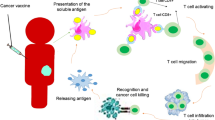
Abstract
Cryoablation is a low-invasive surgical procedure for management of malignant tumors. Tissue destruction is obtained by repeated deep freezing and thawing and results in coagulative necrosis and in apoptosis. This procedure induces the release of tumor-associated antigens and proinflammatory factors into the microenvironment. Local administration of immature dendritic cells (DCs) potentiates the immune response induced by cryoablation. To further augment the induction of long-lasting antitumor immunity, we investigated the clinical value of combining cryoimmunotherapy consisting of cryoablation and inoculation of immature DCs with administration of the immune adjuvant, CpG oligodeoxynucleotides. Injection of the murine Lewis lung carcinoma, D122-luc-5.5 that expresses the luciferase gene, results in spontaneous metastases, which can be easily monitored in vivo. The local tumor was treated by the combined treatment. The clinical outcome was assessed by monitoring tumor growth, metastasis in distant organs, overall survival, and protection from tumor recurrence. The nature of the induced T cell responses was analyzed. Combined cryoimmunotherapy results in reduced tumor growth, low metastasis and significantly prolonged survival. Moreover, this treatment induces antitumor memory that protected mice from rechallenge. The underlying suggested mechanisms are the generation of tumor-specific type 1 T cell responses, subsequent induction of cytotoxic T lymphocytes, and generation of systemic memory. Our data highlight the combined cryoimmunotherapy as a novel antitumor vaccine with promising preclinical results. Adjustment of this technique into practice will provide the therapeutic benefits of both, ablation of the primary tumor and induction of robust antitumor and antimetastatic immunity.






Similar content being viewed by others
References
Tsivian M, Polascik TJ (2010) Focal cryotherapy for prostate cancer. Curr Urol Rep 11:147–151
Rioja J, Tzortzis V, Mamoulakis C, Laguna MP (2010) Cryotherapy for renal tumors: current status and contemporary developments. Actas Urol Esp (English Edition) 34:309–317
Ng KM, Chua TC, Saxena A, Zhao J, Chu F et al (2012) Two decades of experience with hepatic cryotherapy for advanced colorectal metastases. Ann Surg Oncol 19:1276–1283
Greenwald BD, Dumot JA, Abrams JA, Lightdale CJ, David DS et al (2010) Endoscopic spray cryotherapy for esophageal cancer: safety and efficacy. Gastrointest Endosc 71:686–693
Lee S-H, Choi W-J, Sung S-W, Kim Y-K, Kim C-H et al (2011) Endoscopic cryotherapy of lung and bronchial tumors: a systematic review. Korean J Intern Med 26:137–144
Nishida H, Yamamoto N, Tanzawa Y, Tsuchiya H (2011) Cryoimmunology for malignant bone and soft-tissue tumors. Int J Clin Oncol 16:109–117
Sabel MS (2009) Cryo-immunology: a review of the literature and proposed mechanisms for stimulatory versus suppressive immune responses. Cryobiology 58:1–11
Sidana A, Chowdhury WH, Fuchs EJ, Rodriguez R (2010) Cryoimmunotherapy in urologic oncology. Urology 75:1009–1014
Redondo P, del Olmo J, López-Diaz de Cerio A, Inoges S, Marquina M et al (2007) Imiquimod enhances the systemic immunity attained by local cryosurgery destruction of melanoma lesions. J Investig Dermatol 127:1673–1680
Udagawa M, Kudo-Saito C, Hasegawa G, Yano K, Yamamoto A et al (2006) Enhancement of immunologic tumor regression by intratumoral administration of dendritic cells in combination with cryoablative tumor pretreatment and Bacillus Calmette-Guerin cell wall skeleton stimulation. Clin Cancer Res 12:7465–7475
Hoffmann NE, Coad JE, Huot CS, Swanlund DJ, Bischof JC (2001) Investigation of the mechanism and the effect of cryoimmunology in the Copenhagen rat. Cryobiology 42:59–68
Shibata T, Suzuki K, Yamashita T, Takeichi N, Mark M, Hosokawa M, Kobayashi HAM (1998) Immunological analysis of enhanced spontaneous metastasis in WKA rats following cryosurgery. Anticancer Res 18(4A):2483–2486
Machlenkin A, Goldberger O, Tirosh B, Paz A, Bar-haim E et al (2005) Combined dendritic cell cryotherapy of tumor induces systemic antimetastatic immunity. Clin Cancer Res 11:4955–4961
Krieg AM (2006) Therapeutic potential of toll-like receptor 9 activation. Nat Rev Drug Discov 5:471–484
Vollmer J, Krieg AM (2009) Immunotherapeutic applications of CpG oligodeoxynucleotide TLR9 agonists. Adv Drug Deliv Rev 61:195–204
den Brok MHMGM, Sutmuller RPM, Nierkens S, Bennink EJ, Toonen LWJ et al (2006) Synergy between in situ cryoablation and TLR9 stimulation results in a highly effective in vivo dendritic cell vaccine. Cancer Res 66:7285–7292
Eisenbach L, Hollander N, Greenfeld L, Yakor H, Segal S et al (1984) The differential expression of H-2 K versus H-2D antigens, distinguishing high- metastatic from low- metastatic clones, is correlated with the immunogenic properties of the tumor cells. Int J Cancer 34:567–573
