Abstract
Rationale
Mazindol, an appetite suppressant, inhibits the reuptake of dopamine in the synaptic cleft. It has been considered that mazindol might enhance dopamine transmission in the human brain. However, there has been no study that investigated the extracellular dopamine concentration in vivo.
Objective
Using positron emission tomography (PET), we aimed to measure the effect of mazindol on the extracellular dopamine concentration and to evaluate how mazindol affects the dopamine system in the healthy human brain.
Methods
Eleven healthy individuals (six males, five females, age 30.9 ± 4.9 years) were enrolled in this study. Each participant was scanned with [11C]raclopride on 1 day without any medicine as baseline condition, and on another day with mazindol as drug condition. In the drug condition, participants took mazindol 0.5 mg (N = 5) or 1.5 mg (N = 6) 2 h before the PET scan. Plasma concentrations of mazindol were measured before the injection of [11C]raclopride, and urine concentrations of mazindol were measured after the scan.
Results
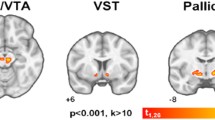
After taking mazindol, the calculated decrease in binding potential (ΔBP) in the striatum was 1.74 % for 0.5 mg and 8.14 for 1.5 mg, and the correlation with the blood concentration of mazindol was significant (P = 0.0016, R 2 = 0.69). ΔBP was not significantly correlated with the urine concentration of mazindol (P = 0.84, R 2 = 0.005).
Conclusions
Mazindol increased the extracellular concentration of dopamine in the human brain, and its effect was dose dependent. A single administration of mazindol, even at usual dosage, elevated dopamine concentration similarly to other addictive drugs, suggesting that the risk of dependence may increase with the mazindol dose.


Similar content being viewed by others
References
Carboni E, Imperato A, Perezzani L, Di Chiara G (1989) Amphetamine, cocaine, phencyclidine and nomifensine increase extracellular dopamine concentrations preferentially in the nucleus accumbens of freely moving rats. Neuroscience 28(3):653–651
Carruba MO, Picotti GB, Zambotti F, Mantegazza P (1977) Mazindol and amphetamine as inhibitors of the uptake and release of 3H-dopamine by rat striatal synaptosomes. Naunyn Schmiedebergs Arch Pharmacol 298(1):1–5
Engstrom RG, Kelly LA, Gogerty JH (1975) The effects of 5-hydroxy-5(4'-chlorophenyl)-2, 3-dihydro-5H-imidazo (2, 1-a) isoindole (mazindol, SaH 42–548) on the metabolism of brain norepinephrine. Arch Int Pharmacodyn Ther 214(2):308–321
Farde L, Hall H, Ehrin E, Sedvall G (1986) Quantitative analysis of D2 dopamine receptor binding in the living human brain by PET. Science 231(4735):258–261
Innis RB, Cunningham VJ, Delforge J, Fujita M, Gjedde A, Gunn RN, Holden J, Houle S, Huang SC, Ichise M, Iida H, Ito H, Kimura Y, Koeppe RA, Knudsen GM, Knuuti J, Lammertsma AA, Laruelle M, Logan J, Maguire RP, Mintun MA, Morris ED, Parsey R, Price JC, Slifstein M, Sossi V, Suhara T, Votaw JR, Wong DF, Carson RE (2007) Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J Cereb Blood Flow Metab 27(9):1533–1539
Ito K, Hayashi Y, Nishi N, Maki Y, Asano Y, Kikuiri T, Ogino S (1995) A case of the appearance of psychiatric symptoms upon the use of mazindol (syokuyokuyokuseizai (mazindol) touyo ga seishinnsyoujyou hatsugen ni kannyo shita to omowareru ichi syourei). Clin Psychiatry (Jpn) 37:519–521
Ito H, Sudo Y, Suhara T, Okubo Y, Halldin C, Farde L (2001) Error analysis for quantification of [11C]FLB 457 binding to extrastriatal D2 dopamine receptors in the human brain. Neuroimaging 13(3):531–539
Kaneko S, Harima H, Wakejima T, Okazaki Y (2008) A case of abuse of mazindol, administered as alternative to stimulants, resulting in recurrence of hallucination and delusion (kakuseizai daitai to shite mazindol wo rannyou shi gennkakumousou no sainenn wo teishita kakuseizaiseishinbyou no ichi rei). Psychiatry (Jpn) 12(6):1347–4790
