Abstract
Rationale
Several studies have documented impairments in memory processes as a result of ketamine administration; however, few studies have compared the profile of cognitive effects of ketamine to other drugs.
Objectives
The aim of this study was to compare the cognitive effects of ketamine with those of triazolam in healthy volunteers.
Methods
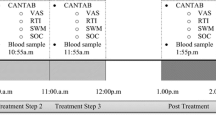
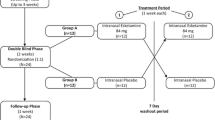
Doses of ketamine (0.2, 0.4 mg/kg intramuscular (i.m.)), triazolam (0.2, 0.4 mg/70 kg p.o.), and double-dummy placebos were administered to 20 volunteers under repeated measures, counterbalanced, double-blind conditions. Peak physiological, psychomotor, subjective, and cognitive effects were examined.
Results
Ketamine impaired balance when balance was assessed early in the task order, whereas triazolam impaired psychomotor coordination and divided attention irrespective of task order. Triazolam also tended to produce greater effects on working memory and episodic memory tasks than ketamine at doses that produced lower subjective effects and higher estimates of performance.
Conclusions
Ketamine produces less cognitive impairment than triazolam at doses that produced greater subjective effects. Thus ketamine does not produce the underestimation of cognitive impairment typically seen with triazolam.




Similar content being viewed by others
References
Bowdle TA, Radant AD, Cowley DS, Kharasch ED, Strassman RJ, Roy-Byrne PP (1998) Psychedelic effects of ketamine in healthy volunteers: relationship to steady-state plasma concentrations. Anesthesiology 88:82–88
Carter LP, Griffiths RR (2009) Principles of laboratory assessment of drug abuse liability and implications for clinical development. Drug Alcohol Depend 105(Suppl 1):S14–S25
Carter LP, Griffiths RR, Mintzer MZ (2009) Cognitive, psychomotor, and subjective effects of sodium oxybate and triazolam in healthy volunteers. Psychopharmacol (Berl) 206:141–154
Carter LP, Richards BD, Mintzer MZ, Griffiths RR (2006) Relative abuse liability of GHB in humans: a comparison of psychomotor, subjective, and cognitive effects of supratherapeutic doses of triazolam, pentobarbital, and GHB. Neuropsychopharmacol 31:2537–2551
deWit H, Pierri J, Johanson CE (1989) Reinforcing and subjective effects of diazepam in nondrug-abusing volunteers. Pharmacol Biochem Behav 33:205–213
Goodman LA, Kruskal WH (1954) Measures of association for cross-classifications. JASA 49:732–764
Honey GD, Honey RA, O’Loughlin C, Sharar SR, Kumaran D, Suckling J, Menon DK, Sleator C, Bullmore ET, Fletcher PC (2005) Ketamine disrupts frontal and hippocampal contribution to encoding and retrieval of episodic memory: an fMRI study. Cereb Cortex 15:749–759
Honey GD, O’loughlin C, Turner DC, Pomarol-Clotet E, Corlett PR, Fletcher PC (2006) The effects of a subpsychotic dose of ketamine on recognition and source memory for agency: implications for pharmacological modelling of core symptoms of schizophrenia. Neuropsychopharmacology 31:413–423
Hood RW Jr, Ghorbani N, Watson PJ, Ghramaleki AF, Bing MN, Davison HK, Morris RJ, Williamson WP (2001) Dimensions of the mysticism scale: confirming the three-factor structure in the United States and Iran. J Sci Study Relig 40:691–705
Johnson M, Richards W, Griffiths R (2008) Human hallucinogen research: guidelines for safety. J Psychopharmacol 22:603–620
Jones HE, Garrett BE, Griffiths RR (2001) Reinforcing effects of oral cocaine: contextual determinants. Psychopharmacol (Berl) 154:143–152
Keppel G (1991) Design and analysis: a researcher’s handbook, 3rd edn. Prentice Hall, Englewood Cliffs, NJ
Kleykamp BA, Griffiths RR, Mintzer MZ (2010) Dose effects of triazolam and alcohol on cognitive performance in healthy volunteers. Exp Clin Psychopharmacol 18:1–16
Koriat A, Goldsmith M (1996) Monitoring and control processes in the strategic regulation of memory accuracy. Psychol Rev 103:490–517
Krystal JH, Karper LP, Bennett A, D’Souza DC, Abi-Dargham A, Morrissey K, Abi-Saab D, Bremner JD, Bowers MB Jr, Suckow RF, Stetson P, Heninger GR, Charney DS (1998) Interactive effects of subanesthetic ketamine and subhypnotic lorazepam in humans. Psychopharmacol (Berl) 135:213–229
Krystal JH, Perry EB Jr, Gueorguieva R, Belger A, Madonick SH, Abi-Dargham A, Cooper TB, Macdougall L, Abi-Saab W, D’Souza DC (2005) Comparative and interactive human psychopharmacologic effects of ketamine and amphetamine: implications for glutamatergic and dopaminergic model psychoses and cognitive function. Arch Gen Psychiatry 62:985–994
LaPorte DJ, Blaxton TA, Michaelidis T, Robertson DU, Weiler MA, Tamminga CA, Lahti AC (2005) Subtle effects of ketamine on memory when administered following stimulus presentation. Psychopharmacol (Berl) 180:385–390
