
Deoxyribonucleic acid
- ER:
-
Endoplasmic reticulum
- FDA:
-
US Food and Drug Administration
- Flt3-L:
-
Fms-related tyrosine kinase 3 ligand
- GM-CSF:
-
Granulocyte-macrophage colony-stimulating factor
- HMGB1:
-
High-mobility group box 1
- Hsp70:
-
Heat shock protein 70
- ICAM-1:
-
Intercellular adhesion molecule-1
- IFN:
-
Interferon
- IL:
-
Interleukin
- iNOS:
-
Inducible nitric oxide synthase
- MDSC:
-
Myeloid-derived suppressor cell
- MHC:
-
Major histocompatibility complex
- NSCLC:
-
Non-small cell lung cancer
- PD-1:
-
Programmed cell death protein 1
- ROS:
-
Reactive oxygen species
- RT:
-
Radiotherapy
- RT5:
-
Rat insulin promoter (RIP)1-Tag5 tumor mouse model
- TNF:
-
Tumor necrosis factor
- X-ray:
-
Ionizing radiation
- zVAD-fmk:
-
Z-Val-Ala-DL-Asp-FMK
References
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. doi:10.1016/j.cell.2011.02.013
Mittal D, Gubin MM, Schreiber RD, Smyth MJ (2014) New insights into cancer immunoediting and its three component phases–elimination, equilibrium and escape. Curr Opin Immunol 27:16–25. doi:10.1016/j.coi.2014.01.004
Dunn GP, Old LJ, Schreiber RD (2004) The immunobiology of cancer immunosurveillance and immunoediting. Immunity 21:137–148. doi:10.1016/j.immuni.2004.07.017
Kepp O, Senovilla L, Vitale I et al (2014) Consensus guidelines for the detection of immunogenic cell death. Oncoimmunology 3:e955691. doi:10.4161/21624011.2014.955691
Apetoh L, Ghiringhelli F, Tesniere A et al (2007) Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med 13:1050–1059. doi:10.1038/nm1622
Vacchelli E, Aranda F, Eggermont A, Galon J, Sautes-Fridman C, Cremer I, Zitvogel L, Kroemer G, Galluzzi L (2014) Trial watch: chemotherapy with immunogenic cell death inducers. Oncoimmunology 3:e27878. doi:10.4161/onci.27878
Kroemer G, El-Deiry WS, Golstein P et al (2005) Classification of cell death: recommendations of the nomenclature committee on cell death. Cell Death Differ 12(Suppl 2):1463–1467. doi:10.1038/sj.cdd.4401724
Salomaa SI, Wright EG, Hildebrandt G, Kadhim MA, Little MP, Prise KM, Belyakov OV (2010) Editorial. Non-DNA targeted effects. Mutat Res 687:1–2. doi:10.1016/j.mrfmmm.2010.01.018
Hodge JW, Ardiani A, Farsaci B, Kwilas AR, Gameiro SR (2012) The tipping point for combination therapy: cancer vaccines with radiation, chemotherapy, or targeted small molecule inhibitors. Semin Oncol 39:323–339. doi:10.1053/j.seminoncol.2012.02.006
Frey B, Rubner Y, Kulzer L, Werthmoller N, Weiss EM, Fietkau R, Gaipl US (2014) Antitumor immune responses induced by ionizing irradiation and further immune stimulation. Cancer Immunol Immunother 63:29–36. doi:10.1007/s00262-013-1474-y
Gaipl US, Multhoff G, Scheithauer H, Lauber K, Hehlgans S, Frey B, Rodel F (2014) Kill and spread the word: stimulation of antitumor immune responses in the context of radiotherapy. Immunotherapy 6:597–610. doi:10.2217/imt.14.38
Demaria S, Ng B, Devitt ML, Babb JS, Kawashima N, Liebes L, Formenti SC (2004) Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int J Radiat Oncol Biol Phys 58:862–870. doi:10.1016/j.ijrobp.2003.09.012
Werthmöller N, Frey B, Wunderlich R, Fietkau R, Gaipl US (2015) Modulation of radiochemoimmunotherapy-induced B16 melanoma cell death by the pan-caspase inhibitor zVAD-fmk induces anti-tumor immunity in a HMGB1-, nucleotide- and T-cell-dependent manner. Cell Death Dis 6:e1761. doi:10.1038/cddis.2015.129
Vandenabeele P, Galluzzi L, Vanden Berghe T, Kroemer G (2010) Molecular mechanisms of necroptosis: an ordered cellular explosion. Nat Rev Mol Cell Biol 11:700–714. doi:10.1038/nrm2970
Klug F, Prakash H, Huber PE et al (2013) Low-dose irradiation programs macrophage differentiation to an iNOS(+)/M1 phenotype that orchestrates effective T cell immunotherapy. Cancer Cell 24:589–602. doi:10.1016/j.ccr.2013.09.014
