Abstract
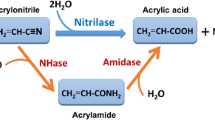
The nhhBAG gene of Rhodococcus rhodochrous M33 that encodes nitrile hydratase (NHase), converting acrylonitrile into acrylamide, was cloned and expressed in Corynebacterium glutamicum under the control of an ilvC promoter. The specific enzyme activity in recombinant C. glutamicum cells was about 13.6 μmol/min/mg dry cell weight (DCW). To overexpress the NHase, five types of plasmid variants were constructed by introducing mutations into 80 nucleotides near the translational initiation region (TIR) of nhhB. Of them, pNBM4 with seven mutations showed the highest NHase activity, exhibiting higher expression levels of NhhB and NhhA than wild-type pNBW33, mainly owing to decreased secondary-structure stability and an introduction of a conserved Shine-Dalgarno sequence in the translational initiation region. In a fed-batch culture of recombinant Corynebacterium cells harboring pNBM4, the cell density reached 53.4 g DCW/L within 18 h, and the specific and total enzyme activities were estimated to be 37.3 μmol/min/mg DCW and 1,992 μmol/min/mL, respectively. The use of recombinant Corynebacterium cells for the production of acrylamide from acrylonitrile resulted in a conversion yield of 93 % and a final acrylamide concentration of 42.5 % within 6 h when the total amount of fed acrylonitrile was 456 g.





Similar content being viewed by others
References
Brennan BA, Alms G, Nelson MJ, Durney LT, Scarrow RC (1996) Nitrile hydratase from Rhodococcus rhodochrous J1 contains a non-corrin cobalt ion with two sulfur ligands. J Am Chem Soc 118:9194–9195
Date M, Itaya H, Matsui H, Kikuchi Y (2006) Secretion of human epidermal growth factor by Corynebacterium glutamicum. Lett Appl Microbiol 42:66–70
de Smit MH, van Duin J (1994) Control of translation by mRNA secondary structure in Escherichia coli. A quantitative analysis of literature data. J Mol Biol 244:144–150
Hermann T (2003) Industrial production of amino acids by coryneform bacteria. J Biotechnol 104:155–172
Jana S, Deb JK (2005) Strategies for efficient production of heterologous proteins in Escherichia coli. Appl Microbiol Biotechnol 67:289–298
Kim S, Oriel P (2000) Cloning and expression of the nitrile hydratase and amidase genes from Bacillus sp. BR449 into Escherichia coli. Enzyme Microb Technol 27:492–501
Kim BY, Kim JC, Lee HH, Hyun HH (2001) Fed-batch fermentation for production of nitrile hydratase by Rhodococcus rhodochrous M33. Biotechnol Bioproc Eng 6:11–17
Kobayashi M, Shimizu S (1998) Metalloenzyme nitrile hydratase: structure, regulation, and application to biotechnology. Nat Biotechnol 16:733–736
Kobayashi M, Nishiyama M, Nagasawa T, Horinouchi S, Beppu T, Yamada H (1991) Cloning, nucleotide sequence, and expression in Escherichia coli of two cobalt-containing nitrile hydratase genes from Rhodococcus rhodochrous J1. Biochim Biophys Acta 1129:23–33
Komeda H, Kobayashi M, Shimizu S (1996) Characterization of the gene cluster of high-molecular-mass nitrile hydratase (H-NHase) induced by its reaction product in Rhodococcus rhodochrous Jl. Proc Natl Acad Sci U S A 93:4267–4272
Komeda H, Kobayashi M, Shimizu S (1997) A novel transporter involved in cobalt uptake. Proc Natl Acad Sci U S A 94:36–41
Kudla G, Murray AW, Tollervey D, Plotkin JB (2009) Coding-sequence determinants of gene expression in Escherichia coli. Science 324:255–258
Liebeton K, Eck J (2004) Identification and expression in E. coli of novel nitrile hydratases from the metagenome. Eng Life Sci 4:557–562
Martín JF, Barreiro C, González-Lavado E, Barriuso M (2003) Ribosomal RNA and ribosomal proteins in Corynebacteria. J Biotechnol 104:41–53
Myagchenkov VA, Kurenkov VF (1991) Applications of acrylamide polymers and copolymers—a review. Polym Plast Technol 30(2–3):109–135
Nagasawa T, Takeuchi K, Nardi-Dei V, Mihara Y, Yamada H (1991) Optimum culture conditions for the production of cobalt-containing nitrile hydratase by Rhodococcus rhodochrous J1. Appl Microbiol Biotechnol 34:783–788