Lofwall MR, Griffiths RR, Mintzer MZ (2006) Cognitive and subjective acute dose effects of intramuscular ketamine in healthy adults. Exp Clin Psychopharmacol 14:439–449
McLeod DR, Griffiths RR, Bigelow GE, Yingling J (1982) An automated version of the digit symbol substitution test (DSST). Behav Res Methods Instrum 14:463–466
Mintzer MZ, Frey JM, Yingling JE, Griffiths RR (1997) Triazolam and zolpidem: a comparison of their psychomotor, cognitive, and subjective effects in healthy volunteers. Behav Pharmacol 8:561–574
Mintzer MZ, Griffiths RR (2002) Alcohol and triazolam: differential effects on memory, psychomotor performance and subjective ratings of effects. Behav Pharmacol 13:653–658
Mintzer MZ, Griffiths RR (2003a) Lorazepam and scopolamine: a single-dose comparison of effects on human memory and attentional processes. Exp Clin Psychopharmacol 11:56–72
Mintzer MZ, Griffiths RR (2003b) Triazolam–amphetamine interaction: dissociation of effects on memory versus arousal. J Psychopharmacol 17:17–29
Mintzer MZ, Griffiths RR (2007a) A triazolam/amphetamine dose-effect interaction study: dissociation of effects on memory versus arousal. Psychopharmacol (Berl) 192:425–440
Mintzer MZ, Griffiths RR (2007b) Differential effects of scopolamine and lorazepam on working memory maintenance versus manipulation processes. Cogn Affect Behav Neurosci 7:120–129
Mintzer MZ, Kleykamp BA, Griffiths RR (2010) Dose effects of triazolam and scopolamine on metamemory. Exp Clin Psychopharmacol 18:17–31
Morgan CJ, Mofeez A, Brandner B, Bromley L, Curran HV (2004a) Ketamine impairs response inhibition and is positively reinforcing in healthy volunteers: a dose–response study. Psychopharmacol (Berl) 172:298–308
Morgan CJ, Muetzelfeldt L, Curran HV (2010) Consequences of chronic ketamine self-administration upon neurocognitive function and psychological wellbeing: a 1-year longitudinal study. Addiction 105:121–133
Morgan CJ, Riccelli M, Maitland CH, Curran HV (2004b) Long-term effects of ketamine: evidence for a persisting impairment of source memory in recreational users. Drug Alcohol Depend 75:301–308
Muetzelfeldt L, Kamboj SK, Rees H, Taylor J, Morgan CJ, Curran HV (2008) Journey through the K-hole: phenomenological aspects of ketamine use. Drug Alcohol Depend 95:219–229
Mumford GK, Rush CR, Griffiths RR (1995) Abecarnil and alprazolam in humans: behavioral, subjective and reinforcing effects. J Pharmacol Exp Ther 272:570–580
Nelson TO (1984) A comparison of current measures of the accuracy of feeling-of-knowing predictions. Psychol Bull 95:109–133
Parwani A, Weiler MA, Blaxton TA, Warfel D, Hardin M, Frey K, Lahti AC (2005) The effects of a subanesthetic dose of ketamine on verbal memory in normal volunteers. Psychopharmacol (Berl) 183:265–274
Rowland LM, Astur RS, Jung RE, Bustillo JR, Lauriello J, Yeo RA (2005) Selective cognitive impairments associated with NMDA receptor blockade in humans. Neuropsychopharmacology 30:633–639
Rush CR, Frey JM, Griffiths RR (1999) Zaleplon and triazolam in humans: acute behavioral effects and abuse potential. Psychopharmacology 145:39–51
Snodgrass JG, Corwin J (1988) Pragmatics of measuring recognition memory: applications to dementia and amnesia. J Exp Psychol Gen 117:34–50
Sternberg S (1969) The discovery of processing stages: extensions of Donder’s method. Acta Psychologica 30:276–315
Strassman RJ, Qualls CR, Uhlenhuth EH, Kellner R (1994) Dose response study of N, N-dimethyltryptamine in humans. II. Subjective effects and preliminary results of a new rating scale. Arch Gen Psychiatry 51:98–108
Acknowledgments
The authors thank Crystal Barnhouser, Julia Kane, and John Yingling for technical assistance, and Paul Nuzzo and Linda Felch for data analysis. This study was supported by National Institute on Drug Abuse Research Grants R01 DA03889 and T32 DA07209
Conflict of interest
In the past 3 years, Lawrence Carter has served as a Special Government Employee for FDA and as a consultant for Dan’s Plan, LLC, Jazz Pharmaceuticals, Inc., KemPharm, Inc., and UCB, SA. He is currently employed by Jazz Pharmaceuticals, Inc. and owns stock options in the company. During the past 3 years, on issues related to drug abuse liability, Roland Griffiths has been a consultant to or has received contracts or grants from: Abbott Laboratories, Alexza Pharmaceuticals, Bristol-Myers Squibb, Hoffman-La Roche Inc., Jazz Pharmaceuticals, Merck & Co., Neurocrine Biosciences, Novartis, Pharmacia Corporation, Pfizer, Sanofi-Aventis, Somaxon Pharmaceuticals, Transcept Pharmaceuticals Inc., Vanda Pharmaceuticals, and Wyeth Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Carter, L.P., Kleykamp, B.A., Griffiths, R.R. et al. Cognitive effects of intramuscular ketamine and oral triazolam in healthy volunteers. Psychopharmacology 226, 53–63 (2013). https://doi.org/10.1007/s00213-012-2883-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-012-2883-x