Krumholz WV, White L (1970) Clinical evaluation of Mazindol in chronic schizophrenics. Curr Ther Res Clin Exp 12(9):609–610
Kurihara M, Hirose T, Henmi T, Mori A, Yanagida T (1985) A study of the problem of dependency of Mazindol –A clinical open survey. Clin Eval 13:517–547
Lammertsma AA, Hume SP (1996) Simplified reference tissue model for PET receptor studies. Neuroimaging 4(3 Pt 1):153–158
Laruelle M (2000) Imaging synaptic neurotransmission with in vivo binding competition techniques: a critical review. J Cereb Blood Flow Metab 20(3):423–451
Minami T, Oomura Y, Sugimori M, Hynes M (1985) Direct action of mazindol on guinea-pig ventromedial hypothalamic neurons: intracellular studies in slice preparation. Brain Res Bull 15(1):29–31
Mozley LH, Gur RC, Mozley PD, Gur RE (2001) Striatal dopamine transporters and cognitive functioning in healthy men and women. Am J Psychiatry 158(9):1492–1499
Nakajima N, Hayashi H, Takahashi K, Taylor P, Takahashi S, Hashimoto K (1986) Chronic toxicity test of mazindol in rats by oral 26-week administration. A Clin Rep (Jpn) 20(4):2243–2278
Narendran R, Mason NS, Laymon CM, Lopresti BJ, Velasquez ND, May MA, Kendro S, Martinez D, Mathis CA, Frankle WG (2010) A comparative evaluation of the dopamine D2/3 agonist radiotracer [11C]( )-N-propyl-norapomorphine and antagonist [11C]raclopride to measure amphetamine-induced dopamine release in the human striatum. J Pharmacol Exp Ther 333(2):533–539
Parker EM, Cubeddu LX (1988) Comparative effects of amphetamine, phenylethylamine and related drugs on dopamine efflux, dopamine uptake and mazindol binding. J Pharmacol Exp Ther 245(1):199–210
Sikdar SK, Oomura Y, Inokuchi A (1985) Effects of mazindol on rat lateral hypothalamic neurons. Brain Res Bull 15(1):33–38
Skinbjerg M, Liow JS, Seneca N, Hong J, Lu S, Thorsell A, Heilig M, Pike VW, Halldin C, Sibley DR, Innis RB et al (2010) D2 dopamine receptor internalization prolongs the decrease of radioligand binding after amphetamine: a PET study in a receptor internalization-deficient mouse model. Neuroimaging 50(4):1402–1407
Suhara T, Sudo Y, Okauchi T, Maeda J, Kawabe K, Suzuki K, Okubo Y, Nakashima Y, Ito H, Tanada S, Halldin C, Farde L (1999) Extrastriatal dopamine D2 receptor density and affinity in the human brain measured by 3D PET. Int J Neuropsychopharmacol 2(2):73–82
Takahashi H, Fujimura Y, Hayashi M, Takano H, Kato M, Okubo Y, Kanno I, Ito H, Suhara T (2008) Enhanced dopamine release by nicotine in cigarette smokers: a double-blind, randomized, placebo-controlled pilot study. Int J Neuropsychopharmacol 11(3):413–417
Volkow ND, Fowler JS, Wang GJ (2002) Role of dopamine in drug reinforcement and addiction in humans: results from imaging studies. Behav Pharmacol 13(5–6):355–366
Wand GS, Oswald LM, McCaul ME, Wong DF, Johnson E, Zhou Y, Kuwabara H, Kumar A (2007) Association of amphetamine-induced striatal dopamine release and cortisol responses to psychological stress. Neuropsychopharmacology 32(11):2310–2320
Yanagida T, Wakasa Y, Oinuma N (1982) Drug dependence potential of mazindol tested in rhesus monkeys. Preclin Rep Cent Inst Exp Anim (Jpn) 8(3):247–257
Acknowledgments
This work was partially supported by a grant from the Ministry of Education, Culture, Sports, Science and Technology (MEXT, Japan). We thank Minoru Sakurai, Koji Nagaya, Koji Kanaya, Masaya Suda, Megumi Takei, and Kazuyoshi Honjo (Clinical Imaging Centre for Healthcare, Nippon Medical School) for their assistance in performing MRI and PET examinations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakayori, T., Tateno, A., Arakawa, R. et al. Effect of mazindol on extracellular dopamine concentration in human brain measured by PET. Psychopharmacology 231, 2321–2325 (2014). https://doi.org/10.1007/s00213-013-3392-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-013-3392-2