Nagasawa T, Shimizu H, Yamada H (1993) The superiority of the third-generation catalyst, Rhodococcus rhodochrous J1 nitrile hydratase, for industrial production of acrylamide. Appl Microbiol Biotechnol 40:189–195
Nesvera J, Patek M (2011) Tools for genetic manipulations in Corynebacterium glutamicum and their applications. Appl Microbiol Biotechnol 90(5):1641–1654
Neupert J, Karcher D, Bock R (2008) Design of simple synthetic RNA thermometers for temperature-controlled gene expression in Escherichia coli. Nucleic Acids Res 36:e124
Nojiri M, Yohda M, Odaka M, Matsushita Y, Tsujimura M, Yoshida T, Dohmae N, Takio K, Endo I (1999) Functional expression of nitrile hydratase in Escherichia coli: requirement of a nitrile hydratase activator and post-translational modification of a ligand cysteine. J Biochem 125:696–704
Ogawa J, Shimizu S (2002) Industrial microbial enzymes: their discovery by screening and use in large-scale production of useful chemicals in Japan. Curr Opin Biotechnol 13(4):367–375
Park YS, Seo SW, Hwang S, Chu HS, Ahn JH, Kim TW, Kim DM, Jung GY (2007) Design of 5'-untranslated region variants for tunable expression in Escherichia coli. Biochem Biophys Res Commun 356:136–141
Park JU, Jo JH, Kim YJ, Chung SS, Lee JH, Lee HH (2008) Construction of heat-inducible expression vector of Corynebacterium glutamicum and C. ammoniagenes: fusion of lambda operator with promoters isolated from C. ammoniagenes. J Microbiol Biotechnol 18:639–647
Patek M, Eikmanns BJ, Patek J, Sahm H (1996) Promoters from Corynebacterium glutamicum: cloning, molecular analysis and search for a consensus motif. Microbiology 142:1297–1309
Prasad S, Bhalla TC (2010) Nitrile hydratases (NHase): at the interface of academia and industry. Biotechnol Adv 28:725–741
Salis HM, Mirsky EA, Voigt CA (2009) Automated design of synthetic ribosome binding sites to control protein expression. Nat Biotechnol 27:946–950
Schneider J, Eberhardt D, Wendisch VF (2012) Improving putrescine production by Corynebacterium glutamicum by fine-tuning ornithine transcarbamoylase activity using a plasmid addiction system. Appl Microbiol Biotechnol 95(1):169–178
Seo SW, Yang J, Jung GY (2009) Quantitative correlation between mRNA secondary structure around the region downstream of the initiation codon and translational efficiency in Escherichia coli. Biotechnol Bioeng 104:611–616
Srivastava P, Deb JK (2005) Gene expression systems in corynebacteria. Protein Expr Purif 40(2):221–229
Toru J, Masayuki I, Hideaki Y (2012) Biorefinery applications of Corynebacterium glutamicum. In: Tatsumi N, Inui M (eds) Corynebacterium glutamicum: biology and biotechnology. Springer, London, pp 149–172
Tsao D, Shabalina SA, Gauthier J, Dokholyan NV, Diatchenko L (2011) Disruptive mRNA folding increases translational efficiency of catechol-O-methyltransferase variant. Nucleic Acids Res 39:6201–6212
Van der Rest ME, Lange C, Molenaar D (1999) A heat shock following electroporation induces highly efficient transformation of Corynebacterium glutamicum with xenogeneic plasmid DNA. Appl Microbiol Biotechnol 52:541–545
Wieschalka S, Blombach B, Bott M, Eikmanns BJ (2013) Bio-based production of organic acids with Corynebacterium glutamicum. Microb Biotechnol 6(2):87–102
Yamada H, Kobayashi M (1996) Nitrile hydratase and its application to industrial production of acrylamide. Biosci Biotechnol Biochem 60:1391–1400
Yanenko et al. (1998) Strain of R. rhodochrous as a producer of nitrile hydratase. US Patent 5827699
Zuker M (2003) Mfold Web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res 31:3406–3415
Acknowledgments
This work was financially supported by Tongsuh Petrochemical Corp., Ltd.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Mi-Suk Kang and Sang-Soo Han equally contributed to this work.
Rights and permissions
About this article
Cite this article
Kang, MS., Han, SS., Kim, MY. et al. High-level expression in Corynebacterium glutamicum of nitrile hydratase from Rhodococcus rhodochrous for acrylamide production. Appl Microbiol Biotechnol 98, 4379–4387 (2014). https://doi.org/10.1007/s00253-014-5544-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-5544-7