Garbi N, Arnold B, Gordon S, Hammerling GJ, Ganss R (2004) CpG motifs as proinflammatory factors render autochthonous tumors permissive for infiltration and destruction. J Immunol 172:5861–5869. doi:10.4049/jimmunol.172.10.5861
Frey B, Hehlgans S, Rödel F, Gaipl US (2015) Modulation of inflammation by low and high doses of ionizing radiation: implications for benign and malign diseases. Cancer Lett 368:230–237. doi:10.1016/j.canlet.2015.04.010
Witek M, Blomain ES, Magee MS, Xiang B, Waldman SA, Snook AE (2014) Tumor radiation therapy creates therapeutic vaccine responses to the colorectal cancer antigen GUCY2C. Int J Radiat Oncol Biol Phys 88:1188–1195. doi:10.1016/j.ijrobp.2013.12.043
Kulzer L, Rubner Y, Deloch L, Allgauer A, Frey B, Fietkau R, Dorrie J, Schaft N, Gaipl US (2014) Norm- and hypo-fractionated radiotherapy is capable of activating human dendritic cells. J Immunotoxicol 11:328–336. doi:10.3109/1547691x.2014.880533
Zeng J, See AP, Phallen J et al (2013) Anti-PD-1 blockade and stereotactic radiation produce long-term survival in mice with intracranial gliomas. Int J Radiat Oncol Biol Phys 86:343–349. doi:10.1016/j.ijrobp.2012.12.025
Dewan MZ, Galloway AE, Kawashima N, Dewyngaert JK, Babb JS, Formenti SC, Demaria S (2009) Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti-CTLA-4 antibody. Clin Cancer Res 15:5379–5388. doi:10.1158/1078-0432.ccr-09-0265
Ganss R, Ryschich E, Klar E, Arnold B, Hammerling GJ (2002) Combination of T-cell therapy and trigger of inflammation induces remodeling of the vasculature and tumor eradication. Cancer Res 62:1462–1470
Burnette BC, Liang H, Lee Y, Chlewicki L, Khodarev NN, Weichselbaum RR, Fu YX, Auh SL (2011) The efficacy of radiotherapy relies upon induction of type I interferon-dependent innate and adaptive immunity. Cancer Res 71:2488–2496. doi:10.1158/0008-5472.can-10-2820
Rubner Y, Wunderlich R, Ruhle PF et al (2012) How does ionizing irradiation contribute to the induction of anti-tumor immunity? Front Oncol 2:75. doi:10.3389/fonc.2012.00075
Wunderlich R, Ernst A, Rodel F, Fietkau R, Ott O, Lauber K, Frey B, Gaipl US (2015) Low and moderate doses of ionizing radiation up to 2 Gy modulate transmigration and chemotaxis of activated macrophages, provoke an anti-inflammatory cytokine milieu, but do not impact upon viability and phagocytic function. Clin Exp Immunol 179:50–61. doi:10.1111/cei.12344
Grivennikov SI, Greten FR, Karin M (2010) Immunity, inflammation, and cancer. Cell 140:883–899. doi:10.1016/j.cell.2010.01.025
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. doi:10.1038/nrc3239
Couzin-Frankel J (2013) Breakthrough of the year 2013. Cancer Immunother Sci 342:1432–1433. doi:10.1126/science.342.6165.1432
Eggermont AM, Maio M, Robert C (2015) Immune checkpoint inhibitors in melanoma provide the cornerstones for curative therapies. Semin Oncol 42:429–435. doi:10.1053/j.seminoncol.2015.02.010
Pandha H, Pawelec G (2015) Immune checkpoint targeting as anti-cancer immunotherapy: promises, questions, challenges and the need for predictive biomarkers at ASCO 2015. Cancer Immunol Immunother 64:1071–1074. doi:10.1007/s00262-015-1748-7
Demaria S, Kawashima N, Yang AM, Devitt ML, Babb JS, Allison JP, Formenti SC (2005) Immune-mediated inhibition of metastases after treatment with local radiation and CTLA-4 blockade in a mouse model of breast cancer. Clin Cancer Res 11:728–734
Verbrugge I, Hagekyriakou J, Sharp LL et al (2012) Radiotherapy increases the permissiveness of established mammary tumors to rejection by immunomodulatory antibodies. Cancer Res 72:3163–3174. doi:10.1158/0008-5472.can-12-0210
Twyman-Saint Victor C, Rech AJ, Maity A et al (2015) Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature 520:373–377. doi:10.1038/nature14292