Plaksin D, Gelber C, Feldman M, Eisenbach L (1988) Reversal of the metastatic phenotype in Lewis lung carcinoma cells after transfection with syngeneic H-2 Kb gene. Proc Natl Acad Sci USA 85:4463–4467
Porgador A, Feldman M, Eisenbach L (1989) H-2 Kb transfection of B16 melanoma cells results in reduced tumorigenicity and metastatic competence. J Immunogenet 16:291–303
Lutz MB, Kukutsch N, Ogilvie AL, Rössner S, Koch F et al (1999) An advanced culture method for generating large quantities of highly pure dendritic cells from mouse bone marrow. J Immunol Methods 223:77–92
DeNardo DG, Andreu P, Coussens LM (2010) Interactions between lymphocytes and myeloid cells regulate pro- versus anti-tumor immunity. Cancer Metastasis Rev 29:309–316
Kosmaczewska A, Ciszak L, Potoczek S, Frydecka I (2008) The significance of Treg cells in defective tumor immunity. Arch Immunol Ther Exp (Warsz) 56:181–191
Curiel T (2007) Tregs and rethinking cancer immunotherapy. J Clin Investig 117:1167–1174
Zou W (2006) Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol 6:295–307
Den Brok MHMGM, Sutmuller RPM, van der Voort R, Bennink EJ, Figdor CG et al (2004) In situ tumor ablation creates an antigen source for the generation of antitumor immunity. Cancer Res 64:4024–4029
Dougan M, Dranoff G (2009) Immune therapy for cancer. Annu Rev Immunol 27:83–117
Krieg AM (2008) Toll-like receptor 9 (TLR9) agonists in the treatment of cancer. Oncogene 27:161–167
Klinman DM (2004) Immunotherapeutic uses of CpG oligodeoxynucleotides. Nat Rev Immunol 4:249–258
Figdor CG, de Vries IJM, Lesterhuis WJ, Melief CJM (2004) Dendritic cell immunotherapy: mapping the way. Nat Med 10:475–480
Palucka K, Banchereau J (2012) Cancer immunotherapy via dendritic cells. Nat Rev Cancer 12:265–277
Ueno H, Schmitt N, Klechevsky E, Pedroza-Gonzalez A, Matsui T et al (2010) Harnessing human dendritic cell subsets for medicine. Immunol Rev 234:199–212
Parmiani G, De Filippo A, Novellino L, Castelli C (2007) Unique human tumor antigens: immunobiology and use in clinical trials. J Immunol 178:1975–1979
Bonifaz LC, Bonnyay DP, Charalambous A, Darguste DI, Fujii S-I et al (2004) In vivo targeting of antigens to maturing dendritic cells via the DEC-205 receptor improves T cell vaccination. J Exp Med 199:815–824
Nierkens S, den Brok MH, Roelofsen T, Wagenaars JA, Figdor CG et al (2009) Route of administration of the TLR9 agonist CpG critically determines the efficacy of cancer immunotherapy in mice. PLoS ONE 4:e8368
Nierkens S, den Brok MH, Sutmuller RPM, Grauer OM, Bennink E et al (2008) In vivo colocalization of antigen and CpG [corrected] within dendritic cells is associated with the efficacy of cancer immunotherapy. Cancer Res 68:5390–5396
Vignali DA, Collison LW, Workman CJ (2008) How regulatory T cells work. Nat Rev Immunol 8:523–532
Read S, Malmström V, Powrie F (2000) Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25(+)CD4(+) regulatory cells that control intestinal inflammation. J Exp Med 192:295–302
Marabelle A, Kohrt H, Sagiv-Barfi I, Ajami B, Axtell RC et al (2013) Depleting tumor-specific Tregs at a single site eradicates disseminated tumors. J Clin Investig 123:2447–2463
Sasaki K, Pardee AD, Okada H, Storkus WJ (2008) IL-4 inhibits VLA-4 expression on Tc1 cells resulting in poor tumor infiltration and reduced therapy benefit. Eur J Immunol 38:2865–2873
Fuertes MB, Kacha AK, Kline J, Woo S-R, Kranz DM et al (2011) Host type I IFN signals are required for antitumor CD8 + T cell responses through CD8{alpha} + dendritic cells. J Exp Med 208:2005–2016
Schmidt C (2007) Clinical setbacks for toll-like receptor 9 agonists in cancer. Nat Biotechnol 25:825–826
Jarrossay D, Napolitani G, Colonna M, Sallusto F, Lanzavecchia A (2001) Specialization and complementarity in microbial molecule recognition by human myeloid and plasmacytoid dendritic cells. Eur J Immunol 31:3388–3393
Nierkens S, den Brok MH, Garcia Z, Togher S, Wagenaars J et al (2011) Immune adjuvant efficacy of CpG oligonucleotide in cancer treatment is founded specifically upon TLR9 function in plasmacytoid dendritic cells. Cancer Res 71:6428–6437
Acknowledgments
The authors thank Hagai Tavori for generating the D122-luc-5.5 clone. Israel Science Foundation, the Lewis Family Charitable Trust, and a research grant from the estate of John Hunter (to L. Eisenbach). L. Eisenbach is the incumbent of the George F. Duckwitz Chair of Cancer Research.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alteber, Z., Azulay, M., Cafri, G. et al. Cryoimmunotherapy with local co-administration of ex vivo generated dendritic cells and CpG-ODN immune adjuvant, elicits a specific antitumor immunity. Cancer Immunol Immunother 63, 369–380 (2014). https://doi.org/10.1007/s00262-014-1520-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-014-1520-4