Kim JE, Patel MA, Mangraviti A et al (2015) 143 the combination of anti-TIM-3 and anti-PD-1 checkpoint inhibitors with focused radiation resulted in a synergistic antitumor immune response in a preclinical glioma model. Neurosurgery 62(Suppl 1):212. doi:10.1227/01.neu.0000467105.60300.04
Frey B, Rubner Y, Wunderlich R, Weiss EM, Pockley AG, Fietkau R, Gaipl US (2012) Induction of abscopal anti-tumor immunity and immunogenic tumor cell death by ionizing irradiation—implications for cancer therapies. Curr Med Chem 19:1751–1764
Deng L, Liang H, Burnette B, Beckett M, Darga T, Weichselbaum RR, Fu YX (2014) Irradiation and anti-PD-L1 treatment synergistically promote antitumor immunity in mice. J Clin Invest 124:687–695. doi:10.1172/jci67313
Dovedi SJ, Adlard AL, Lipowska-Bhalla G et al (2014) Acquired resistance to fractionated radiotherapy can be overcome by concurrent PD-L1 blockade. Cancer Res 74:5458–5468. doi:10.1158/0008-5472.can-14-1258
Barker CA, Postow MA (2014) Combinations of radiation therapy and immunotherapy for melanoma: a review of clinical outcomes. Int J Radiat Oncol Biol Phys 88:986–997. doi:10.1016/j.ijrobp.2013.08.035
Golden EB, Chhabra A, Chachoua A et al (2015) Local radiotherapy and granulocyte-macrophage colony-stimulating factor to generate abscopal responses in patients with metastatic solid tumours: a proof-of-principle trial. Lancet Oncol 16:795–803. doi:10.1016/s1470-2045(15)00054-6
Alexandrov LB, Nik-Zainal S, Wedge DC et al (2013) Signatures of mutational processes in human cancer. Nature 500:415–421. doi:10.1038/nature12477
Page DB, Postow MA, Callahan MK, Allison JP, Wolchok JD (2014) Immune modulation in cancer with antibodies. Annu Rev Med 65:185–202. doi:10.1146/annurev-med-092012-112807
Philips GK, Atkins M (2015) Therapeutic uses of anti-PD-1 and anti-PD-L1 antibodies. Int Immunol 27:39–46. doi:10.1093/intimm/dxu095
Kline J, Bishop MR (2015) Update on checkpoint blockade therapy for lymphoma. J Immunother Cancer 3:33. doi:10.1186/s40425-015-0079-8
Specht HM, Ahrens N, Blankenstein C et al (2015) Heat shock protein 70 (Hsp70) peptide activated natural killer (NK) cells for the treatment of patients with non-small cell lung cancer (NSCLC) after radiochemotherapy (RCTx)—from preclinical studies to a clinical phase ii trial. Front Immunol 6:162. doi:10.3389/fimmu.2015.00162
Barker HE, Paget JT, Khan AA, Harrington KJ (2015) The tumour microenvironment after radiotherapy: mechanisms of resistance and recurrence. Nat Rev Cancer 15:409–425. doi:10.1038/nrc3958
Ngiow SF, McArthur GA, Smyth MJ (2015) Radiotherapy complements immune checkpoint blockade. Cancer Cell 27:437–438. doi:10.1016/j.ccell.2015.03.015
Frey B, Gaipl US (2015) Radio-immunotherapy: the focused beam expands. Lancet Oncol 16:742–743. doi:10.1016/s1470-2045(15)00055-8
Acknowledgments
This work was partially funded by the German Federal Ministry of Education and Research (BMBF; m4 Cluster, 16EX1021R and GREWIS, 02NUK017G) and the European Commission (DoReMi, European Atomic Energy Community’s Seventh Framework Programme (FP7/2007-2011) under Grant Agreement No. 249689).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no competing interests.
Additional information
This paper is a Focussed Research Review based on a presentation given at the Fourth International Conference on Cancer Immunotherapy and Immunomonitoring (CITIM 2015), held in Ljubljana, Slovenia, 27th–30th April 2015. It is part of a series of Focussed Research Reviews and meeting report in Cancer Immunology, Immunotherapy.
Rights and permissions
About this article
Cite this article
Derer, A., Frey, B., Fietkau, R. et al. Immune-modulating properties of ionizing radiation: rationale for the treatment of cancer by combination radiotherapy and immune checkpoint inhibitors. Cancer Immunol Immunother 65, 779–786 (2016). https://doi.org/10.1007/s00262-015-1771-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-015-1771-8